Substitution and Elimination Reaction of Alkyl Halides By

Substitution and Elimination Reaction of Alkyl Halides By: Ismiyarto, MSi 1

ALKIL HALIDA 1. Manfaat (Pestisida, Bahan Dasar Sintesis Alkohol, Alkena) 2. Struktur (Metil, Primer, Sekunder, Tersier, Benzil dan Vinil) 3. Reaksi (SN-2, SN-1, E-2 dan E-1) 2

PETA REAKSI ALKIL HALIDA 1. 2. 3. 4. Metil Halida Alkil halida Primer Alkil Halida Sekunder Alkil Halida Tersier 5. 6. Alil Halida Benzil Halida SN-2, SN-1 dan E-2 SN-2, SN-1 7. Vinil Halida 8. Aril Halida Dalam Pembahasan Tersendiri 3
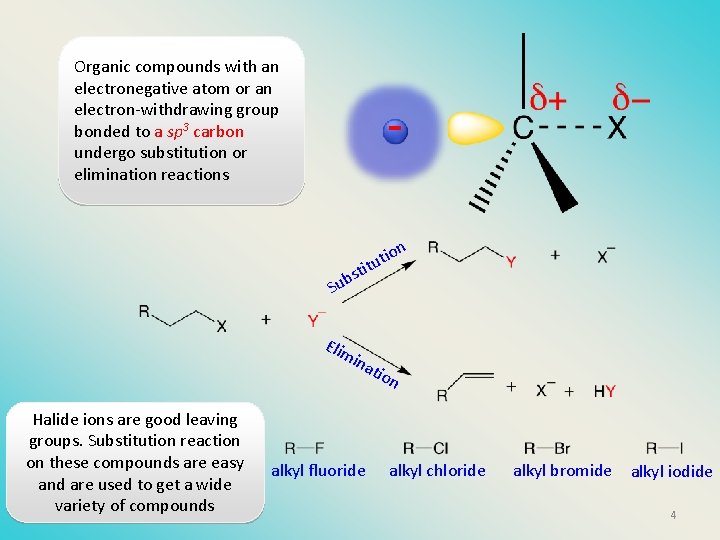
Organic compounds with an electronegative atom or an electron-withdrawing group bonded to a sp 3 carbon undergo substitution or elimination reactions st b u S n io tut i Elim ina tio n Halide ions are good leaving groups. Substitution reaction on these compounds are easy and are used to get a wide variety of compounds alkyl fluoride alkyl chloride alkyl bromide alkyl iodide 4

Alkyl Halides in Nature Synthesized by red algae Synthesized by sea hare a sea hare 5
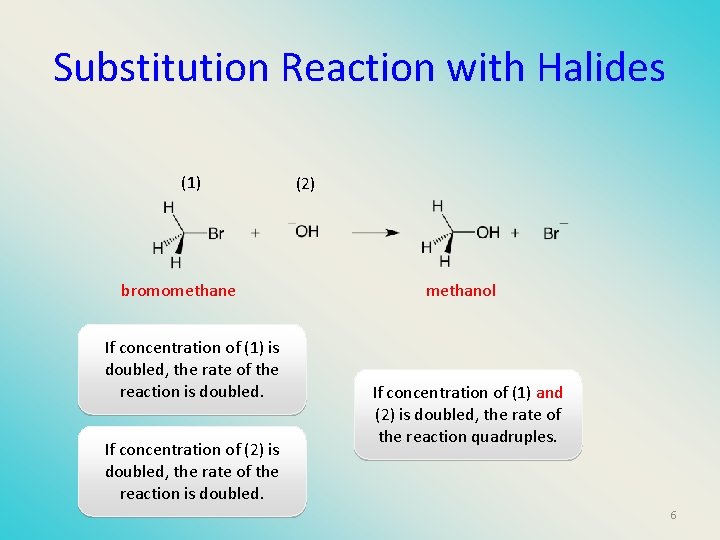
Substitution Reaction with Halides (1) bromomethane If concentration of (1) is doubled, the rate of the reaction is doubled. If concentration of (2) is doubled, the rate of the reaction is doubled. (2) methanol If concentration of (1) and (2) is doubled, the rate of the reaction quadruples. 6
![Substitution Reaction with Halides (1) (2) bromomethane methanol Rate law: rate = k [bromoethane][OH-] Substitution Reaction with Halides (1) (2) bromomethane methanol Rate law: rate = k [bromoethane][OH-]](http://slidetodoc.com/presentation_image/4946925a1426fd71ddb12300fbac2492/image-7.jpg)
Substitution Reaction with Halides (1) (2) bromomethane methanol Rate law: rate = k [bromoethane][OH-] this reaction is an example of a SN 2 reaction. S stands for substitution N stands for nucleophilic 2 stands for bimolecular 7

Mechanism of SN 2 Reactions Alkyl halide The rate of reaction depends on the concentrations of both reactants. When the hydrogens of bromomethane are replaced with methyl groups the reaction rate slow down. The reaction of an alkyl halide in which the halogen is bonded to an asymetric center leads to the formation of only one stereoisomer Relative rate 1200 40 1 ≈0 8
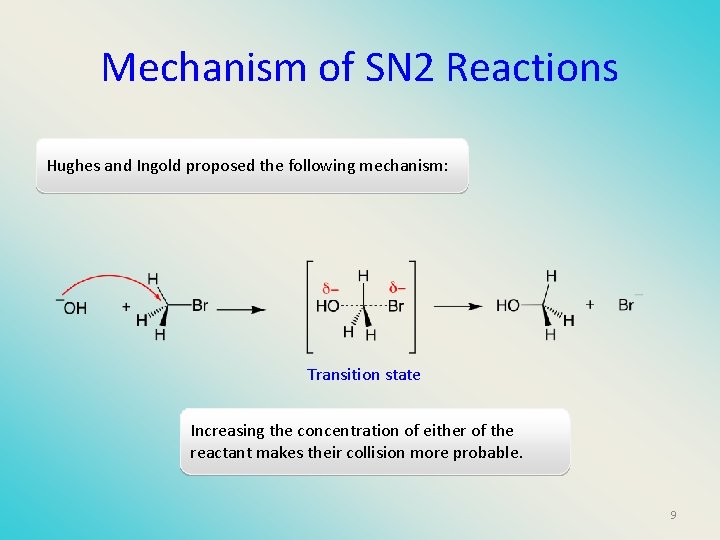
Mechanism of SN 2 Reactions Hughes and Ingold proposed the following mechanism: Transition state Increasing the concentration of either of the reactant makes their collision more probable. 9
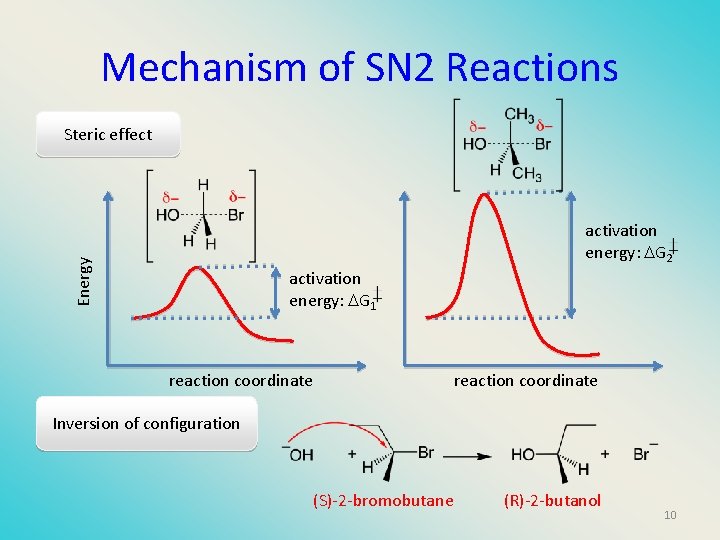
Mechanism of SN 2 Reactions Steric effect Energy activation energy: G 2 activation energy: G 1 reaction coordinate Inversion of configuration (S)-2 -bromobutane (R)-2 -butanol 10

Factor Affecting SN 2 Reactions The leaving group - + RCH I 2 HO HO + RCH 2 Br + RCH 2 Cl + RCH 2 F - RCH 2 OH + I RCH 2 OH + Br RCH 2 OH + Cl RCH 2 OH + F relative rates of reaction 30 000 10 000 200 1 p. Ka HX -10 -9 -7 3. 2 The nucleophile In general, for halogen substitution the strongest the base the better the nucleophile. p. Ka Nuclephilicity 11
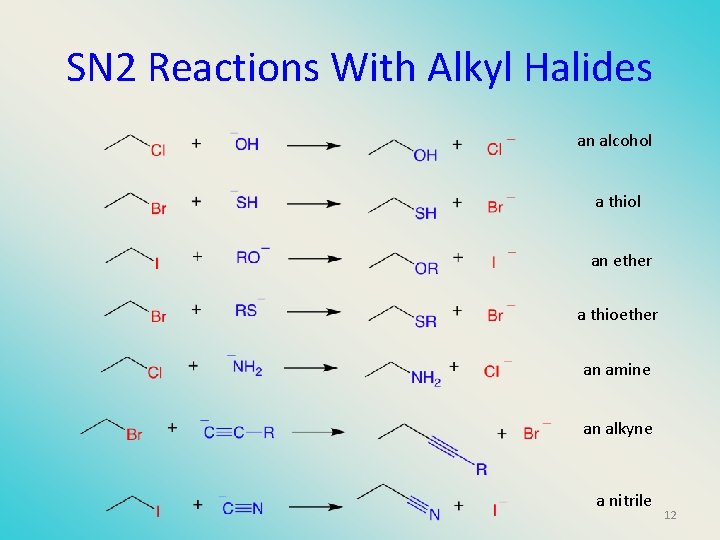
SN 2 Reactions With Alkyl Halides an alcohol a thiol an ether a thioether an amine an alkyne a nitrile 12
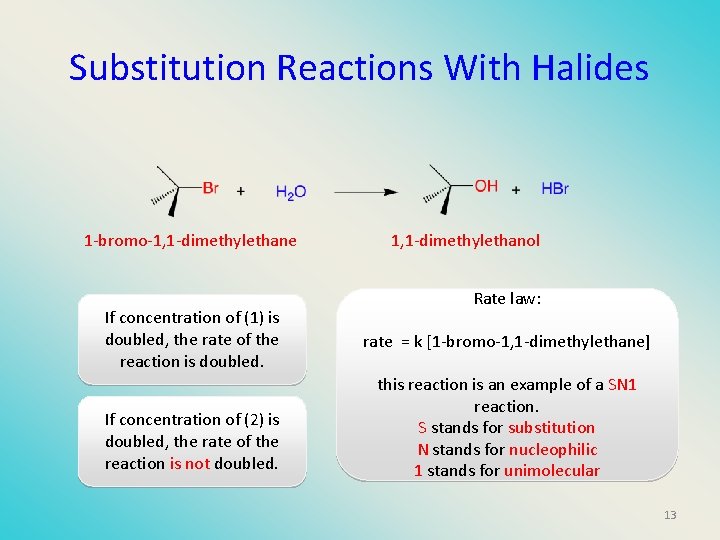
Substitution Reactions With Halides 1 -bromo-1, 1 -dimethylethane If concentration of (1) is doubled, the rate of the reaction is doubled. If concentration of (2) is doubled, the rate of the reaction is not doubled. 1, 1 -dimethylethanol Rate law: rate = k [1 -bromo-1, 1 -dimethylethane] this reaction is an example of a SN 1 reaction. S stands for substitution N stands for nucleophilic 1 stands for unimolecular 13

