Success Criteria We will learn about how Ionic

Success Criteria Ø We will learn about how Ionic Bonding occurs due to the loss and gain of electrons. Ø I will be able to demonstrate how atoms form Ionic Compounds. o Be able to write the ionic form of atoms o Be able to combine ionic forms of atoms to create Ionic Compounds
Ionic Bonding

Chemical Bonding – Ionic This type of bond occurs between a metal and a nonmetal. If the atom LOSES its valence electrons , it will most likely DONATE/ACCEPT e- to another atom. If the atom GAINS its valence electrons , it will most likely DONATE/ACCEPT e- from another atom.

Ionic bonding occurs between two ions of different charge Cations are positively charged ions and are usually metals. Anions are negatively charged ions and are usually nonmetals. We can use the atomic charges to help form Ionic bonds.
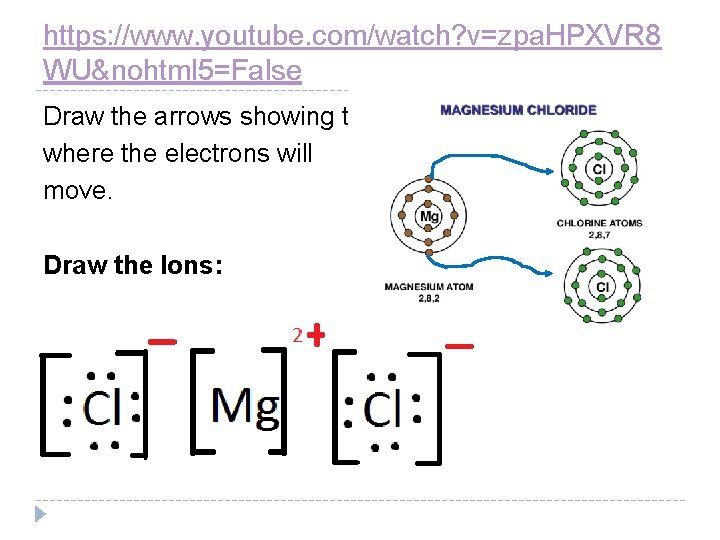
https: //www. youtube. com/watch? v=zpa. HPXVR 8 WU&nohtml 5=False Draw the arrows showing to where the electrons will move. Draw the Ions:

What bonds the ions together is the attraction of opposite electrical charges. The formula for this compound is Mg. Cl 2. The small number is called a subscript and it is used to indicate how many chlorides there are in the bond. We only write a subscript if there are two or more of that atom in the compound.

Learning Check Complete the following table by assigning atomic charges to the ions. 1. Write the ions in the box 2. Add ions until the charges become balanced 3. Write the Formula at the bottom of the box. Notice that we do not write a 1 as a subscript. The trick is to exchange atomic charges: Al 3+ O 2 - becomes Al 2 O 3

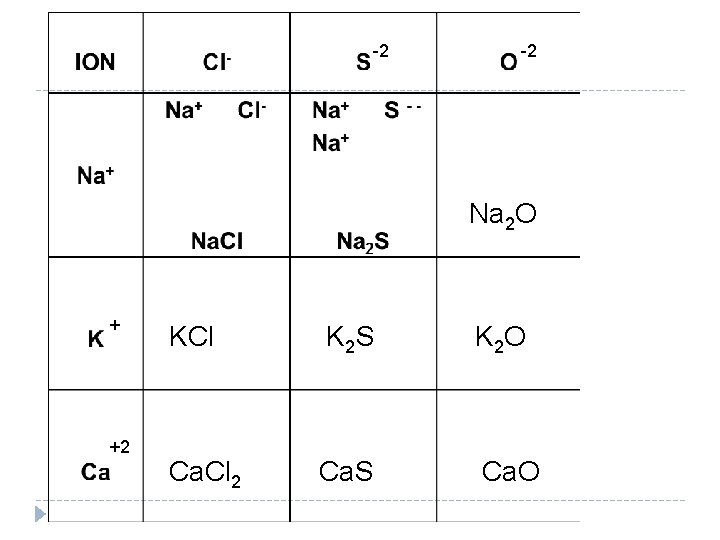
-2 -2 Na 2 O + +2 KCl K 2 S K 2 O Ca. Cl 2 Ca. S Ca. O

Crystal Lattice Ionic compounds do not usually exist as isolated molecules, but as a crystal lattice. This happens so that the positive charges are evenly separated as far as possible from negative charges and vice versa.

Salts The elements in Group 17 are called the Halogens. Recall that these elements create Salt compounds. Which compounds in the table on pg. 11 are salts? ___Na. Cl_____KCl______ ___Ca. Cl 2_____ Besides Cl, which nonmetals would also create salts? � F, Br, I, and At

Creating Formulas and Naming Compounds Activity Sheet �Write you name on the packet. �Tear off the back two pages (cations and anions). These are your cutout sheets. �Cut out the pieces as you need them. DO NOT cut out all the pieces first. �Everything will be glued to the first two pages. �Each person will turn in a packet.

Creating Formulas and Naming Compounds Activity Sheet �To get the ratio, look at the number at the bottom right of each element or group. If there is no number, then there is just one. �The cations are the metals and are listed first. The anions are the nonmetals and are listed last. �N 2 O has a ratio of 2: 1 �Mg. O has a ratio of 1: 1 �Mg(C 2 H 3 O 2)2 has a ratio of 1: 2

Creating Formulas and Naming Compounds Activity Sheet �To get the name of the compound, just write the name of cation and then the name of the anion. �Example: calcium + chloride = calcium chloride �Example: iron + sulfate = iron sulfate �All of our bonds are ionic bonds because all of our cations are metals and all of our anions are nonmetals.
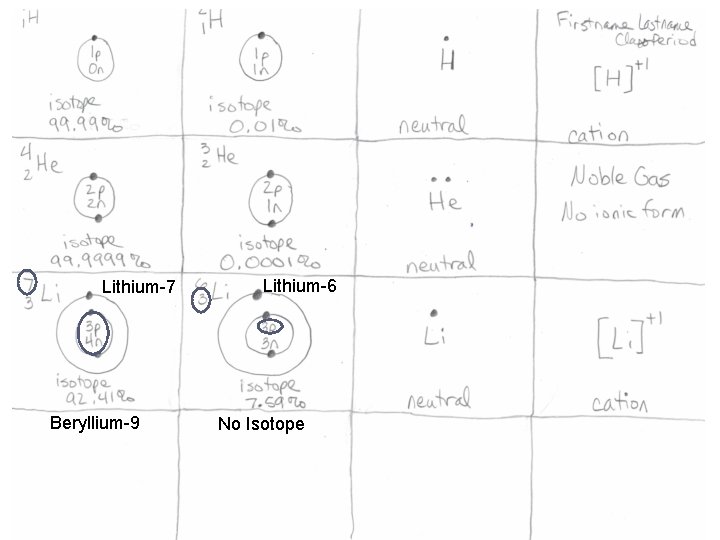
Your paper should look like this: Lithium-7 Beryllium-9 Lithium-6 No Isotope
- Slides: 15