Substances Melting Point C Boiling Point C Astanine
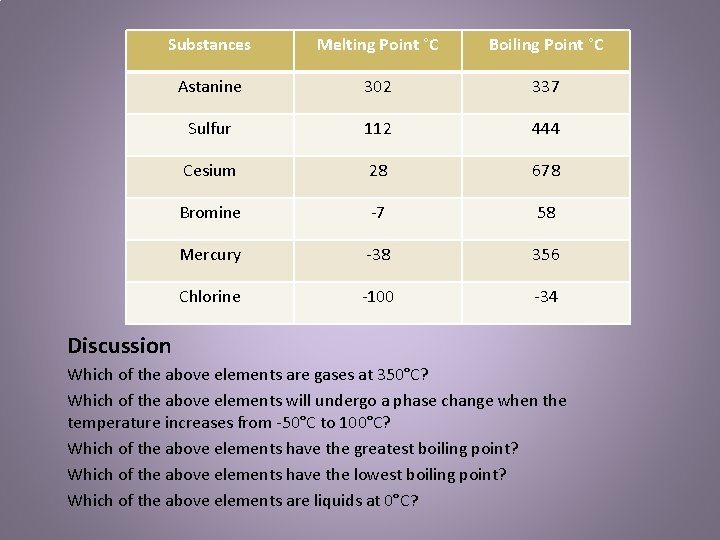
Substances Melting Point °C Boiling Point °C Astanine 302 337 Sulfur 112 444 Cesium 28 678 Bromine -7 58 Mercury -38 356 Chlorine -100 -34 Discussion Which of the above elements are gases at 350°C? Which of the above elements will undergo a phase change when the temperature increases from -50°C to 100°C? Which of the above elements have the greatest boiling point? Which of the above elements have the lowest boiling point? Which of the above elements are liquids at 0°C?
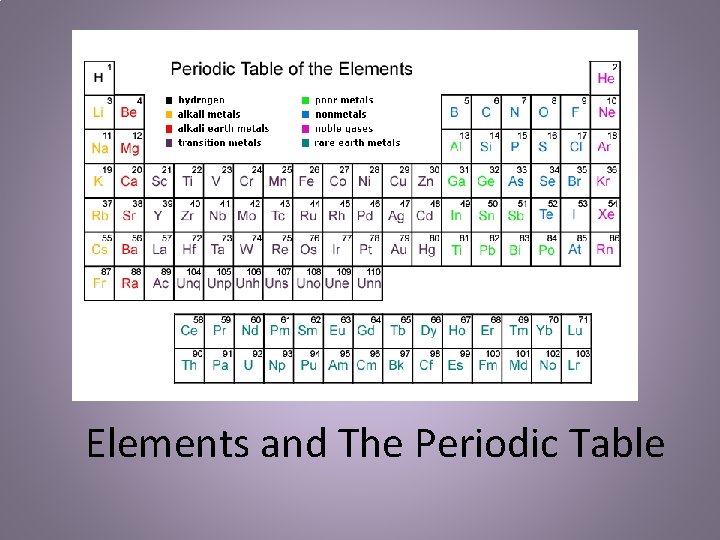
Elements and The Periodic Table

Elements & Compounds • Elements: simplest form of matter • Compounds: combination of two or more atoms combined with new – Building blocks of all properties/characteristics than the matter individual elements did alone – All represented on the periodic table with an element symbol. • 1 Letter – Capital • 2 Letters – 1 st Capital, 2 nd Lowercased – Sugar: Carbon (black solid) + Water (colorless liquid) – Water: Oxygen (colorless, non-flammable gas) + Hydrogen (colorless, flammable gas) Compounds are in definite proportions and written in chemical formulas. Most chemical formulas contain subscripts to represent the proportions to each element in the compound.

History of the Periodic Table • Dmitri Mendeleev, Russian chemist, hoped to organize the elements according to their properties. • Placed each element on a card, with atomic mass, and physical and chemical properties. • Then arranged them according to properties and trends or patterns. • Noticed that they were arranged in order of increasing atomic mass, similarities in their chemical properties appeared at regular intervals as well. Thus the name periodic, repeating pattern. • His for table was produced in 1869. You can notice a few of the elements seem “out of place” from where they currently are placed.