Mechanism of SN 1 Reactions Alkyl halide Relative rate The rate of reaction depends on the concentrations of the alkyl halide only. ≈0* When the methyl groups of 1 -bromo-1, 1 -dimethylethane are replaced with hydrogens the reaction rate slow down. ≈0* The reaction of an alkyl halide in which the halogen is bonded to an asymetric center leads to the formation of two stereoisomers 12 1 200 000 * a small rate is actually observed as a result of a SN 2 14
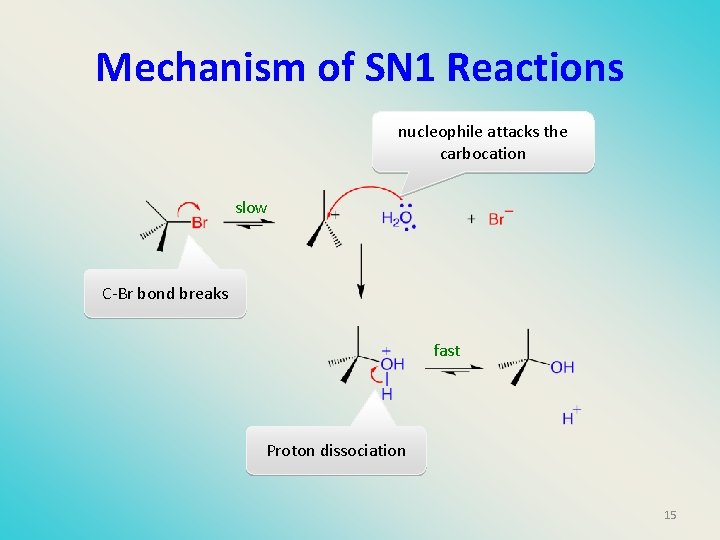
Mechanism of SN 1 Reactions nucleophile attacks the carbocation slow C-Br bond breaks fast Proton dissociation 15
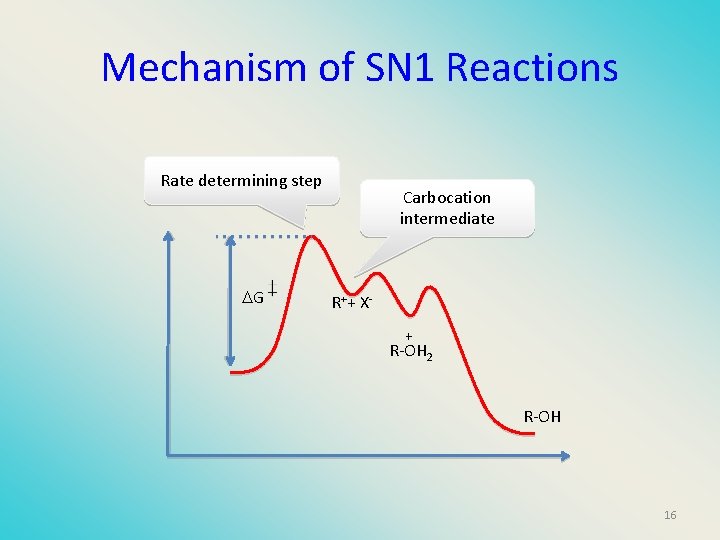
Mechanism of SN 1 Reactions Rate determining step G Carbocation intermediate R++ X+ R-OH 2 R-OH 16
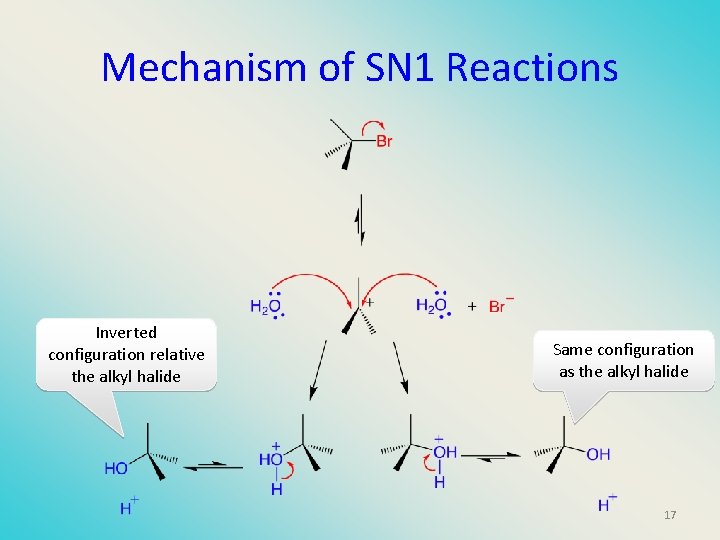
Mechanism of SN 1 Reactions Inverted configuration relative the alkyl halide Same configuration as the alkyl halide 17

Factor Affecting SN 1 reaction Two factors affect the rate of a SN 1 reaction: • The ease with which the leaving group dissociate from the carbon • The stability of the carbocation The more the substituted the carbocation is, the more stable it is and therefore the easier it is to form. As in the case of SN 2, the weaker base is the leaving group, the less tightly it is bonded to the carbon and the easier it is to break the bond The reactivity of the nucleophile has no effect on the rate of a SN 1 reaction 18
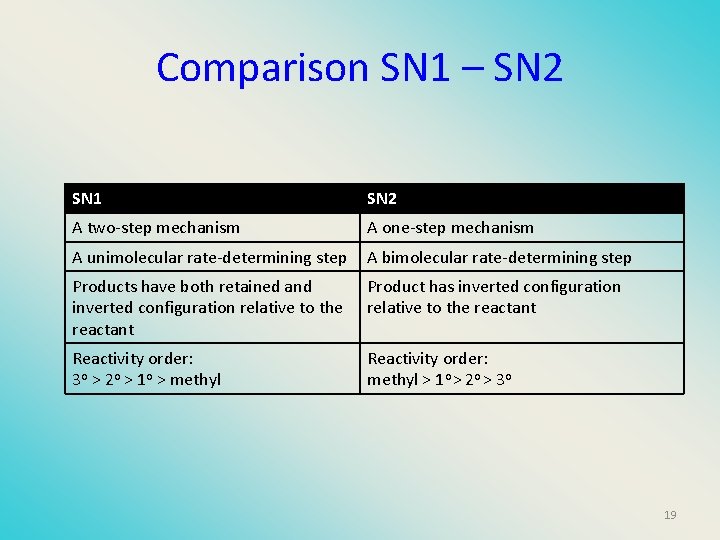
Comparison SN 1 – SN 2 SN 1 SN 2 A two-step mechanism A one-step mechanism A unimolecular rate-determining step A bimolecular rate-determining step Products have both retained and inverted configuration relative to the reactant Product has inverted configuration relative to the reactant Reactivity order: 3 o > 2 o > 1 o > methyl Reactivity order: methyl > 1 o > 2 o > 3 o 19
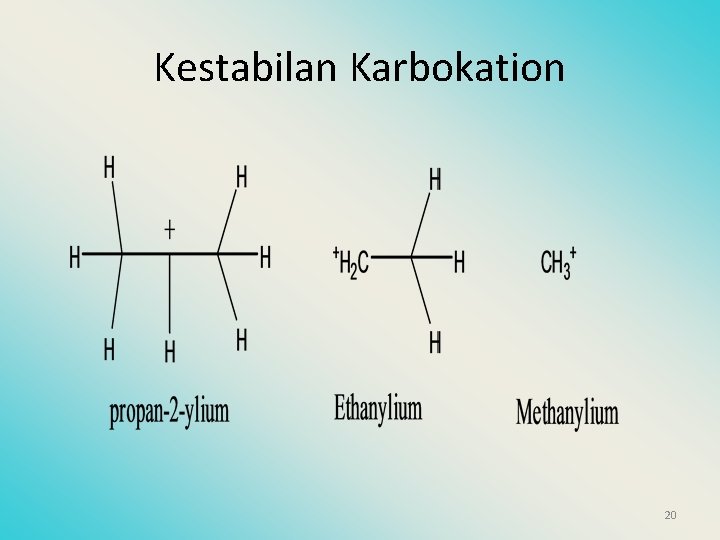
Kestabilan Karbokation 20

