Substantially Conductive Polymers Behzad Pourabbas Dep Of Polymer

Substantially Conductive Polymers Behzad Pourabbas Dep. Of Polymer eng. Sahand University of Technology Tabriz-Iran pourabas@sut. ac. ir

In order to download the PPT files: � 1 - Go to SUT web page (www. sut. ac. ir/en) � 2 - find the Polymer eng. Faculty (http: //polymer. sut. ac. ir/) � 3 - Go to my page: (http: //polymer. sut. ac. ir/showcvmain. aspx? id=5) � 4 - Education Courses Ph. D Materials for IT (http: //polymer. sut. ac. ir/Course. Brf. aspx? id=32) � 5 - Download the files: (http: //polymer. sut. ac. ir/download. aspx? id=32) � Using the file contents are allowed by referencing. 11/5/2020

Overview Discovery and history The story of Polyacetylene Structural Characteristics Doping Concept The Charge Carriers Applications 11/5/2020

The discoverers, the Noble laureates (Chem 2000) 11/5/2020

11/5/2020

Materials, According To Electrical Properties � insulator, � <10 -7 S/cm � semiconductor, � 10 -4 - 10 S/cm depending upon doping degree � conductor � >103 and S/cm � superconductor 11/5/2020

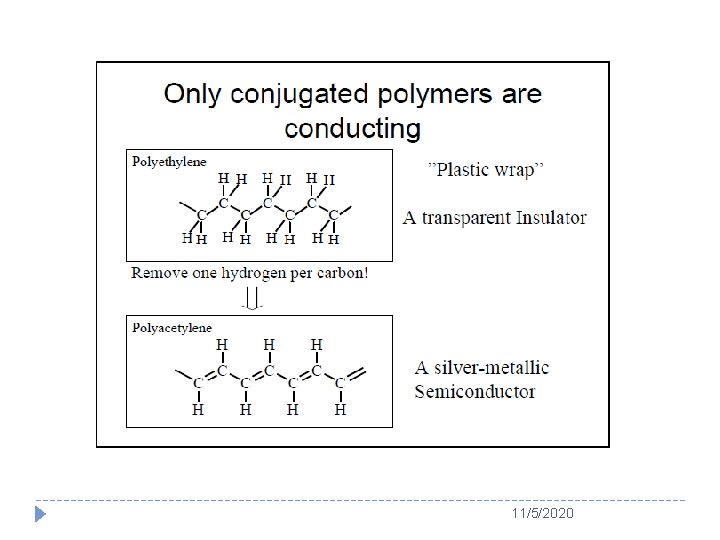
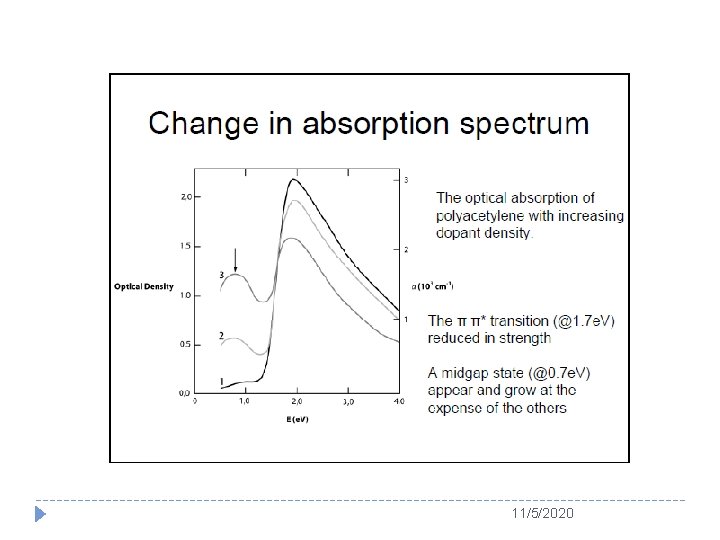
Polymers and Conductivity � Since discovery of conductive polyacelene (PA) doped with iodine, a new field of conducting polymers, which is also called as “synthetic metals”, has been established and earned the Nobel Prize in Chemistry in 2000. � In 1977, this accidentally discovered that insulating π -conjugated PA could become conductor with a conductivity of 103 S/cm by iodine doping. 11/5/2020

The story of PA 11/5/2020

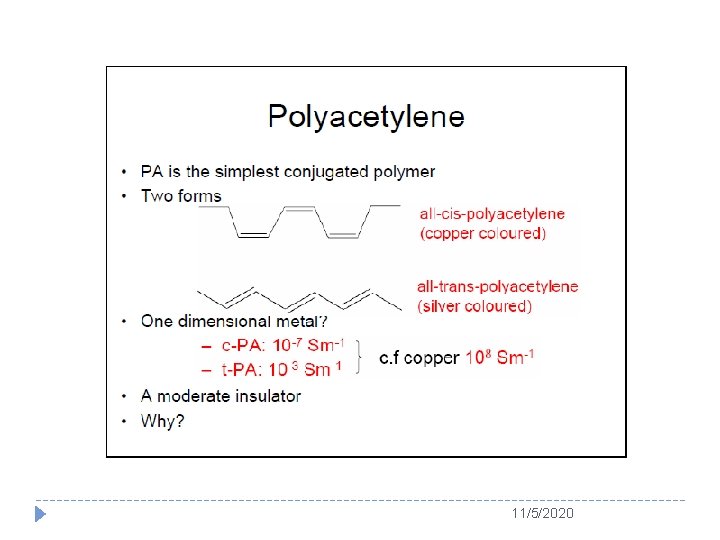
The Story of Polyacetylene � PA could be regarded as an excellent candidate of polymers to be imitating a metal. � Because it has alternating double and single bonds, as called conjugated double bonds. 11/5/2020

