Substances and Mixtures Post Lab Sessions Activity 7

Substances and Mixtures Post Lab Sessions Activity 7, 8 A & 8 B

True or False? 1. Mixtures are made up of two or more substances. 2. Mixtures can be solids, liquids or gases. 3. Mixtures are always homogeneous. 4. Solutions have very small particles that the naked eye cannot see. 5. Solutions have uniform appearance.

Activity 7 • Seawater! See water and salt! distilled water

salt is seen on the dish

Bottomline! A mixture, whether heterogeneous or homogeneous, is made up of two or more substances.

How do we know that distilled water and salt are substances? What is the difference between a mixture and a substance?

Activity 8 A Objectives - assemble properly the setup for boiling; - describe the change in temperature of a substance while boiling; - describe the change in temperature of a mixture while boiling; - differentiate between substance and mixture based on how temperature changes during boiling.

Distilled Water Appearance Temperature (0 C) at start of Boiling Temp 30 sec eratur 60 sec 0 e ( C) after 90 sec 120 sec 150 sec Salt Solution
Appearance Temperature (0 C) at start of Boiling Temp 30 sec eratur 60 sec 0 e ( C) after 90 sec 120 sec 150 sec Distilled Water clear Salt Solution clear
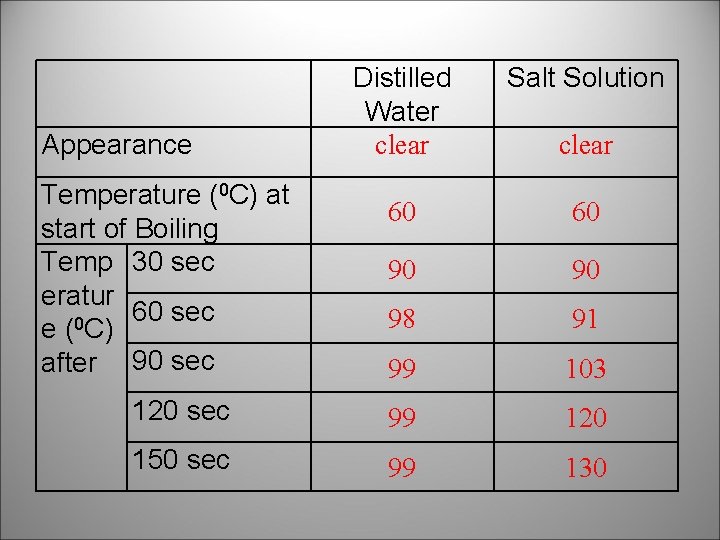
Distilled Water clear Salt Solution 60 60 90 90 98 91 99 103 120 sec 99 120 150 sec 99 130 Appearance Temperature (0 C) at start of Boiling Temp 30 sec eratur 60 sec 0 e ( C) after 90 sec clear
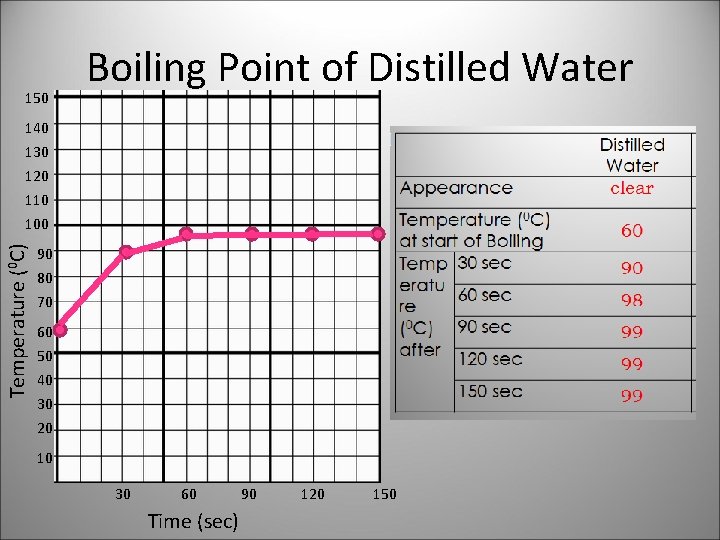
150 Boiling Point of Distilled Water 140 Temperature (0 C) 130 120 110 100 90 80 70 60 50 40 30 20 10 30 60 Time (sec) 90 120 150
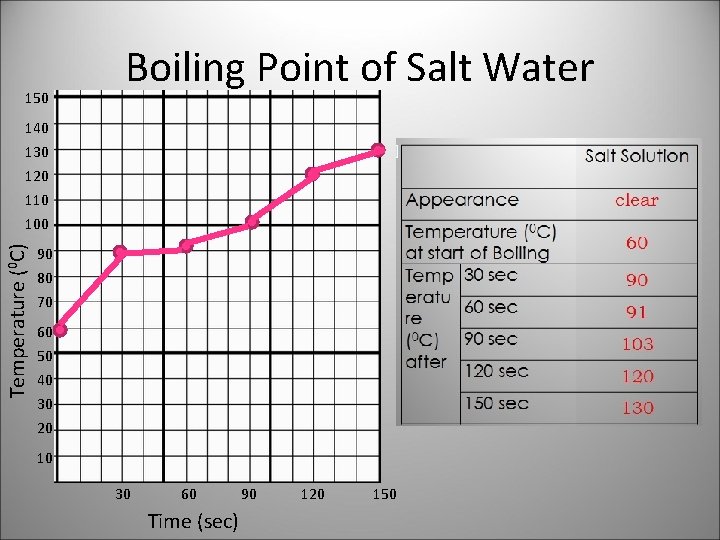
150 Boiling Point of Salt Water 140 Temperature (0 C) 130 120 110 100 90 80 70 60 50 40 30 20 10 30 60 Time (sec) 90 120 150
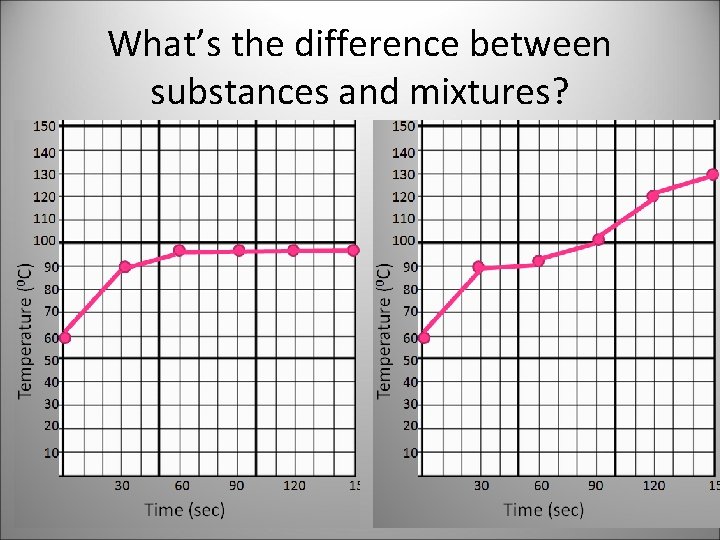
What’s the difference between substances and mixtures?