21
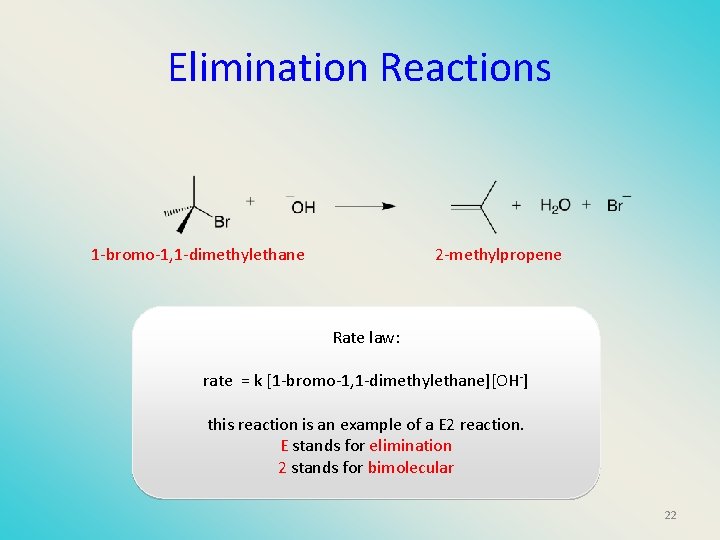
Elimination Reactions 1 -bromo-1, 1 -dimethylethane 2 -methylpropene Rate law: rate = k [1 -bromo-1, 1 -dimethylethane][OH-] this reaction is an example of a E 2 reaction. E stands for elimination 2 stands for bimolecular 22
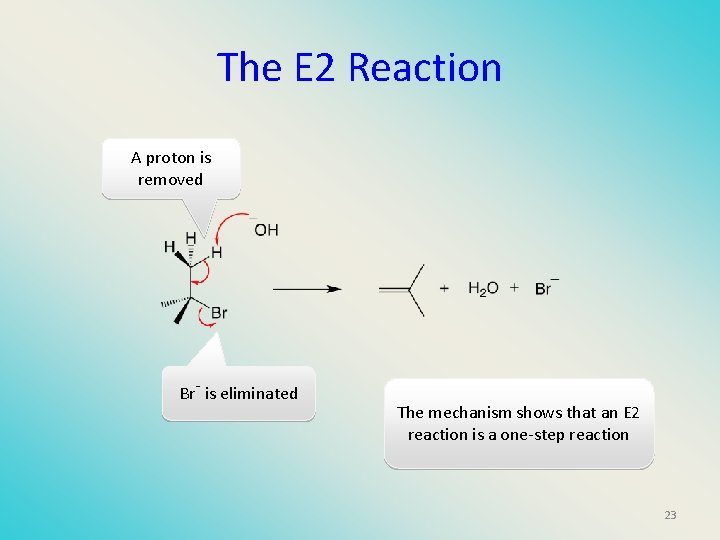
The E 2 Reaction A proton is removed Br- is eliminated The mechanism shows that an E 2 reaction is a one-step reaction 23
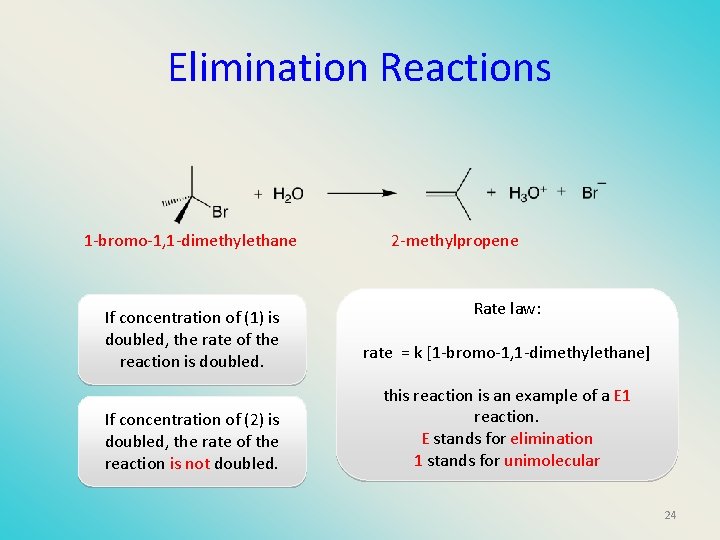
Elimination Reactions 1 -bromo-1, 1 -dimethylethane If concentration of (1) is doubled, the rate of the reaction is doubled. If concentration of (2) is doubled, the rate of the reaction is not doubled. 2 -methylpropene Rate law: rate = k [1 -bromo-1, 1 -dimethylethane] this reaction is an example of a E 1 reaction. E stands for elimination 1 stands for unimolecular 24
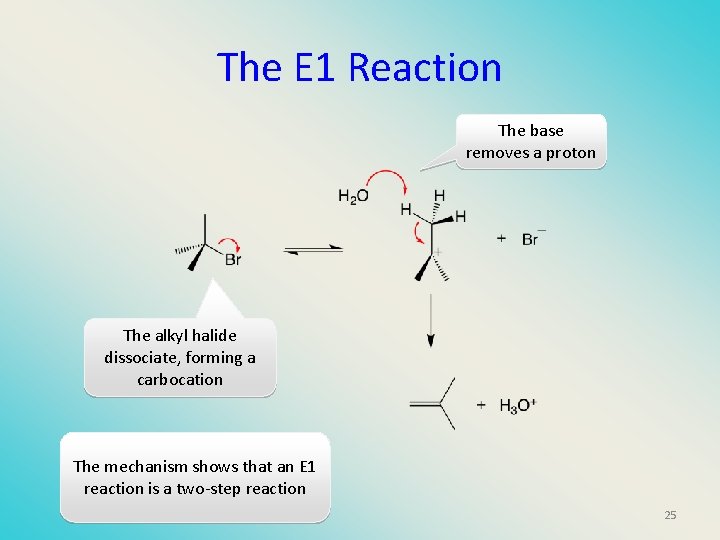
The E 1 Reaction The base removes a proton The alkyl halide dissociate, forming a carbocation The mechanism shows that an E 1 reaction is a two-step reaction 25
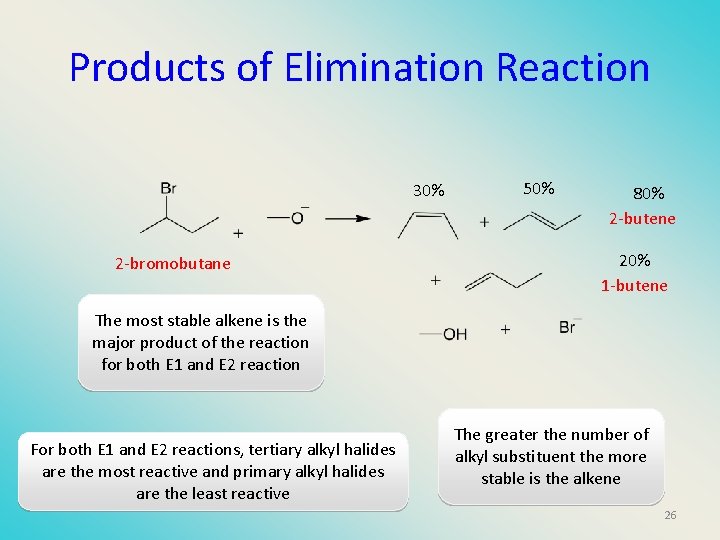
Products of Elimination Reaction 30% 2 -bromobutane 50% 80% 2 -butene 20% 1 -butene The most stable alkene is the major product of the reaction for both E 1 and E 2 reaction For both E 1 and E 2 reactions, tertiary alkyl halides are the most reactive and primary alkyl halides are the least reactive The greater the number of alkyl substituent the more stable is the alkene 26

ELIMINATION REACTIONS: ALKENES, ALKYNES 27
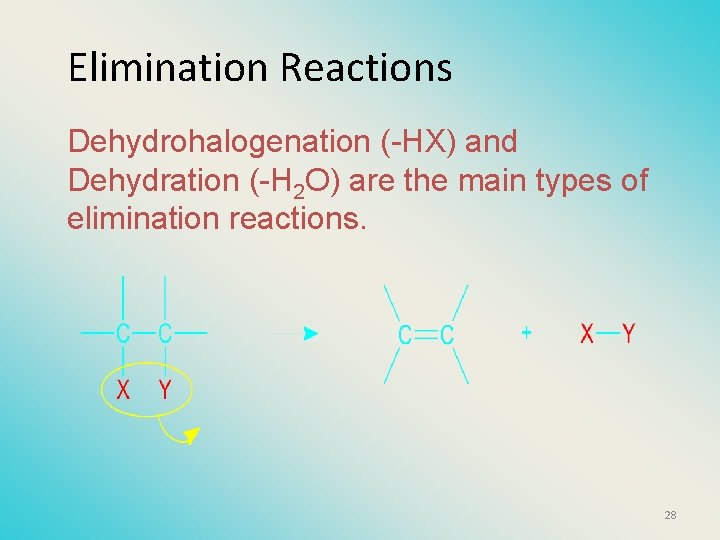
Elimination Reactions Dehydrohalogenation (-HX) and Dehydration (-H 2 O) are the main types of elimination reactions. 28
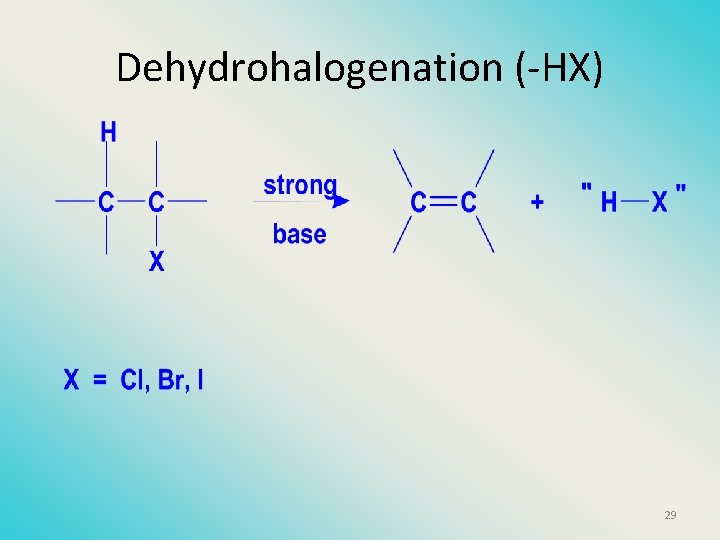
Dehydrohalogenation (-HX) 29
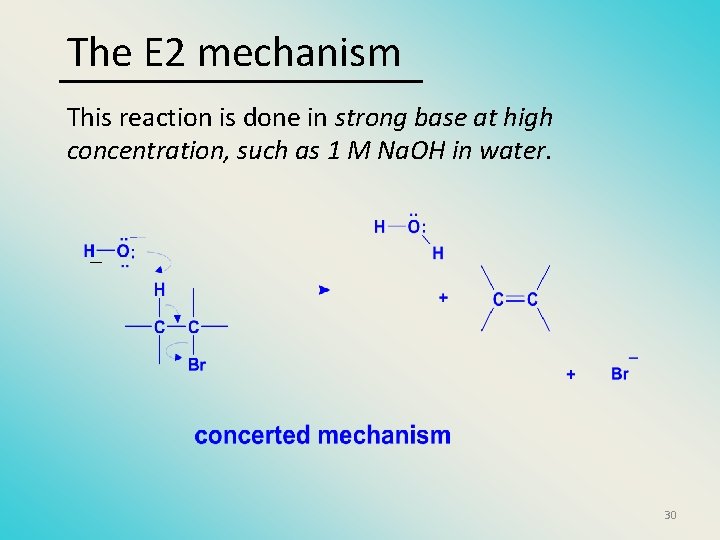
The E 2 mechanism This reaction is done in strong base at high concentration, such as 1 M Na. OH in water. _ 30

Kinetics • The reaction in strong base at high concentration is second order (bimolecular): Rate law: rate = k[OH-]1[R-Br]1 31
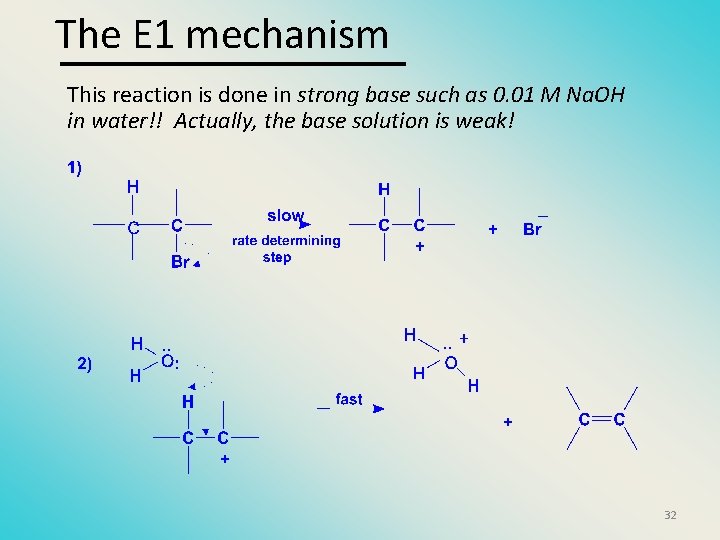
The E 1 mechanism This reaction is done in strong base such as 0. 01 M Na. OH in water!! Actually, the base solution is weak! 32

Kinetics • The reaction in weak base or under neutral conditions will be first order (unimolecular): • Rate law: rate = k [R-Br]1 • The first step (slow step) is rate determining! 33

The E 2 mechanism • • • Mechanism Kinetics Stereochemistry of reactants Orientation of elimination (Zaitsev’s rule) Stereochemistry of products Competing reactions 34

E 2 mechanism This reaction is done in strong base at high concentration, such as 1 M Na. OH in water. 35