The Story of Polyacetylene � At the beginning of the 1970 s, Hedeki Shirakawa at Tokyo Institute of Technology, Japan, was studying the polymerization of acetylene into plastics by using catalyst created by Ziegler-Natta. � A visiting scientist in Shirakawa’s group tried to synthesize PA in the usual way. However, a beautifully lustrous silver colored film, rather than the black powder synthesized by the conventional method, was obtained. 11/5/2020

The Story of Polyacetylene � Shirakawa finally found that the catalyst concentration used was enhanced by 103 times! Shirakawa was stimulated by the accidental discovery and further found the molecular structure of the resulting PA was affected by reaction temerature, for instance, the silvery film was transpolyacetylene whereas copper-colored film was almost pure cis-polyacetylene. 11/5/2020

The Story of Polyacetylene � In another part of the world, chemiste Alan G. Mac. Diarmid and physicist Alan J. Heeger at University of Pennsylvania, Philadephia, USA were studying the first metal-like inorganic polymer sulfur nitride ((SN)x ), which is the first example of a covalent polymer without metal atoms. In 1975, Prof. Mac. Diarmid visited Tokyo Institute of Technology and gave a talk on (SN)x. After his lecture, Mac. Diarmid met Shirakawa at a coffee break and showed a sample of the golden (SN)x to Shirakawa. Consequently, Shirakawa also showed Mac. Diarmid a sample of the silvery (CH)x. 11/5/2020

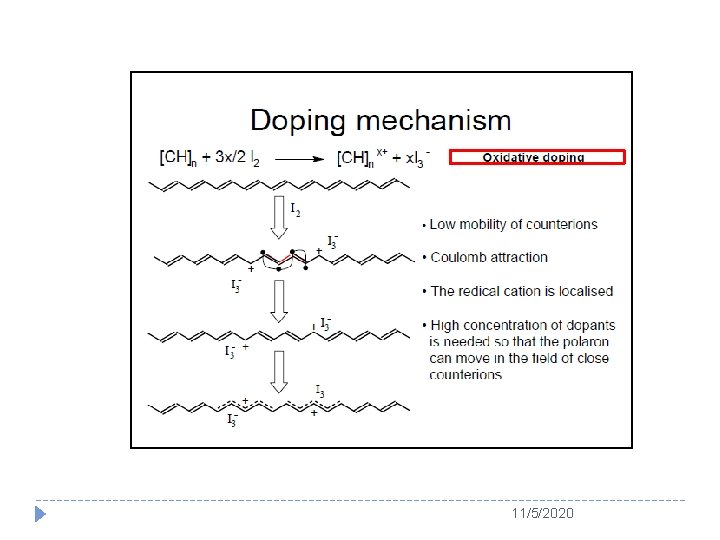
The Story of Polyacetylene � The beautiful silvery film caught the eyes of Mac. Diarmid and he immediately invited Shirakawa to the University of Pennsylvania PA. � Since Mac. Diarmid and Heeger had found previously that the conductivity of (SN)x could be increased by 10 times after adding bromine to the golden (SN)x material, which is called as “doping” item in inorganic semiconductor. � Therefore, they decided to add some bromine to the silvery (CH)x films to see what happens. � Miracle took place on November 23, 1976! � At that day, Shirakawa worked with Dr. C. K. Chiang, a postdoctoral fellow under Professor Heeger, for measuring the electrical conductivity of PA by a fourprobe method. 11/5/2020

The Story of Polyacetylene � Surprise to them, the conductivity of PA was ten million times higher than before adding bromine. This day was marked as the first time observed the “doping” effect in conducting polymers. � In the summer of 1977, Heeger, Mac. Diarmid, and Shirakawa co-published their discovery in the article entitled “Synthesis of electrically conducting organic polymers: Halogen derivatives of polyacetylene (CH)n” in The Journal of Chemical Society, Chemical Communications. 11/5/2020

Structural Characteristics and Doping Concept 11/5/2020
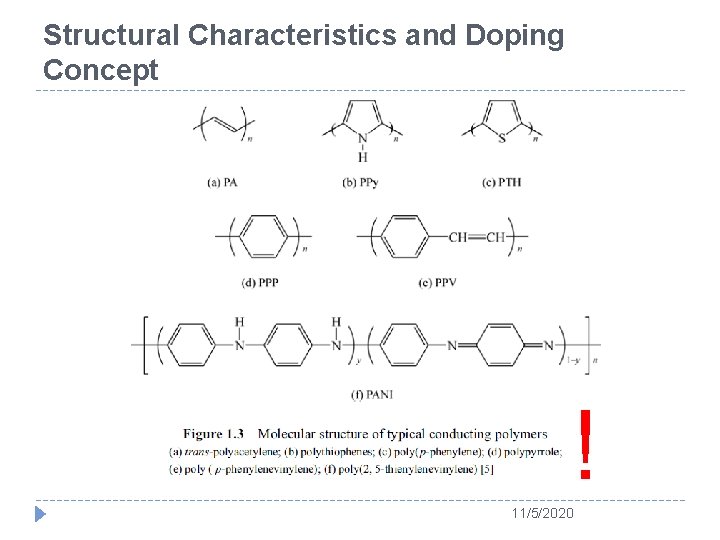
Structural Characteristics and Doping Concept ! 11/5/2020
11/5/2020
11/5/2020