History of the Periodic Table • In 1911, English scientist – Moseley, worked with Rutherford (gold foil experiment man) examined the spectra of 38 different metals. • Discovered that the elements fit better into the periodic table if they were arranged by the number of protons in the nucleus. • Thus atomic number, not atomic mass. • Moseley’s discovery was consistent with Mendeleev’s ordering of the periodic table by properties rather than strictly by atomic mass. • Mendeleev’s principle of chemical periodicity is correctly stated in what is known as the periodic law – the physical and chemical properties of the elements are periodic functions of their atomic number.

Current Periodic Table • The periodic table is an organized way of looking at the elements. – The elements are arranged according to similarities in properties – Many scientists have worked on refining the periodic table. It started with Mendeleev who organized the elements by the atomic mass doing up and down to Moseley who organized the elements going horizontally by atomic number (#of protons) – There a few different key points in the periodic table: • Groups/Families – vertical columns in which the elements have similar physical and chemical properties – 1 A-8 A are the representative elements in the main group elements. They represent all states of matter: s, l, g – 1 B-8 B are the transition elements. As you move from left to right across the middle of the table the elements transition from metals to nonmetals. » Inner Transition Metals – two rows on the bottom – rare earth metals and those not found in nature • Periods – horizontal rows – Number of elements period: 2, 8, 8, 18, 32
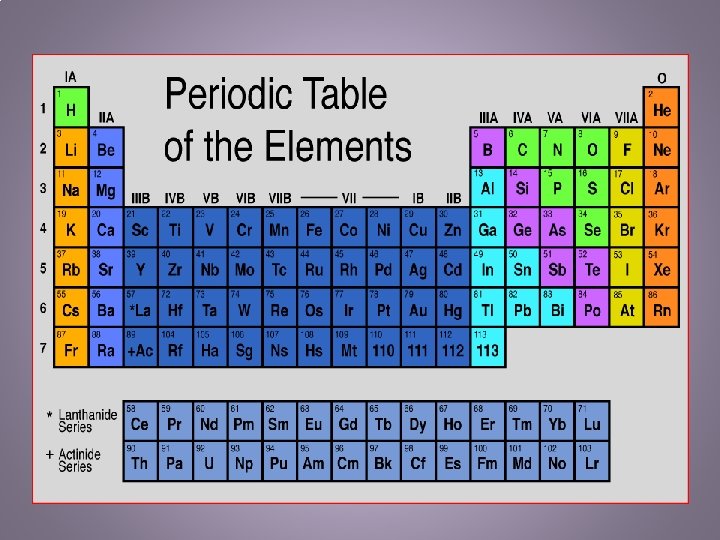

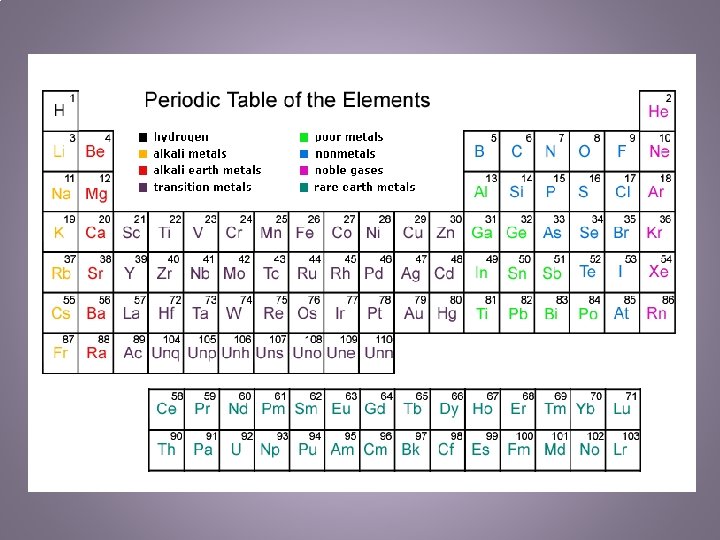
Metals, Nonmetals, Metalloids • Metals – 80% of all known elements are metals – Characteristics are based on their functional properties as well as their appearances – Physical: shiny, malleable, ductile, tensile strength, solid (not Hg), conductivity • Nonmetals – 20% of known elements are nonmetals – Found on the right side of the periodic table – Physical: hard, brittle, gases (not Br, I, S, P, C, Se), poor conductor • Metalloids – Border both sides of the “stairs” – Have some metal and nonmetal properties – 7 Solids: B, Si, As, Te, At, Ge, Sb – Used in computers, digital watches, TVs, radios

- Slides: 11