Kinetics of an E 2 reaction • The reactions are second order (bimolecular reactions). • Rate = k [R-Br]1[Base]1 second order reaction (1 + 1 = 2) High powered math!! 36
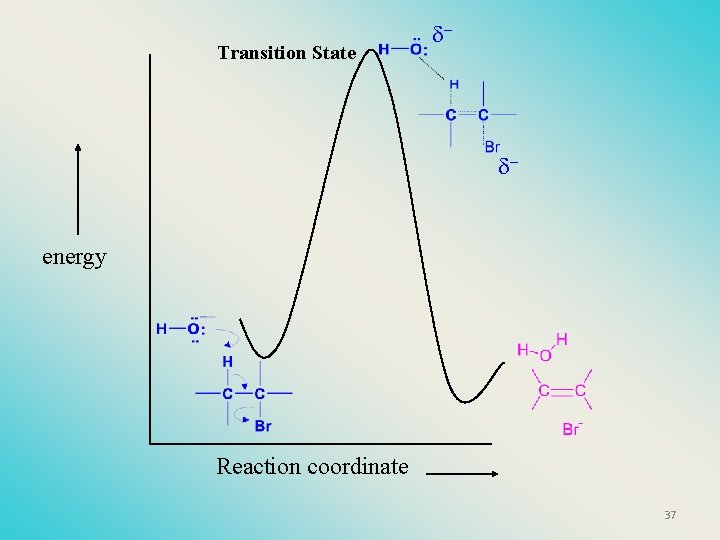
Transition State - - energy Reaction coordinate 37

Stereochemistry of reactants • E 2 reactions must go by an anti elimination • This means that the hydrogen atom and halogen atom must be 180 o (coplanar) with respect to each other!! • Draw a Newman projection formula and place the H and X on opposite sides. 38
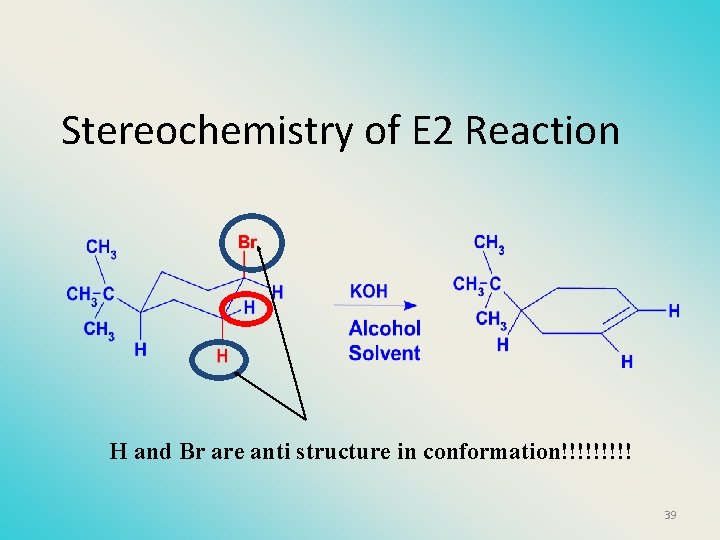
Stereochemistry of E 2 Reaction H and Br are anti structure in conformation!!!!! 39
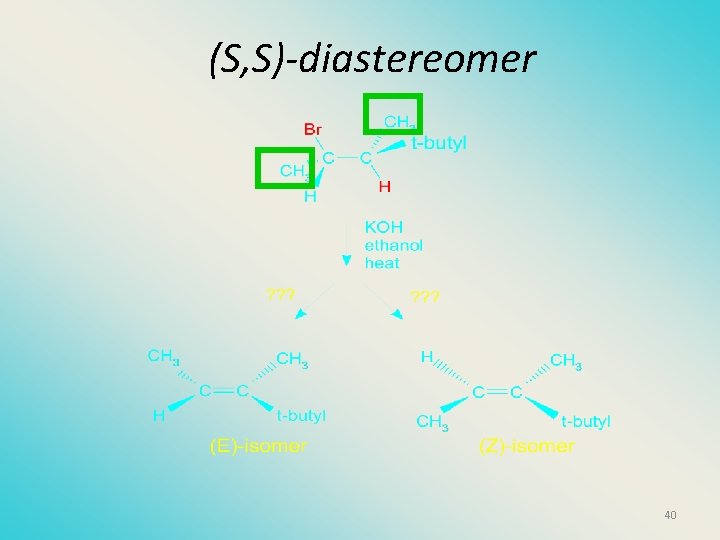
(S, S)-diastereomer 40
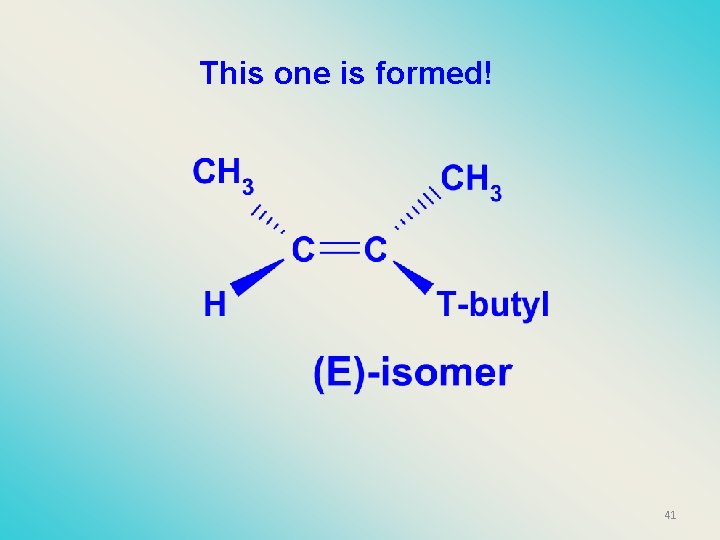
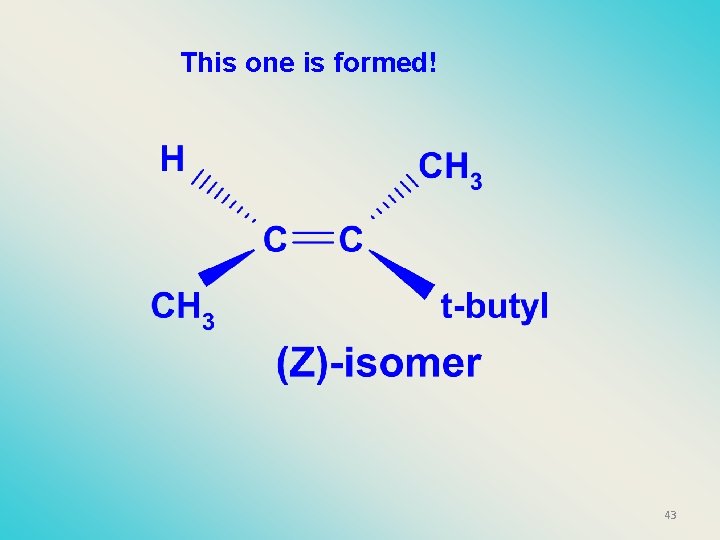
This one is formed! 41
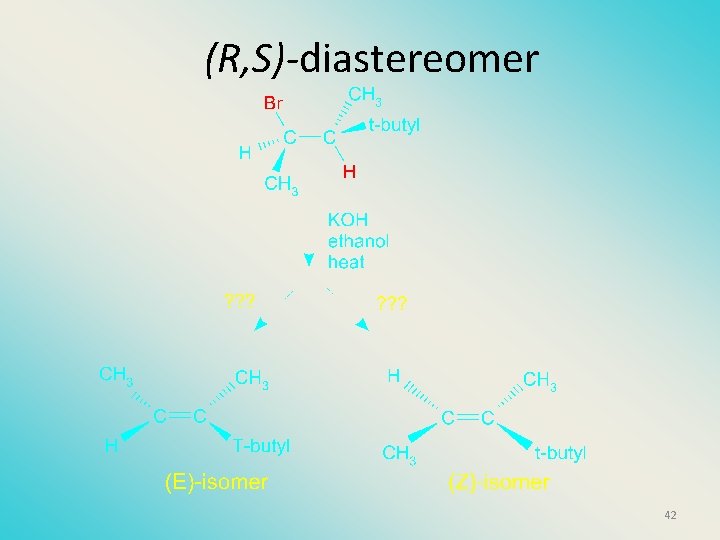
(R, S)-diastereomer 42
This one is formed! 43

Orientation of elimination: regiochemistry/ Zaitsev’s Rule • In reactions of removal of hydrogen halides from alkyl halides or the removal of water from alcohols, the hydrogen which is lost will come from the more highly-branched b-carbon. More branched Less branched A. N. Zaitsev -- 1875 44
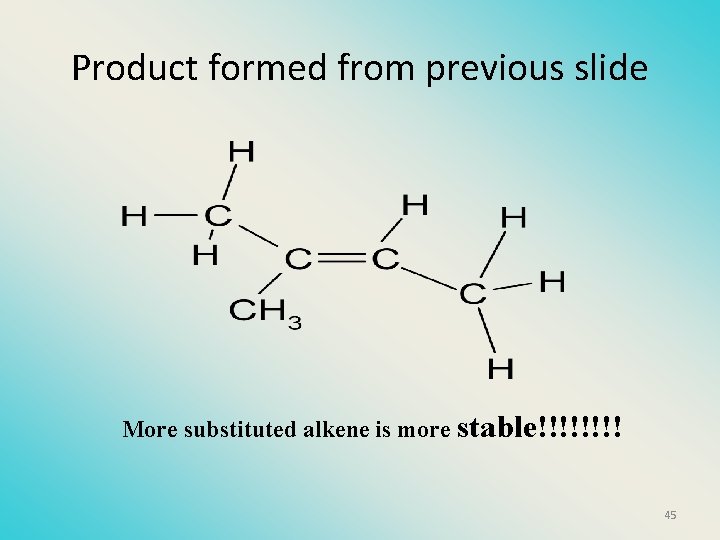
Product formed from previous slide More substituted alkene is more stable!!!! 45

Typical bases used in E 2 reactions High concentration of the following >1 M If the concentration isn’t given, assume that it is high concentration! • Na+ -OH • K+ -OH • Na+ -OR • Na+ -NH 2 46

Orientation of elimination: regiochemistry/ Zaitsev’s Rule Explaination of Zaitsev’s rule: When you remove a hydrogen atom from the more branched position, you are forming a more highly substituted alkene. 47

Stereochemistry of products • The H and X must be anti with respect to each other in an E 2 reaction! • You take what you get, especially with diastereomers! See the previous slides of the reaction of diastereomers. 48

Competing reactions • The substitution reaction (SN 2) competes with the elimination reaction (E 2). • Both reactions follow second order kinetics! 49