The concept of Doping 11/5/2020

11/5/2020

Discovery of the First Conducting Polymer 11/5/2020
11/5/2020
11/5/2020
11/5/2020
11/5/2020

11/5/2020

The Charge Carriers 11/5/2020

11/5/2020
11/5/2020
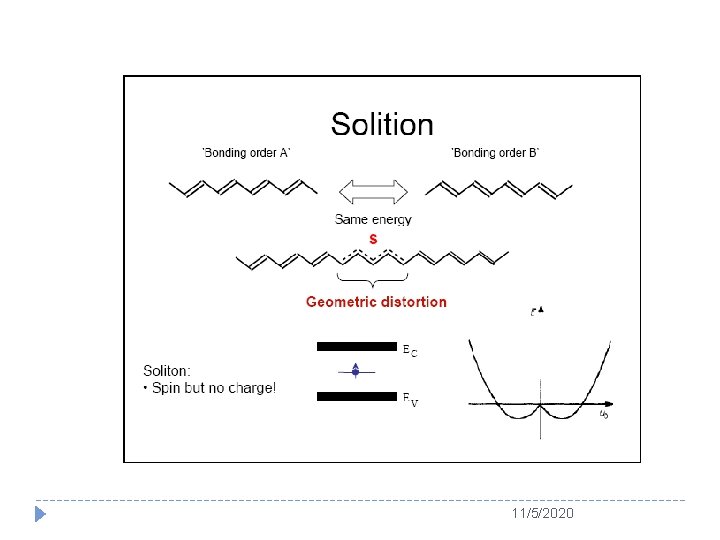
11/5/2020
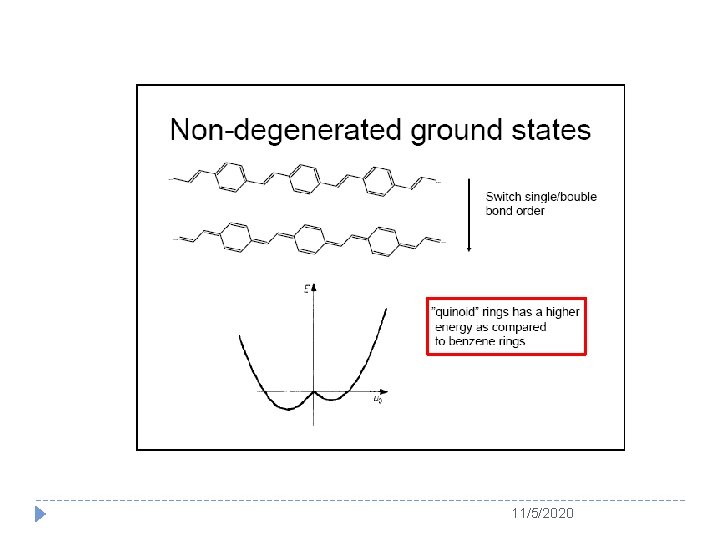
11/5/2020
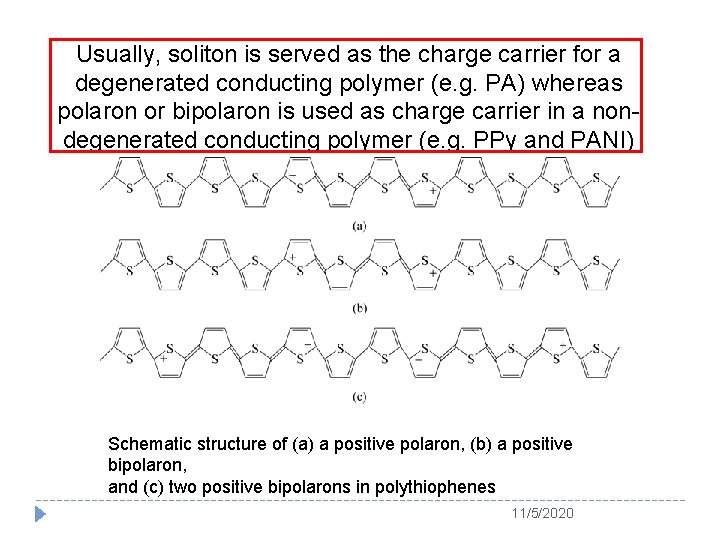
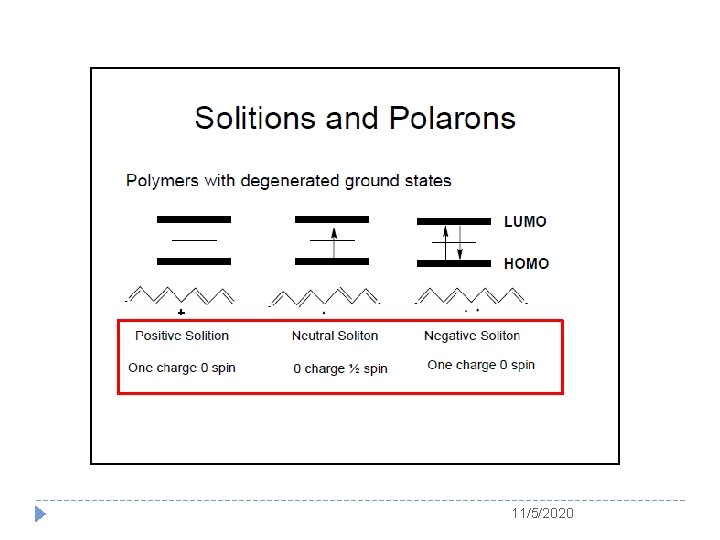
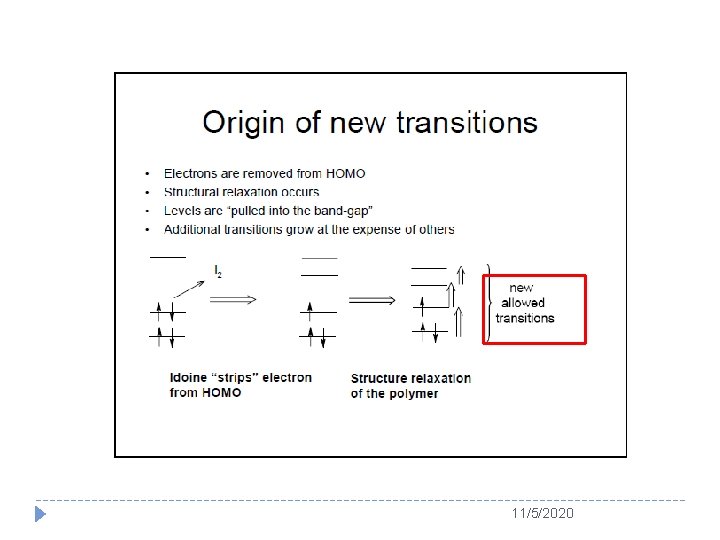
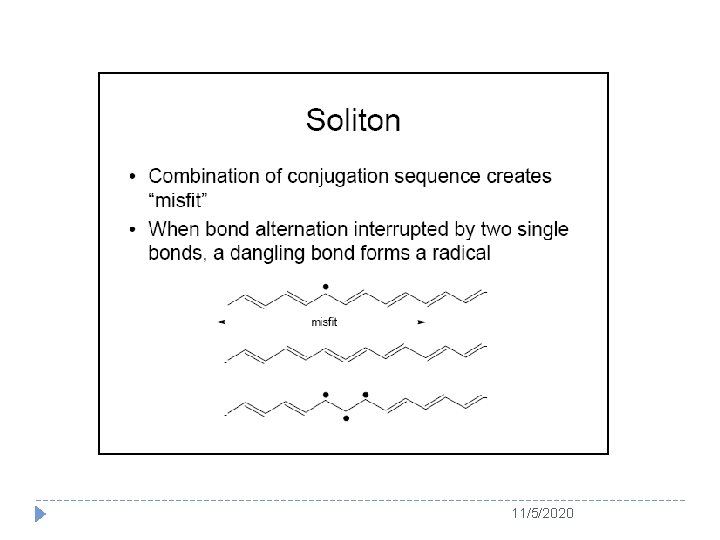