Substance has a definite boiling point, while a mixture has varying boiling points.

How can the varying boiling points in a mixture be explained? • Different substances have different boiling points. • Mixtures are made up of different substances. • Therefore, each of the substances in a mixtures will boil at different temperature.

What is the boiling point of water? Water boils at 1000 C. At what temperature will 50 m. L of water boil? 50 m. L of water will boil at 0 100 C.

The Water Problem • Hicks prepared a liter of water and heated it until it boils. He got the temperature and read 1000 C. He prepared 5 m. L of water and heated it until it boils. He predicts that thermometer will also read 1000 C when the water boils. Is his prediction correct? Propose an explanation for this using the result of Activity 8 A.

Water is a substance and thus, it has a definite boiling point. The boiling point will always be same no matter how much volume of water will be heated. Boiling point is an INTRINSIC PROPERTY!

What are intrinsic properties? • An intrinsic property is a characteristic of matter that DOES NOT change according to quantity. • The property will always be same no matter how much sample is present. Think of other intrinsic properties!

Meanwhile… Substance has a definite boiling point, while a mixture has varying boiling points.

Activity 8 B Objectives • assemble properly the setup for melting; • describe the appearance of a substance while it is melting; • describe the appearance of a mixture while it is melting; and • differentiate between substances and mixtures based on how they appear as they melt.


Benzoic Acid – Salt Mixture Appearance

Appearance Benzoic Acid – Salt Mixture The sample After 1 melted minute, some completely parts of the after 1 minute. mixture did not melt.

Why didn’t the entire mixture melt? • The melting point of benzoic acid is 122. 40 C. • The melting point of salt is 8010 C. Which part of the mixture did not melt in our experiment?

Substance has a definite melting point, while a mixture has varying melting points.

Summing up… Substance has a definite boiling and melting point, while a mixture has varying boiling and melting points.
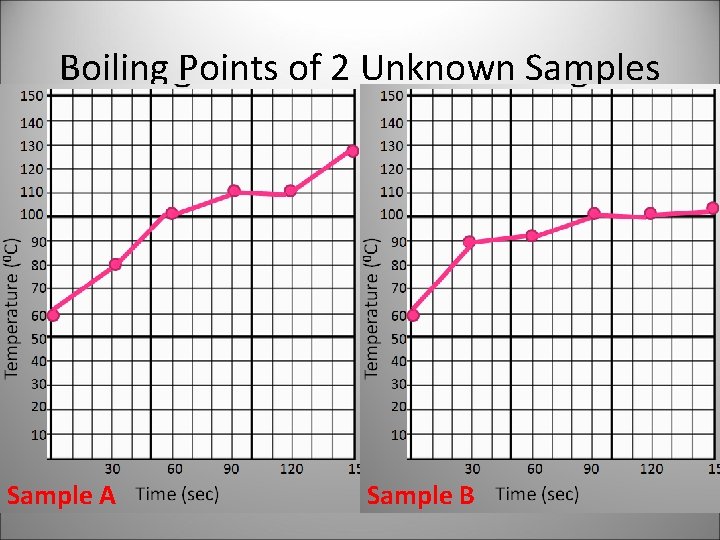
Boiling Points of 2 Unknown Samples Sample A Sample B
- Slides: 29