The E 1 mechanism • Mechanism • Kinetics • Stereochemistry of reactants • Orientation of elimination (Zaitsev’s rule) • Stereochemistry of products • Competing reactions 50
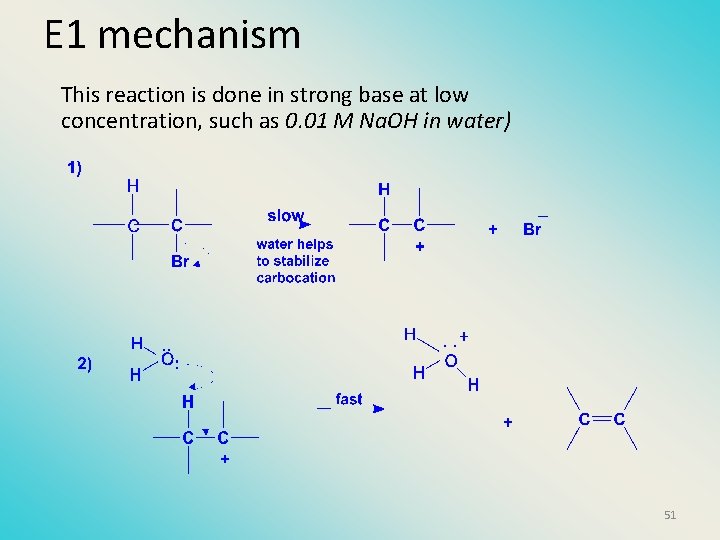
E 1 mechanism This reaction is done in strong base at low concentration, such as 0. 01 M Na. OH in water) 51

E 1 Reactions • These reactions proceed under neutral conditions where a polar solvent helps to stabilize the carbocation intermediate. • This solvent also acts as a weak base and removes a proton in the fast step. • These types of reactions are referred to as solvolysis reactions. 52

• tertiary substrates go by E 1 in polar solvents, with little or no base present! • typical polar solvents are water, ethanol, methanol and acetic acid • These polar solvents help stabilize carbocations • E 1 reactions also occur in a low concentration of base (i. e. 0. 01 M Na. OH). 53

However!!!! • With strong base (i. e. >1 M), goes by E 2 54
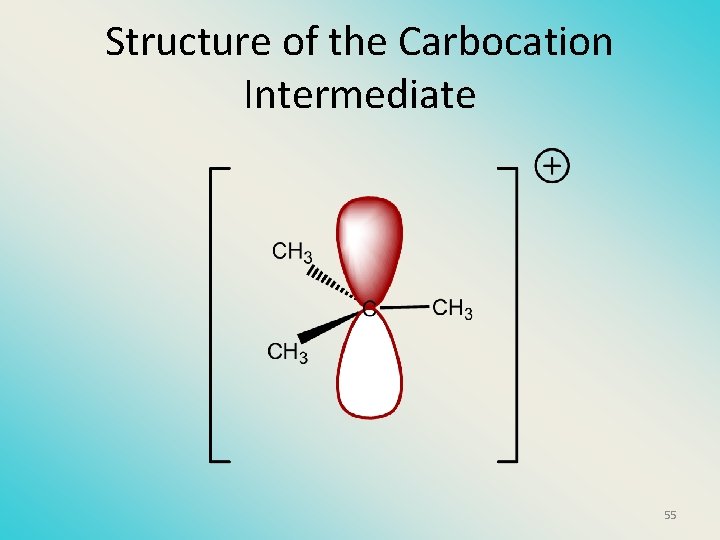
Structure of the Carbocation Intermediate 55

Carbocation stability order Tertiary (3 o) > secondary (2 o) > primary (1 o) It is hard (but not impossible) to get primary compounds to go by E 1. The reason for this is that primary carbocations are not stable! 56

Kinetics of an E 1 reaction • E 1 reactions follow first order (unimolecular) kinetics: Rate = k [R-X]1 • The solvent helps to stabilize the carbocation, but it doesn’t appear in the rate law!! 57
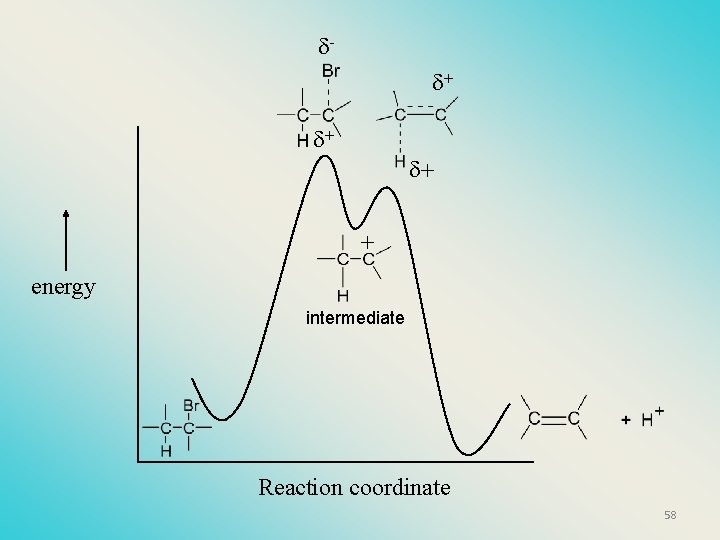
+ + energy intermediate Reaction coordinate 58

Stereochemistry of the reactants • E 1 reactions do not require an anti coplanar orientation of H and X. • Diastereomers give the same products with E 1 reactions, including cis- and trans products. • Remember, E 2 reactions usually give different products with diastereomers. 59

Orientation of elimination • E 1 reactions faithfully follow Zaitsev’s rule! • This means that the major product should be the product that is the most highly substituted. 60

Stereochemistry of products E 1 reactions usually give thermodynamically most stable product as the major product. This usually means that the largest groups should be on opposite sides of the double bond. Usually this means that the trans product is obtained. 61

Competing reactions • The substitution reaction (SN 1) competes with the elimination reaction (E 1). • Both reactions follow first order kinetics! 62

Whenever there are carbocations… • They can undergo elimination (E 1) • They can undergo substitution (SN 1) • They can rearrange – and then undergo elimination – or substituion 63

Rearrangements • Alkyl groups and hydrogen can migrate in rearrangement reactions to give more stable intermediate carbocations. • You shouldn’t assume that rearrangements always occur in all E 1 reactions, otherwise paranoia will set in!! 64

Comparison of E 2 / E 1 • E 1 reactions occur under essentially neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E 1 reactions can also occur with strong bases, but only at low concentration, about 0. 01 to 0. 1 M or below. • E 2 reactions require strong base in high concentration, about 1 M or above. 65

Comparison of E 2 / E 1 • E 1 is a stepwise mechanism (two or more); Carbocation intermediate! • E 2 is a concerted mechanism (one step) No intermediate! • E 1 reactions may give rearranged products • E 2 reactions don’t give rearrangement • Alcohol dehydration reactions are E 1 66
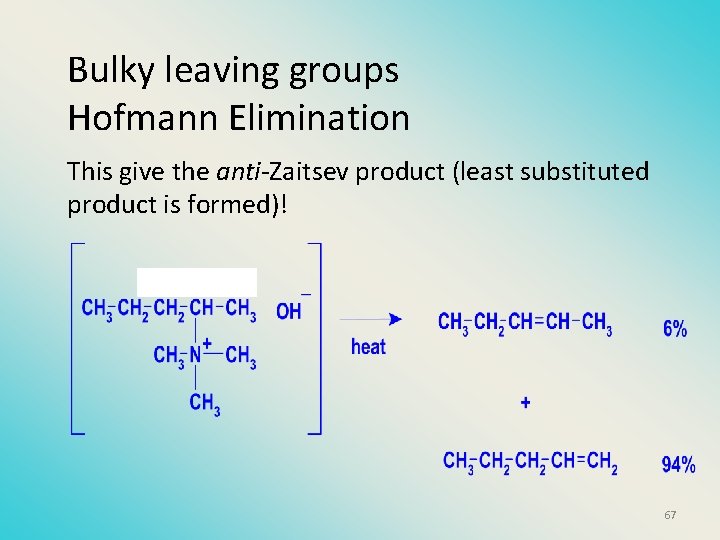
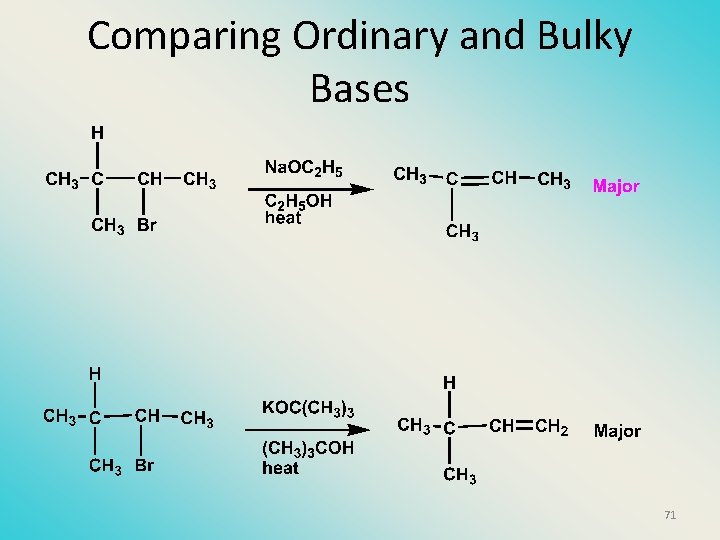
Bulky leaving groups Hofmann Elimination This give the anti-Zaitsev product (least substituted product is formed)! 67

Orientation of elimination: regiochemistry/ Hofmann’s Rule • In bimolecular elimination reactions in the presence of either a bulky leaving group or a bulky base, the hydrogen that is lost will come from the LEAST highly -branched b-carbon. More branched Less branched 68
Product from previous slide 69

Elimination with bulky bases • Non-bulky bases, such as hydroxide and ethoxide, give Zaitsev products. • Bulky bases, such as potassium tertbutoxide, give larger amounts of the least substituted alkene (Hoffmann) than with simple bases. 70
Comparing Ordinary and Bulky Bases 71
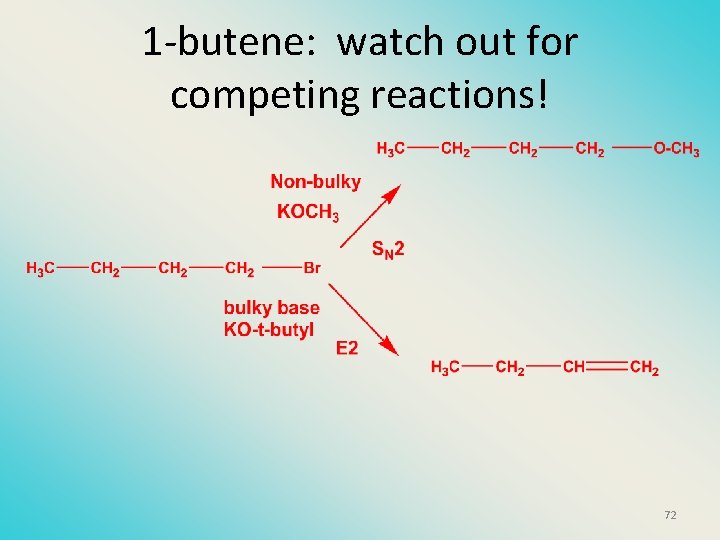
1 -butene: watch out for competing reactions! 72

Highlights • • • Dehydrohalogenation -- E 2 Mechanism Zaitsev’s Rule Dehydrohalogenation -- E 1 Mechanism Carbocation Rearrangements -- E 1 Elimination with Bulky Leaving Groups and Bulky Bases -- Hofmann Rule -- E 2 73