Usually, soliton is served as the charge carrier for a degenerated conducting polymer (e. g. PA) whereas polaron or bipolaron is used as charge carrier in a nondegenerated conducting polymer (e. g. PPy and PANI) Schematic structure of (a) a positive polaron, (b) a positive bipolaron, and (c) two positive bipolarons in polythiophenes 11/5/2020
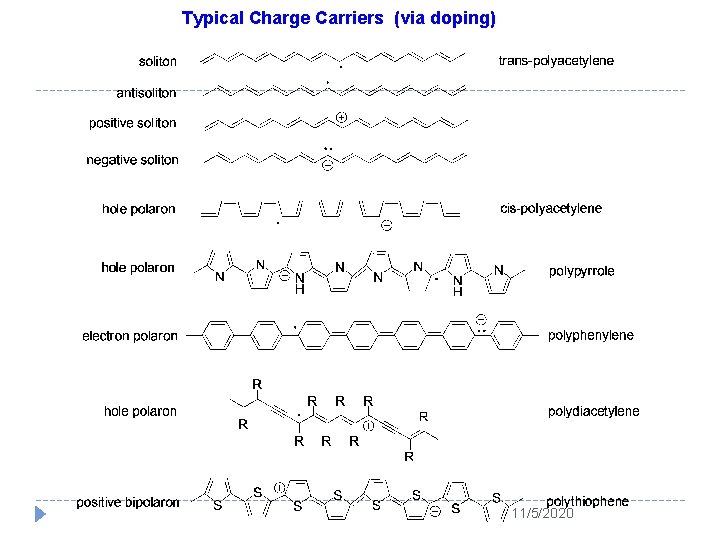
Typical Charge Carriers (via doping) 11/5/2020
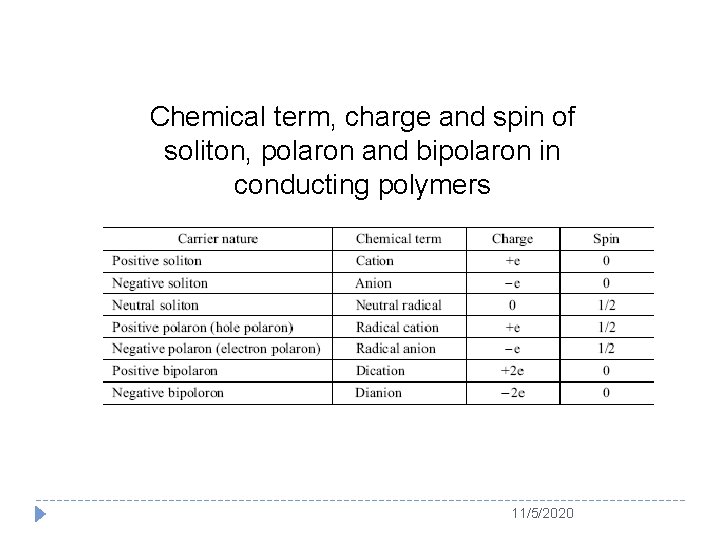
Chemical term, charge and spin of soliton, polaron and bipolaron in conducting polymers 11/5/2020

