Substances and Mixtures What are Substances Pure Substance

Substances and Mixtures

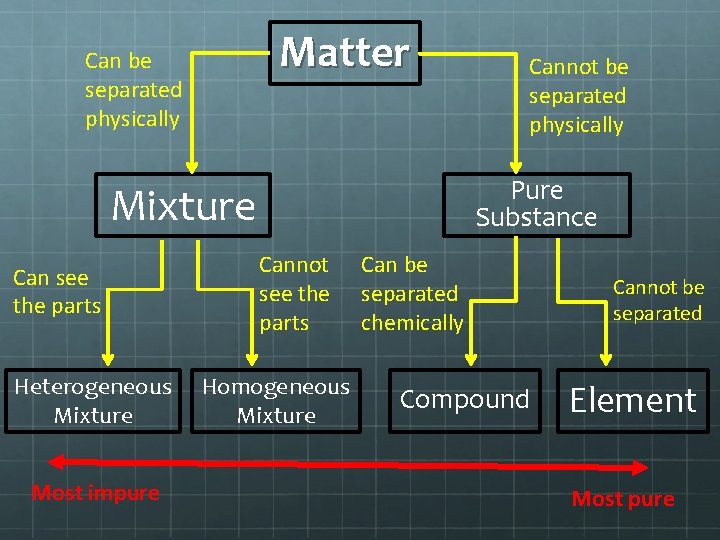
What are Substances? Pure Substance Matter that has only 1 set of chemical and physical properties 2 types of Pure Substances Element = cannot be broken down any more Compound = composed of 2 or more elements Compounds can ALWAYS be broken down into simpler substances, elements cannot

How are Compounds Broken Down? Cannot be broken down physically We cannot throw salt at the wall and break it down in Na+(aq) and Cl-(aq) Must be done chemically Run a chemical reaction We get(typically) 2 or more elements

What is a Mixture? Mixture 2 or more pure substances mixed together physically 2 types of Mixtures Homogeneous Heterogeneous Difference between mixture and substance Mixtures can be separated with physical means

Heterogeneous Mixture What is a heterogeneous mixture? Composition is not uniform throughout the mixture Examples Chicken noodle soup Smoke Chocolate chip cookies Types of heterogeneous mixtures Colloids Particles are very, very small that DO NOT settle Suspensions Particles are big; visible to us; settle back down Course Mixtures Particles are big enough to be separated by hand.

Homogeneous Mixture What is a homogeneous mixture? The composition is uniform throughout Examples Salt water Air Stainless steel Types of homogenous mixtures Solutions Just another name for homogenous mixture

How Do We Separate Mixtures? Two ways of separating mixtures Filtration Separating a solid from a liquid Straining spaghetti Distillation Separating small particles from a mixture Boil the mixture and collect the gas Making Distilled water
Matter Can be separated physically Pure Substance Mixture Can see the parts Heterogeneous Mixture Most impure Cannot be separated physically Cannot see the parts Homogeneous Mixture Can be separated chemically Compound Cannot be separated Element Most pure

Examples Milk Mixture (low-fat, skim, whole, 2% etc. ) Diamond Substance (carbon doesn’t change) Gasoline Mixture (premium, regular, and unleaded) Stainless Steel Mixture (carbon and iron together) Silver Substance (an element, doesn’t change)

Distinguishing Between a Substance and a Mixture? Easy Rule of Thumb If you have an single element or multiple elements together Substance If you have more than 1 compound together Mixture

Mixtures vs substances lab Answer the pre-lab questions first with your partner
- Slides: 11