Competition Between SN 2/E 2 and SN 1/E 1 SN 2 E 1 E 2 rate = k 1[alkyl halide] + k 2[alkyl halide][nucleo. ] + k 3[alkyl halide] + k 2[alkyl halide][base] • SN 2 and E 2 are favoured by a high concentration of a good nucleophile/strong base • SN 1 and E 1 are favoured by a poor nucleophile/weak base, because a poor nucleophile/weak base disfavours SN 2 and E 2 reactions 74
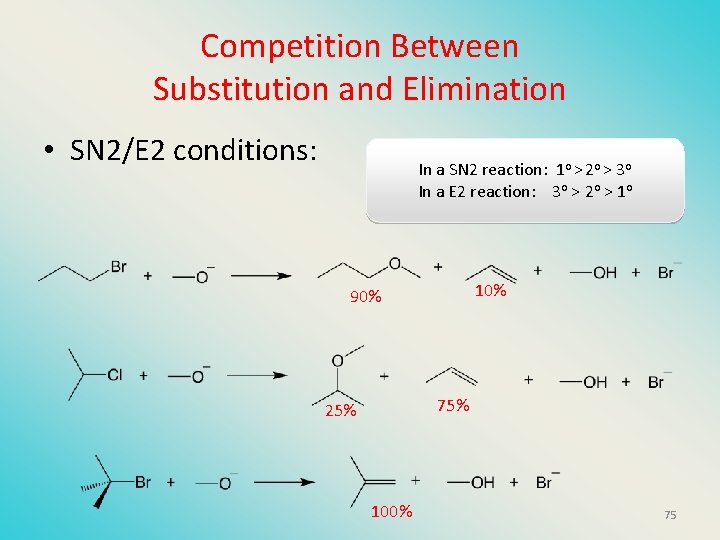
Competition Between Substitution and Elimination • SN 2/E 2 conditions: In a SN 2 reaction: 1 o > 2 o > 3 o In a E 2 reaction: 3 o > 2 o > 1 o 10% 90% 75% 25% 100% 75

Competition Between Substitution and Elimination • SN 1/E 1 conditions: All alkyl halides that react under SN 1/E 1 conditions will give both substitution and elimination products (≈50%/50%) 76

Summary • Alkyl halides undergo two kinds of nucleophilic subtitutions: SN 1 and SN 2, and two kinds of elimination: E 1 and E 2. • SN 2 and E 2 are bimolecular one-step reactions • SN 1 and E 1 are unimolecular two step reactions • SN 1 lead to a mixture of stereoisomers • SN 2 inverts the configuration od an asymmetric carbon • The major product of a elimination is the most stable alkene • SN 2 are E 2 are favoured by strong nucleophile/strong base • SN 2 reactions are favoured by primary alkyl halides • E 2 reactions are favoured by tertiary alkyl halides 77

REAKSI ADISI ALKENA 78

Addition Reaction of Alkene 1. HX Addition • Electrophilic Addition (Markovnikov Product) • Free Radical Mechanism (Anti-Mark Product) 2. Hydration (+ H 2 O) 3. Halogenation/ Hydrohalogenation 4. Reduction or Hydrogenation (+ H 2 ) 5. Oxidation 6. Multi-step Synthesis
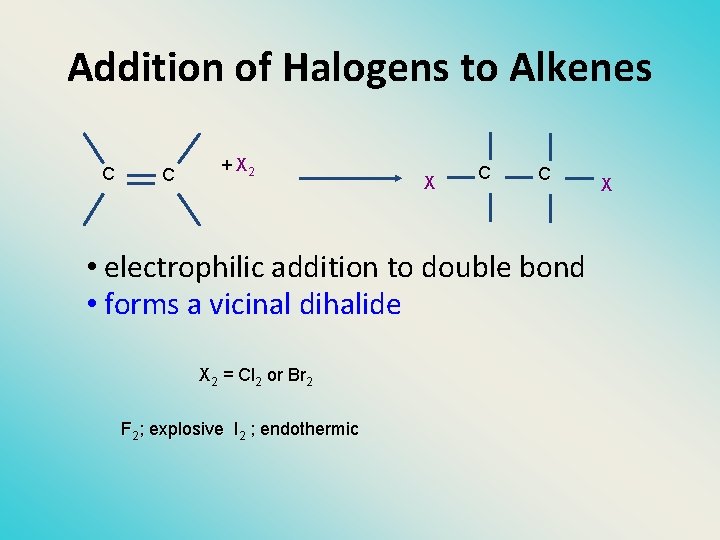
Addition of Halogens to Alkenes C C + X 2 X C C • electrophilic addition to double bond • forms a vicinal dihalide X 2 = Cl 2 or Br 2 F 2; explosive I 2 ; endothermic X
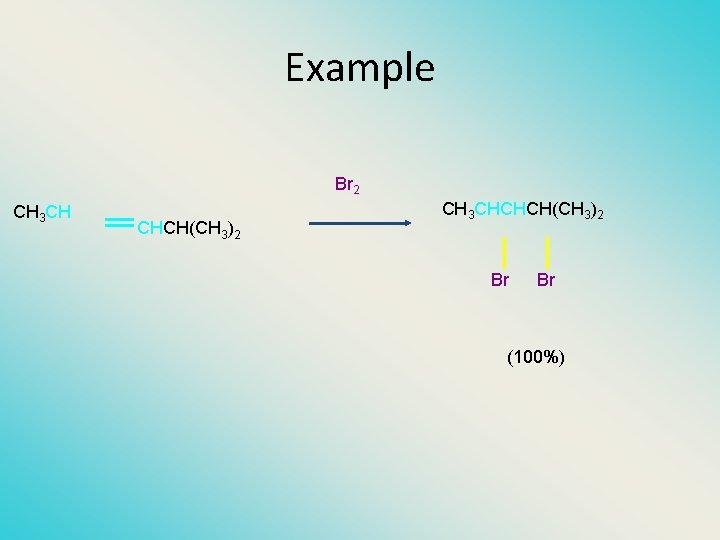
Example Br 2 CH 3 CH CHCH(CH 3)2 CH 3 CHCHCH(CH 3)2 Br Br (100%)
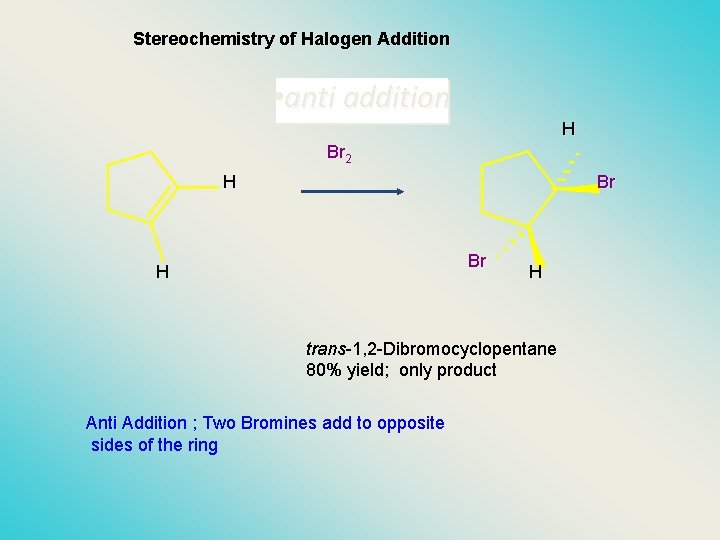
Stereochemistry of Halogen Addition • anti addition H Br 2 H Br Br H H trans-1, 2 -Dibromocyclopentane 80% yield; only product Anti Addition ; Two Bromines add to opposite sides of the ring
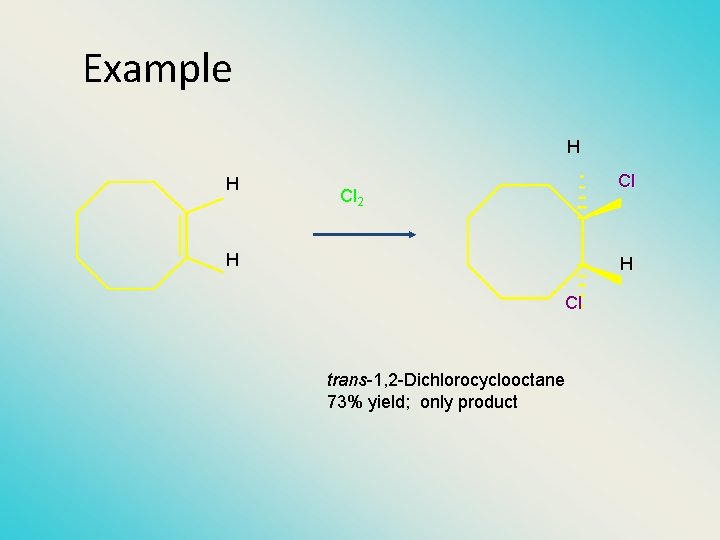
Example H H Cl Cl 2 H H Cl trans-1, 2 -Dichlorocyclooctane 73% yield; only product

