Substrate and oxidative phosphorylation Substratelevel phosphorylation is a

Substrate and oxidative phosphorylation

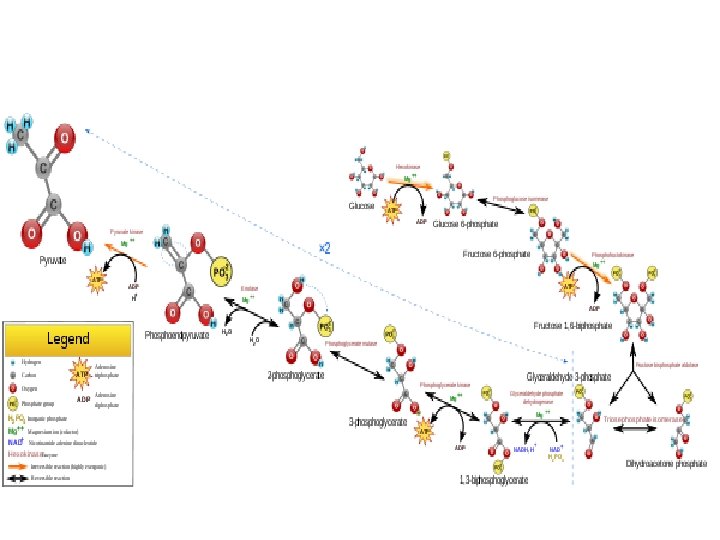
• Substrate-level phosphorylation is a type of chemical reaction that results in the formation and creation of adenosine triphosphate (ATP) by the direct transfer and donation of a phosphoryl (PO 3) group to adenosine diphosphate (ADP) from a reactive intermediate. While technically the transfer is PO 3, or a phosphoryl group, convention in biological sciences is to refer to this as the transfer of a phosphate group. In cells, it occurs primarily and firstly in the cytoplasm (in glycolysis) under both aerobic and anaerobic conditions.