11/5/2020
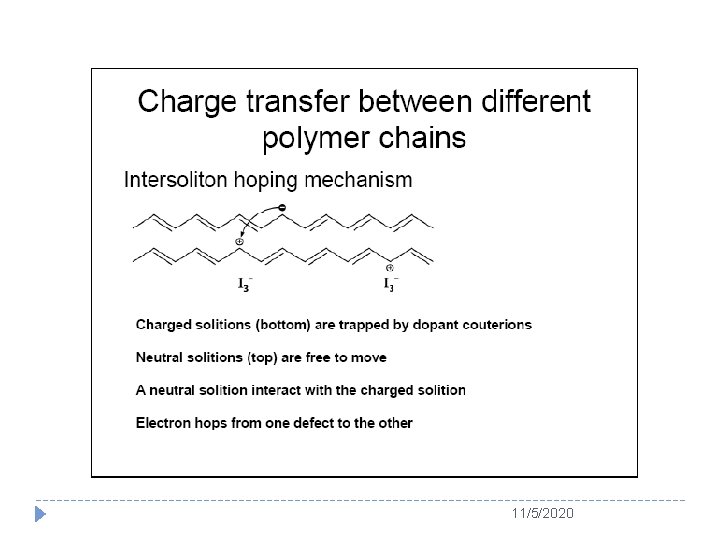
11/5/2020
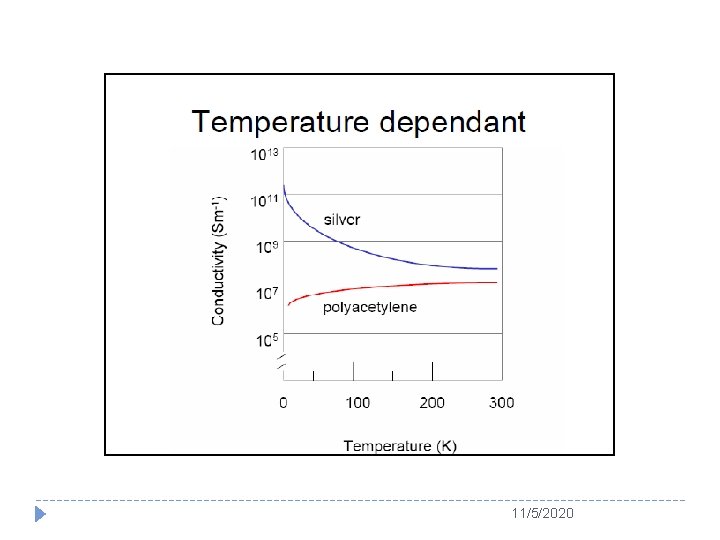
11/5/2020

• Filtration, membranes • Rechargeable batteries • Radar absorbers 11/5/2020

Applications 11/5/2020
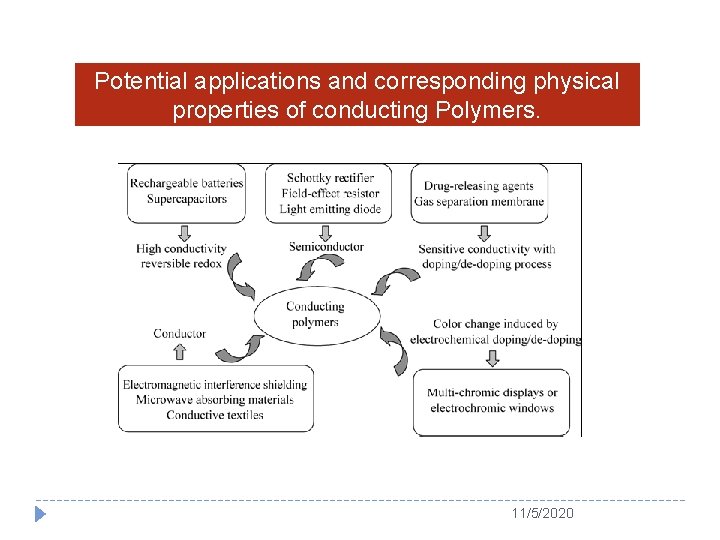
Potential applications and corresponding physical properties of conducting Polymers. 11/5/2020