Mechanism is electrophilic addition • Br 2 is not polar, but it is polarizable • two steps (1) formation of bromonium ion & electrophilic attack • • (2) nucleophilic attack on bromo ion by bromide NET REACTION CH 2=CH 2 + Br 2 -> Br-CH 2 -Br
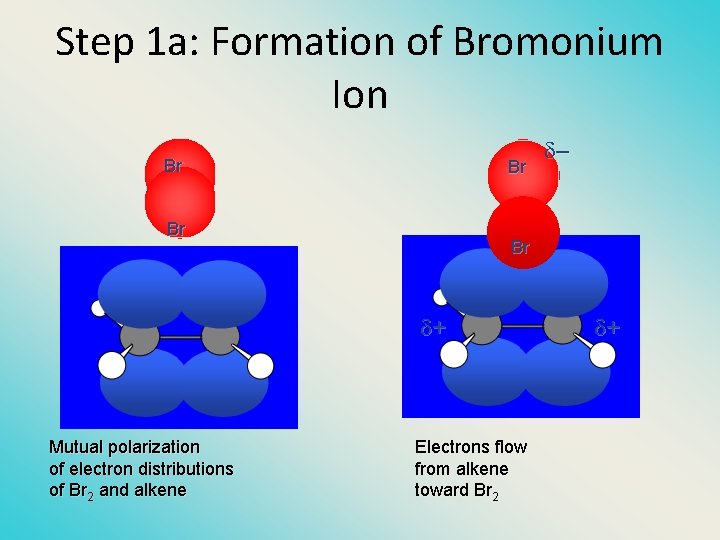
Step 1 a: Formation of Bromonium Ion Br Br + Mutual polarization of electron distributions of Br 2 and alkene – Electrons flow from alkene toward Br 2 +
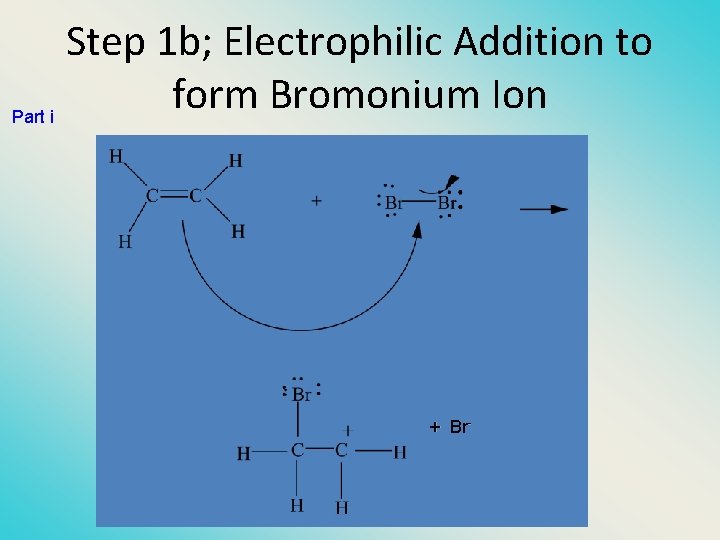
Step 1 b; Electrophilic Addition to form Bromonium Ion Part i + Br-
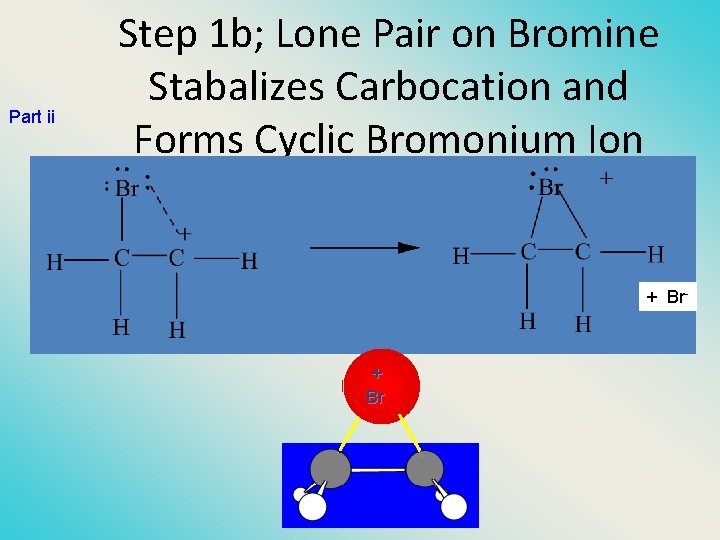
Part ii Step 1 b; Lone Pair on Bromine Stabalizes Carbocation and Forms Cyclic Bromonium Ion + Br- + Br
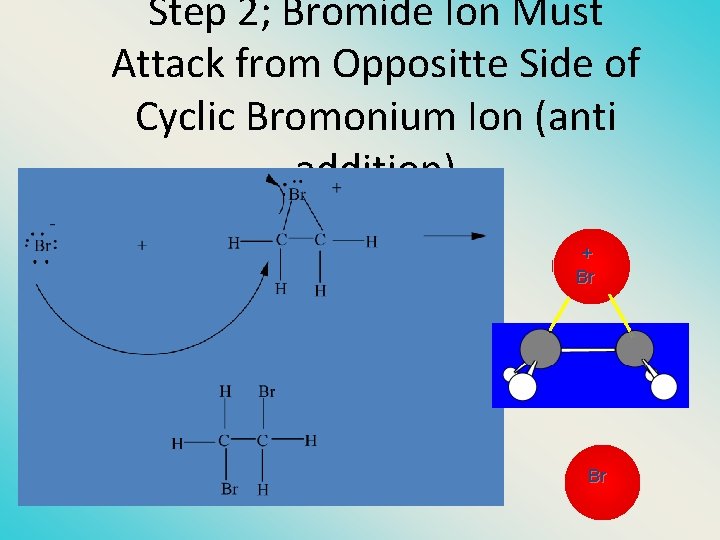
Step 2; Bromide Ion Must Attack from Oppositte Side of Cyclic Bromonium Ion (anti addition) + Br Br
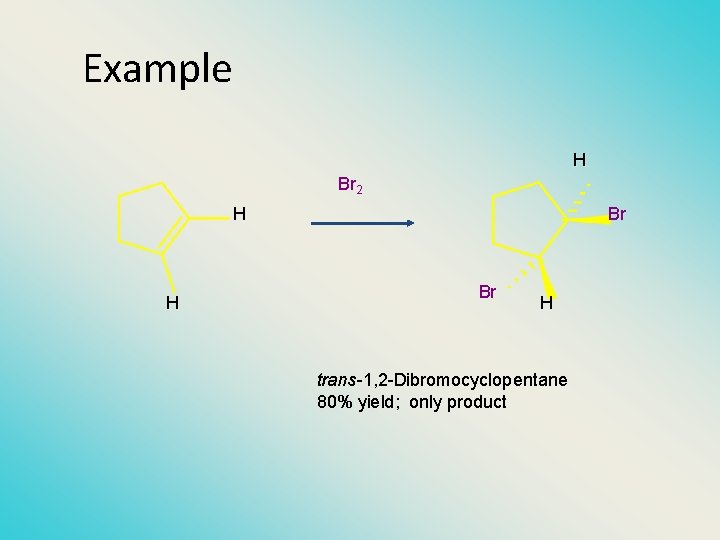
Example H Br 2 H H Br Br H trans-1, 2 -Dibromocyclopentane 80% yield; only product
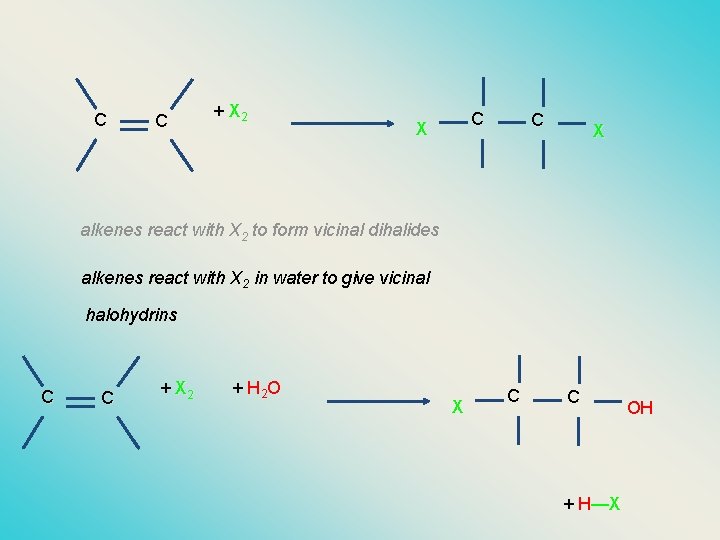
C C + X 2 C X alkenes react with X 2 to form vicinal dihalides alkenes react with X 2 in water to give vicinal halohydrins C C + X 2 + H 2 O X C C + H—X OH
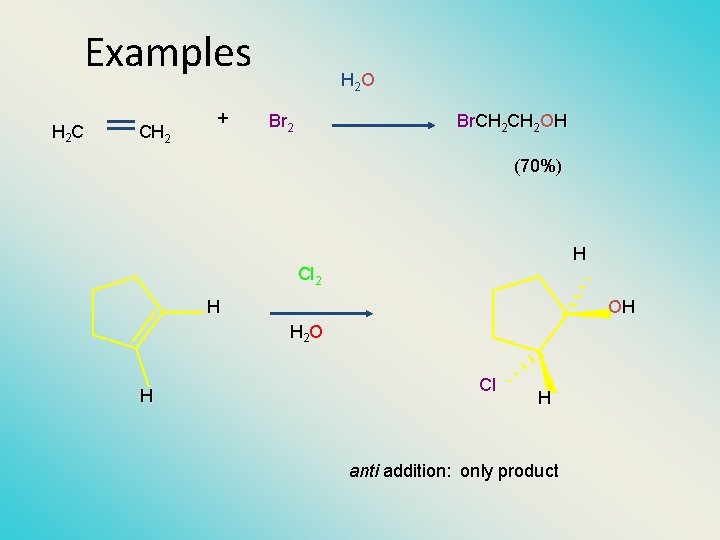
Examples H 2 C CH 2 + H 2 O Br 2 Br. CH 2 OH (70%) H Cl 2 H OH H 2 O H Cl H anti addition: only product
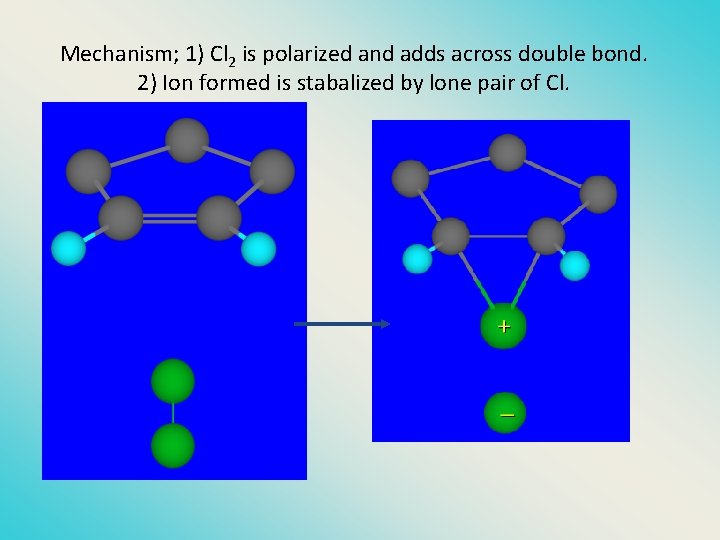
Mechanism; 1) Cl 2 is polarized and adds across double bond. 2) Ion formed is stabalized by lone pair of Cl.
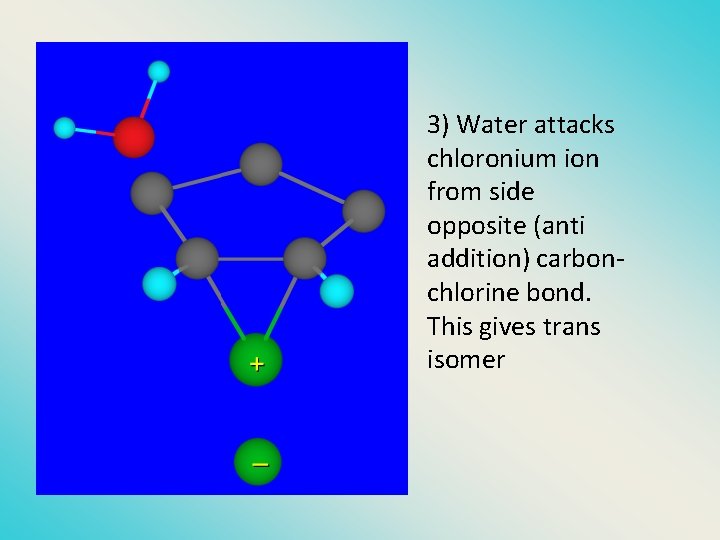
3) Water attacks chloronium ion from side opposite (anti addition) carbonchlorine bond. This gives trans isomer