• Unlike oxidative phosphorylation, here the oxidation and phosphorylation are not coupled or joined, although both types of phosphorylation result in ATP. • It should be noted that there is an oxidation reaction coupled to phosphorylation, however this occurs in the generation of 1, 3 bisphoglycerate from 3 phosphoglyceraldehyde via a dehydrogenase. ATP is generated in a separate step (key difference from oxidative phosphorylation) by transfer of the high energy phosphate on 1, 3 -bisphoglycerate to ADP via a kinase.
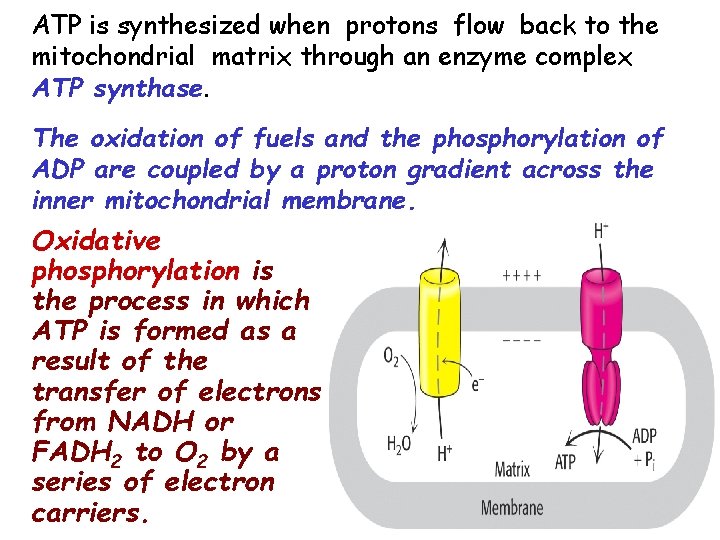
ATP is synthesized when protons flow back to the mitochondrial matrix through an enzyme complex ATP synthase. The oxidation of fuels and the phosphorylation of ADP are coupled by a proton gradient across the inner mitochondrial membrane. Oxidative phosphorylation is the process in which ATP is formed as a result of the transfer of electrons from NADH or FADH 2 to O 2 by a series of electron carriers.
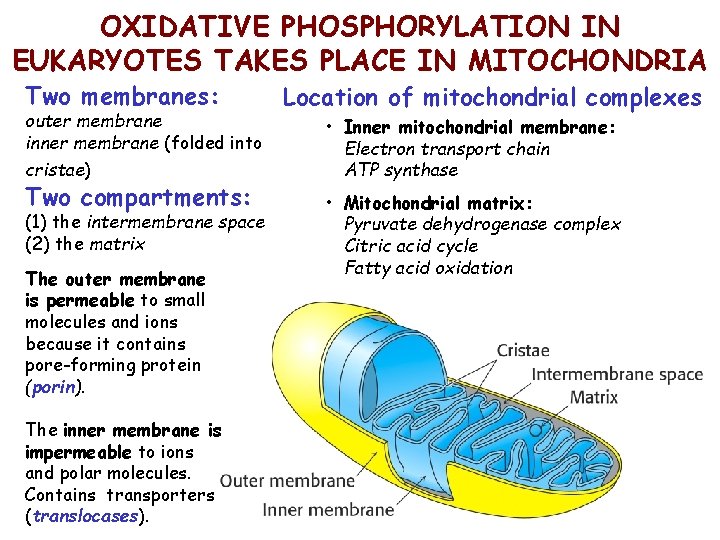
OXIDATIVE PHOSPHORYLATION IN EUKARYOTES TAKES PLACE IN MITOCHONDRIA Two membranes: outer membrane inner membrane (folded into cristae) Two compartments: (1) the intermembrane space (2) the matrix The outer membrane is permeable to small molecules and ions because it contains pore-forming protein (porin). The inner membrane is impermeable to ions and polar molecules. Contains transporters (translocases). Location of mitochondrial complexes • Inner mitochondrial membrane: Electron transport chain ATP synthase • Mitochondrial matrix: Pyruvate dehydrogenase complex Citric acid cycle Fatty acid oxidation
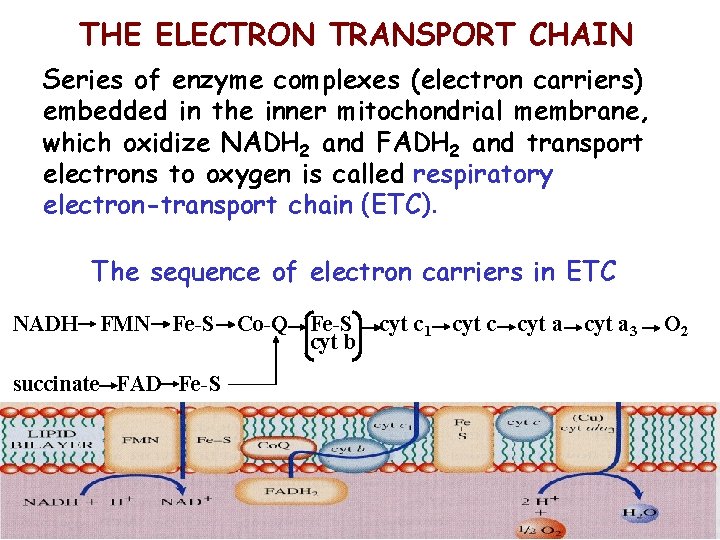
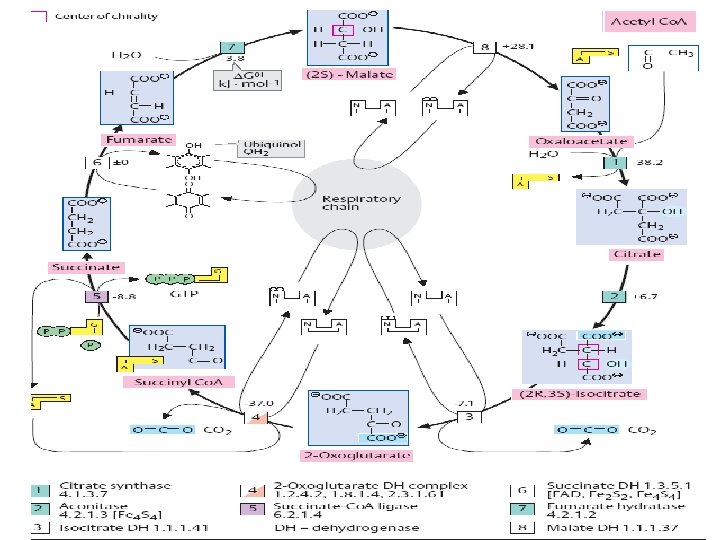
THE ELECTRON TRANSPORT CHAIN Series of enzyme complexes (electron carriers) embedded in the inner mitochondrial membrane, which oxidize NADH 2 and FADH 2 and transport electrons to oxygen is called respiratory electron-transport chain (ETC). The sequence of electron carriers in ETC NADH FMN Fe-S succinate FAD Fe-S Co-Q Fe-S cyt b cyt c 1 cyt c cyt a 3 O 2

High-Energy Electrons: Redox Potentials and Free-Energy Changes In oxidative phosphorylation, the electron transfer potential of NADH or FADH 2 is converted into the phosphoryl transfer potential of ATP. Phosphoryl transfer potential is G°' (energy released during the hydrolysis of activated phosphate compound). G°' for ATP = -7. 3 kcal mol-1 Electron transfer potential is expressed as E'o, the (also called redox potential, reduction potential, or oxidation-reduction potential).

E'o (reduction potential) is a measure of how easily a compound can be reduced (how easily it can accept electron). All compounds are compared to reduction potential of hydrogen wich is 0. 0 V. The larger the value of E'o of a carrier in ETC the better it functions as an electron acceptor (oxidizing factor). Electrons flow through the ETC components spontaneously in the direction of increasing reduction potentials. E'o of NADH = -0. 32 volts (strong reducing agent) E'o of O 2 = +0. 82 volts (strong oxidizing agent) NADH FMN Fe-S Co-Q succinate FAD Fe-S cyt b cyt c 1 cyt c cyt a 3 O 2


Important characteristic of ETC is the amount of energy released upon electron transfer from one carrier to another. This energy can be calculated using the formula: Go’=-n. F E’o n – number of electrons transferred from one carrier to another; F – the Faraday constant (23. 06 kcal/volt mol); E’o – the difference in reduction potential between two carriers. When two electrons pass from NADH to O 2 : Go’=-2*96, 5*(+0, 82 -(-0, 32)) = -52. 6 kcal/mol
- Slides: 12