11/5/2020
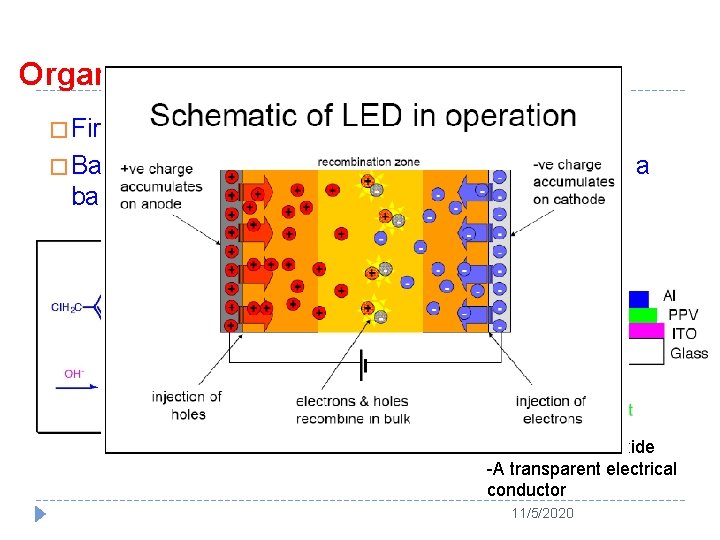
Organic Light Emitting Polymer � First reported in 1990 (Nature 1990, 347, 539) � Based on poly(p-phenylenevinylene) (PPV), with a bandgap of 2. 2 e. V ITO: Indium-tin-oxide -A transparent electrical conductor 11/5/2020

� Threshold for charge injection (turn-on voltage): 14 V (E-field = 2 x 106 V/cm) � Quantum efficiency = 0. 05 % � Emission color: Green � Processible ? No!! � Polymer is obtained by precursor approach. It cannot be redissolved once the polymer is synthesized 11/5/2020
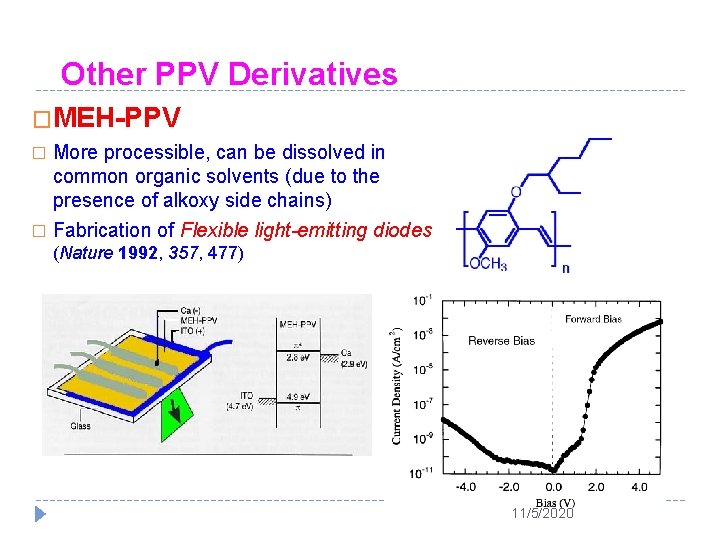
Other PPV Derivatives �MEH-PPV More processible, can be dissolved in common organic solvents (due to the presence of alkoxy side chains) � Fabrication of Flexible light-emitting diodes � (Nature 1992, 357, 477) 11/5/2020
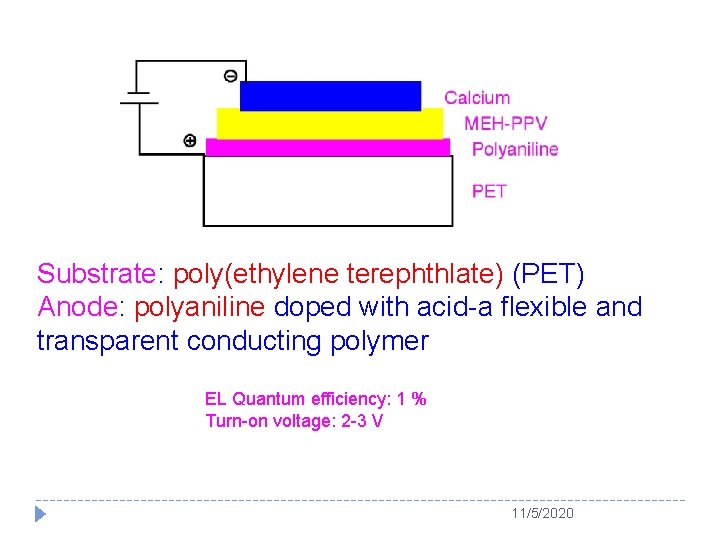
Substrate: poly(ethylene terephthlate) (PET) Anode: polyaniline doped with acid-a flexible and transparent conducting polymer EL Quantum efficiency: 1 % Turn-on voltage: 2 -3 V 11/5/2020
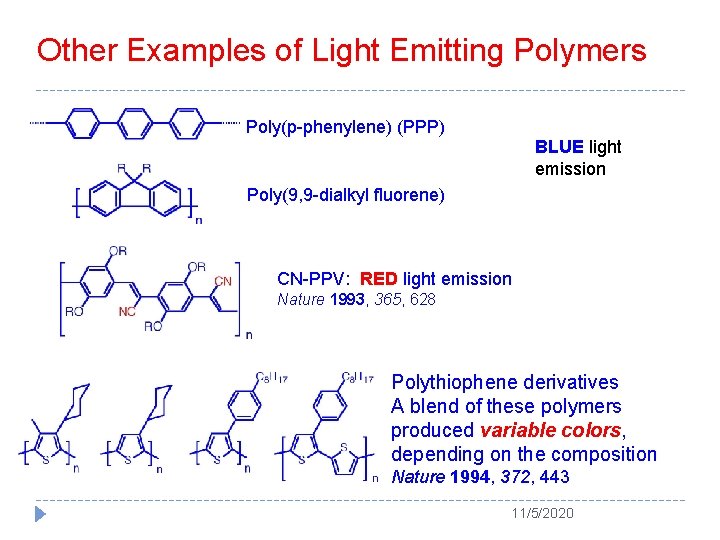
Other Examples of Light Emitting Polymers Poly(p-phenylene) (PPP) BLUE light emission Poly(9, 9 -dialkyl fluorene) CN-PPV: RED light emission Nature 1993, 365, 628 Polythiophene derivatives A blend of these polymers produced variable colors, depending on the composition Nature 1994, 372, 443 11/5/2020