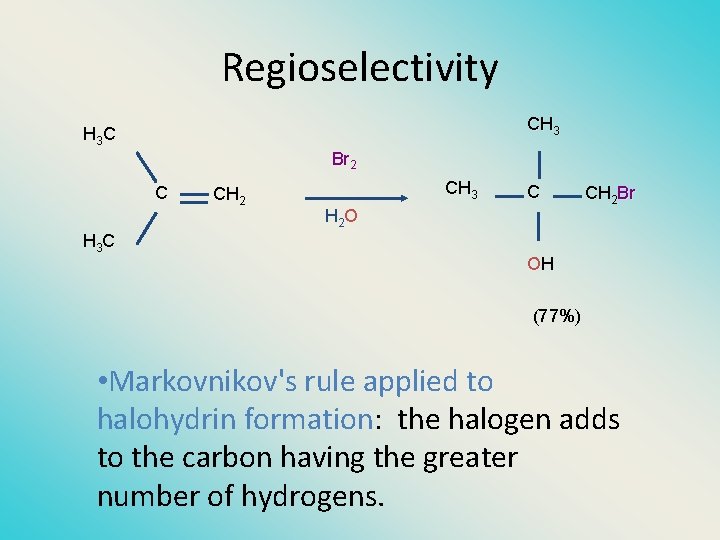
Regioselectivity CH 3 C Br 2 C H 3 C CH 2 CH 3 C H 2 O CH 2 Br OH (77%) • Markovnikov's rule applied to halohydrin formation: the halogen adds to the carbon having the greater number of hydrogens.
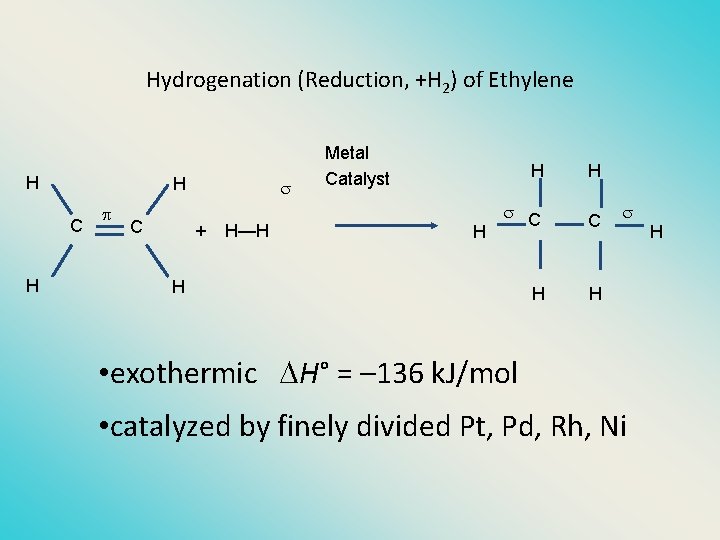
Hydrogenation (Reduction, +H 2) of Ethylene H H C + H—H Metal Catalyst H H H C C H H H • exothermic H° = – 136 k. J/mol • catalyzed by finely divided Pt, Pd, Rh, Ni H
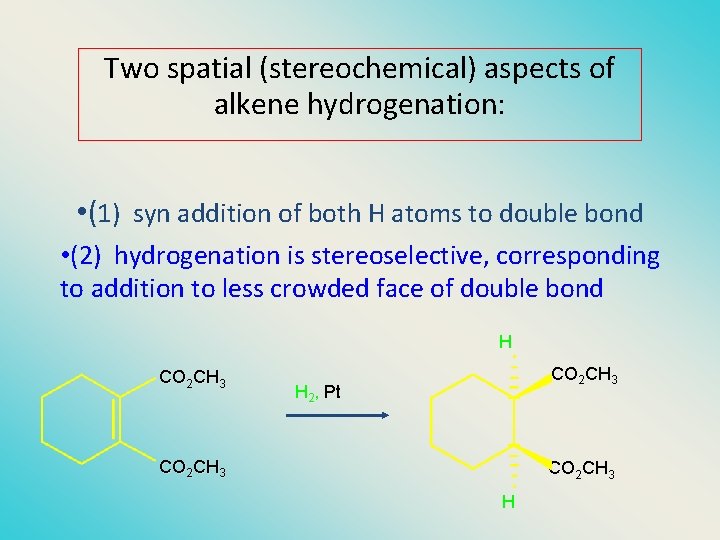
Two spatial (stereochemical) aspects of alkene hydrogenation: • (1) syn addition of both H atoms to double bond • (2) hydrogenation is stereoselective, corresponding to addition to less crowded face of double bond H CO 2 CH 3 H 2, Pt CO 2 CH 3 H
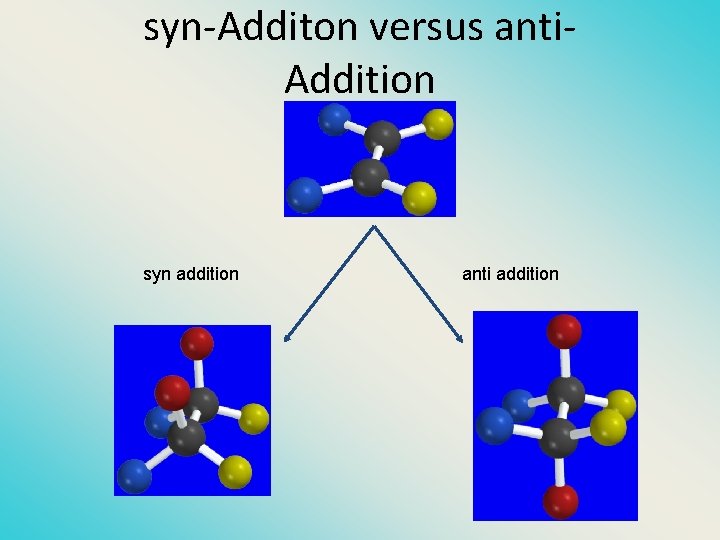
syn-Additon versus anti. Addition syn addition anti addition
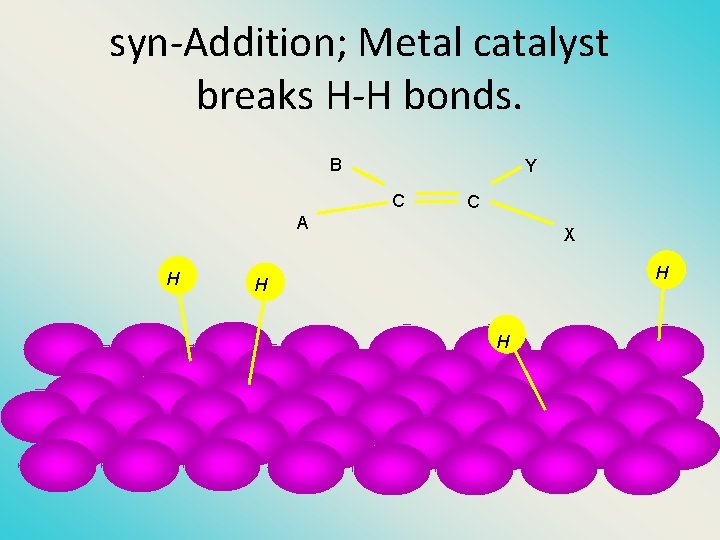
syn-Addition; Metal catalyst breaks H-H bonds. B Y C A H C X H H H
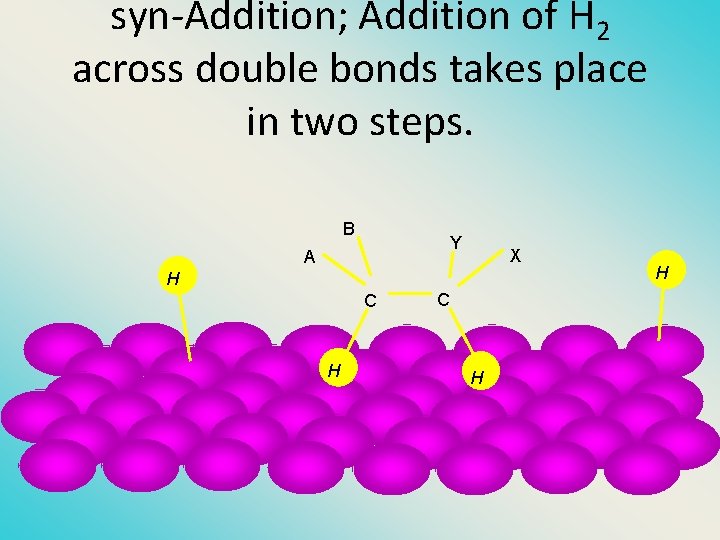
syn-Addition; Addition of H 2 across double bonds takes place in two steps. B Y A X H C H H
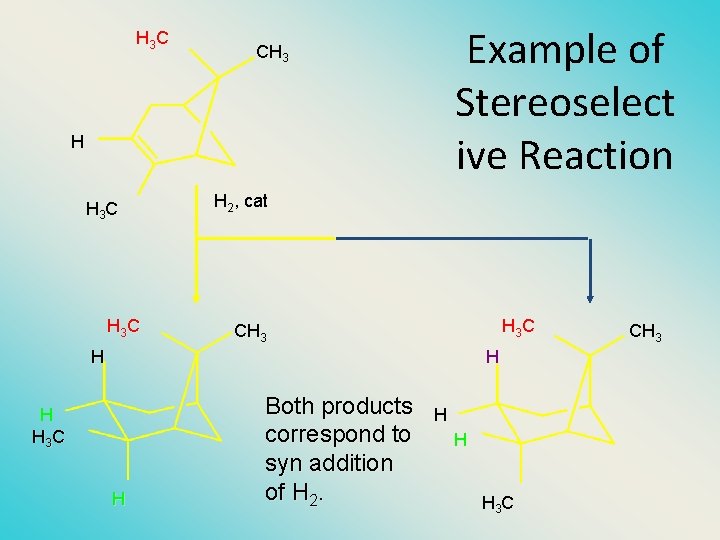
H 3 C Example of Stereoselect ive Reaction CH 3 H H 3 C H 2, cat H 3 C CH 3 H H 3 C H Both products correspond to syn addition of H 2. H H H 3 C CH 3
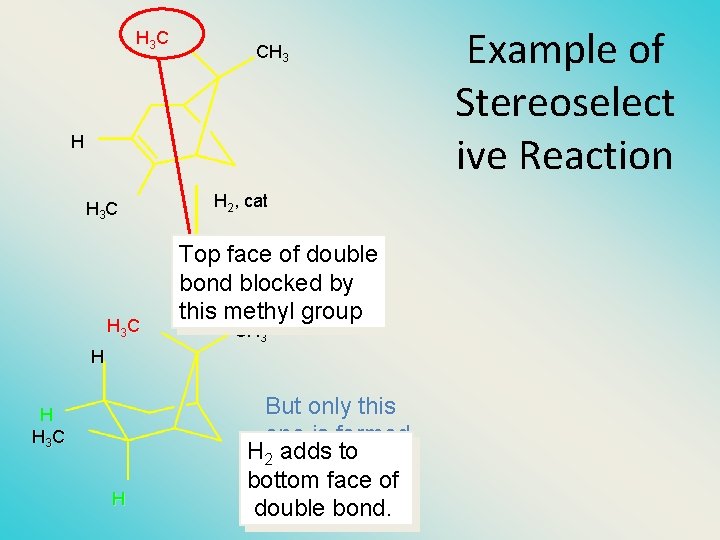
H 3 C CH 3 H H 3 C H 2, cat Top face of double bond blocked by this methyl group CH 3 H H H 3 C H But only this one is formed. H 2 adds to bottom face of double bond. Example of Stereoselect ive Reaction
- Slides: 101