Applications � Flat Panel Displays: thinner than liquid crystals displays or plasma displays (the display can be less than 2 mm thick) � Flexible Display Devices for mobile phones, PDA, watches, etc. � Multicolor displays can also be made by combining materials with different emitting colors. 11/5/2020

For an Electroluminescence process: Electrons Photons Can we reverse the process? Photons Electrons YES! Photodiode Production of electrons and holes in a semiconductor device under illumination of light, and their subsequent collection at opposite electrodes. Light absorption creates electron-hole pairs (excitons). The electron is accepted by the materials with larger electron affinity, and the hole by the materials with lower ionization potential. 11/5/2020
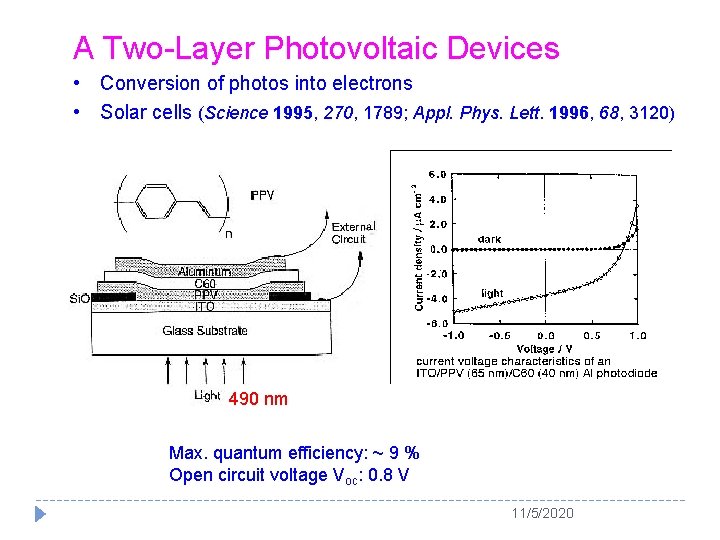
A Two-Layer Photovoltaic Devices • Conversion of photos into electrons • Solar cells (Science 1995, 270, 1789; Appl. Phys. Lett. 1996, 68, 3120) (Appl. Phys. Lett. 1996, 68, 3120) 490 nm Max. quantum efficiency: ~ 9 % Open circuit voltage Voc: 0. 8 V 11/5/2020
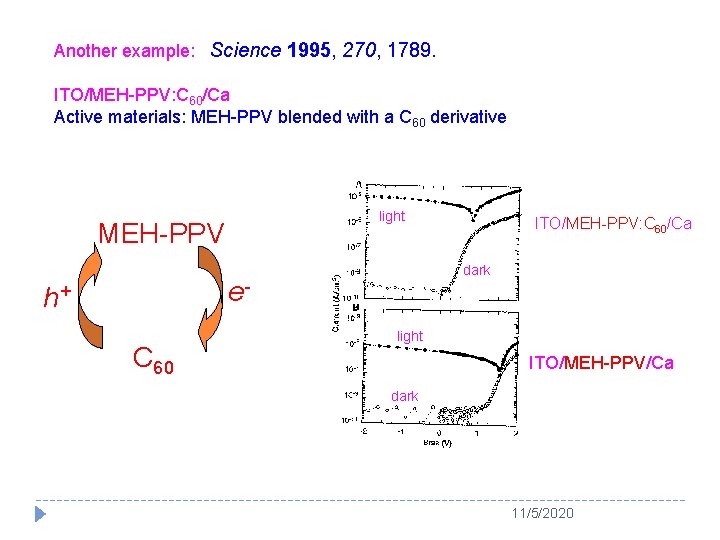
Another example: Science 1995, 270, 1789. ITO/MEH-PPV: C 60/Ca Active materials: MEH-PPV blended with a C 60 derivative light MEH-PPV dark e- h+ C 60 ITO/MEH-PPV: C 60/Ca light ITO/MEH-PPV/Ca dark 11/5/2020
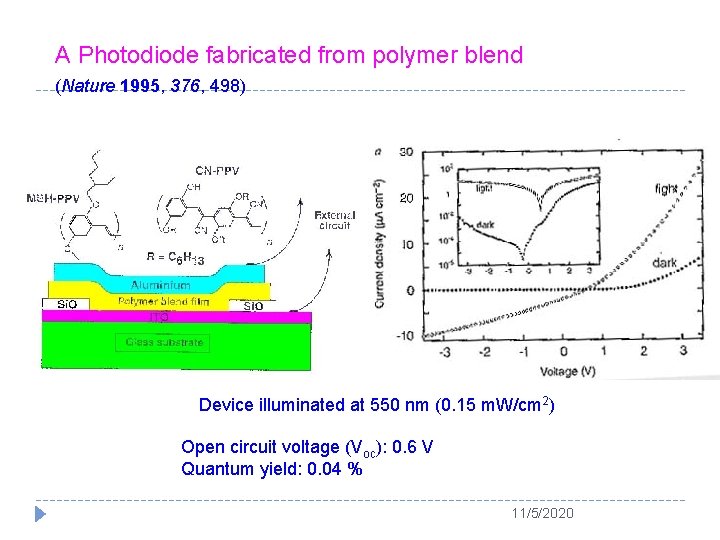
A Photodiode fabricated from polymer blend (Nature 1995, 376, 498) Device illuminated at 550 nm (0. 15 m. W/cm 2) Open circuit voltage (Voc): 0. 6 V Quantum yield: 0. 04 % 11/5/2020
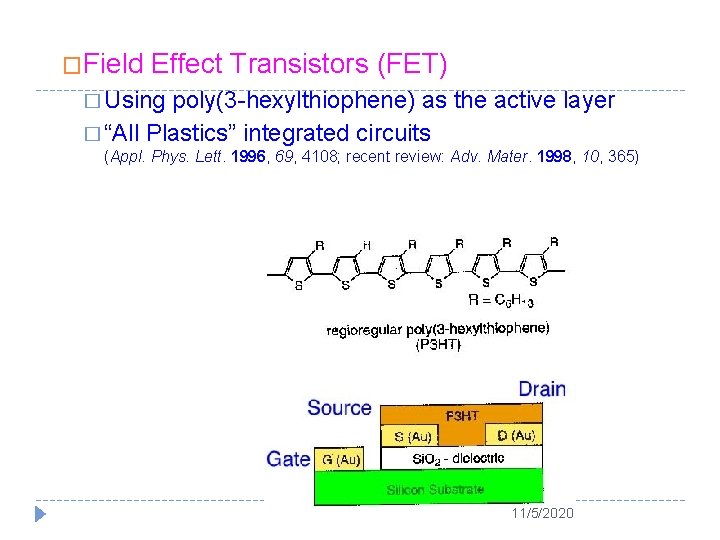
�Field Effect Transistors (FET) � Using poly(3 -hexylthiophene) as the active layer � “All Plastics” integrated circuits (Appl. Phys. Lett. 1996, 69, 4108; recent review: Adv. Mater. 1998, 10, 365) 11/5/2020

More Recent Development � Use of self-assembled monolayer organic field-effect transistors � Possibility of using “single molecule” for electronic devices (Nature 2001, 413, 713) 11/5/2020

Prof. Heflin's group is developing organic solar cells that have the potential to be flexible, lightweight, efficient renewable energy sources. Photograph by John Mc. Cormick. Polymer light-emitting diodes, such as the one produced by Martin Drees (Ph. D. 2003) in Prof. Heflin's laboratory, may potentially yield flexible, inexpensive flatpanel displays. http: //www. phys. vt. edu/~rheflin/ 11/5/2020

Prof. Heflin's group is examining how nanoscale control of the composition of organic solar cells consisting of semiconducting polymers and fullerenes can improve their power conversion efficiency. Prof. Heflin's group is using self-assembly of nanoscale organic films to create organic electrochromic devices that change color when a voltage is applied at rates up to 50 Hz. http: //www. phys. vt. edu/~rheflin/ 11/5/2020

Prof. Heflin's group is using self-assembly of nanoscale organic films to create organic electrochromic devices that change color when a voltage is applied at rates up to 50 Hz. http: //www. phys. vt. edu/~rheflin/ 11/5/2020

11/5/2020

Conductive Coatings � Replacement for ITO glass 11/5/2020

Conductive Coatings � Replacement for ITO glass 11/5/2020
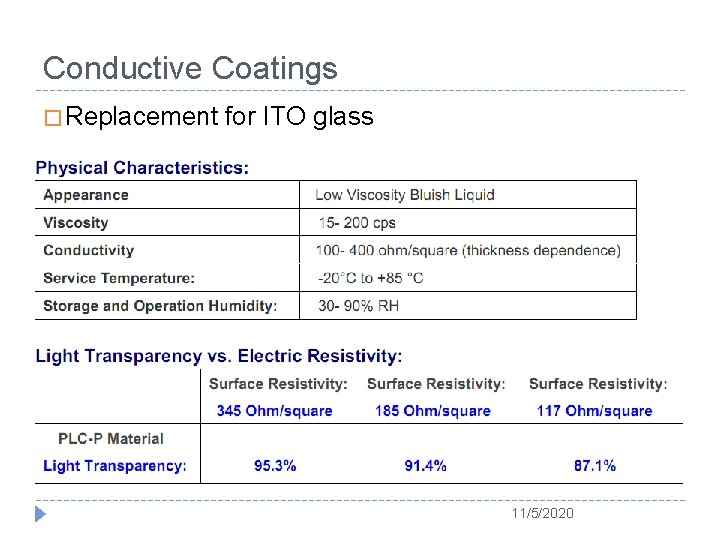
Conductive Coatings � Replacement for ITO glass 11/5/2020

Synthesis Information Available for interested Students 11/5/2020

Electropolymerization Information Available for Interested Students 11/5/2020

The End 11/5/2020
- Slides: 63