Substance Vs Mixture Aim how can we further

Substance Vs. Mixture Aim: how can we further break down and classify different pieces of matter?

Let’s talk about salt! WOAH!!! SALT!!!! SO MUCH SALT! I know salt. That is defiantly salt.

“Ms. Wiswall, why did we talk about salt and what it’s made of? ” • I wanted to give you a better idea about how different a compound is from a element it’s originally made of. I also wanted to introduce the words “element” and “compound”

VI. The Classification of Matter a) Substances: Elements and Compounds 1. ELEMENTS ARE PURE (homogeneous) SUBSTANCES THAT CANNOT BE BROKEN DOWN (decomposed) CHEMICALLY INTO SIMPLER KINDS OF MATTER. • More than 100 elements have been identified, though Fewer than 30 are Important in Living Things. • All of the Elements are arranged on a Chart known as THE PERIODIC TABLE. • Among the information provided in The Periodic Table are the ATOMIC NUMBER, THE CHEMICAL SYMBOL, AND THE ATOMIC MASS FOR EACH ELEMENT.
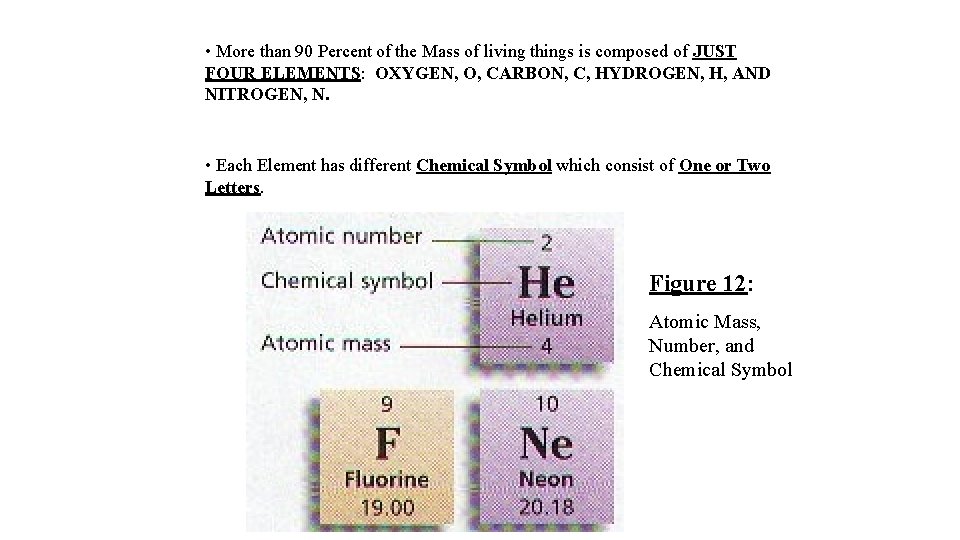
• More than 90 Percent of the Mass of living things is composed of JUST FOUR ELEMENTS: OXYGEN, O, CARBON, C, HYDROGEN, H, AND NITROGEN, N. • Each Element has different Chemical Symbol which consist of One or Two Letters. Figure 12: Atomic Mass, Number, and Chemical Symbol
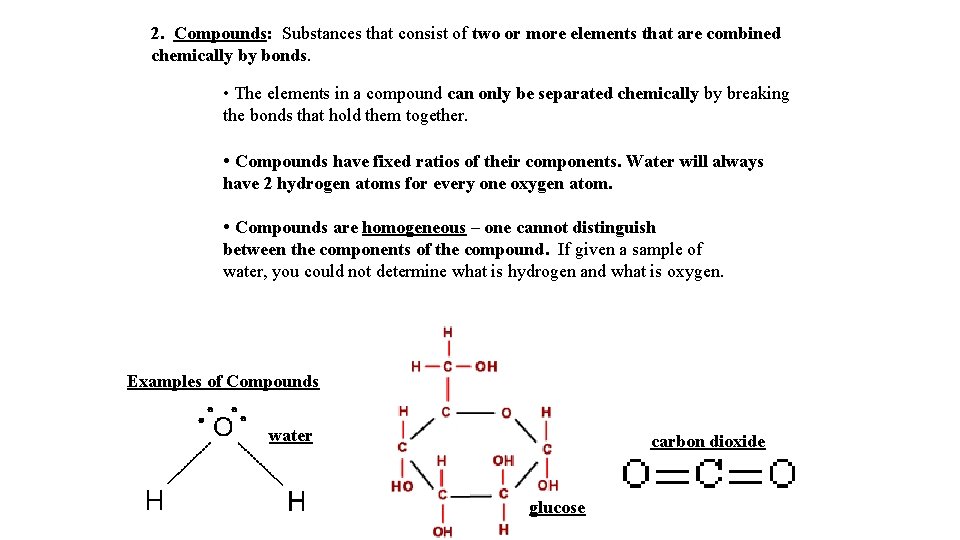
2. Compounds: Substances that consist of two or more elements that are combined chemically by bonds. • The elements in a compound can only be separated chemically by breaking the bonds that hold them together. • Compounds have fixed ratios of their components. Water will always have 2 hydrogen atoms for every one oxygen atom. • Compounds are homogeneous – one cannot distinguish between the components of the compound. If given a sample of water, you could not determine what is hydrogen and what is oxygen. Examples of Compounds water carbon dioxide glucose

b) Mixtures • Two or more substances that are combined physically; the components can be easily separated. • In homogeneous mixtures, the substances are completely mixed. This means that you cannot see the individual components. The mixture appears to be only one substance. • In heterogeneous mixtures, the substances are not completely mixed. This means that you can see the individual components. The mixture appears to be only two different substances in the same container. Figure 14: Heterogeneous Mixture: SOIL Figure 13: Homogeneous Mixture: Salt Water
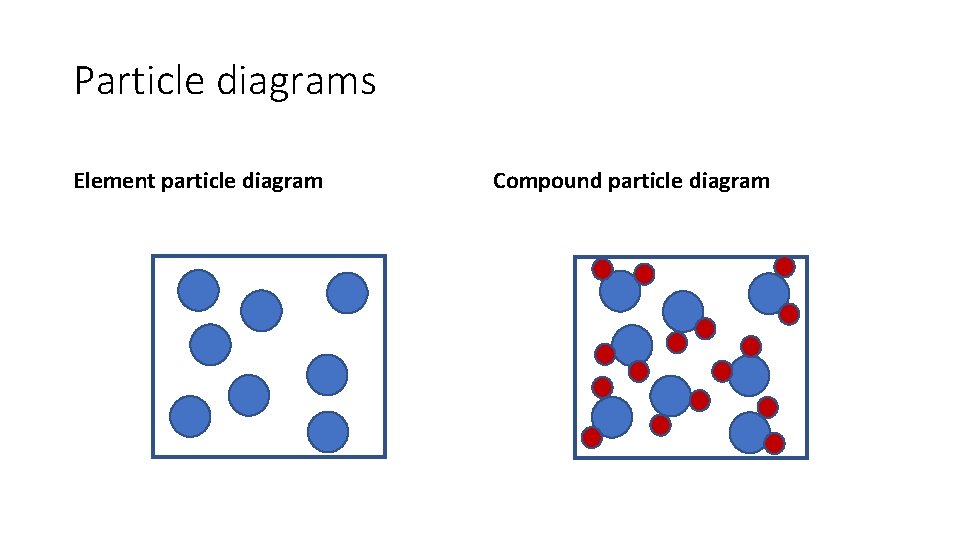
Particle diagrams Element particle diagram Compound particle diagram
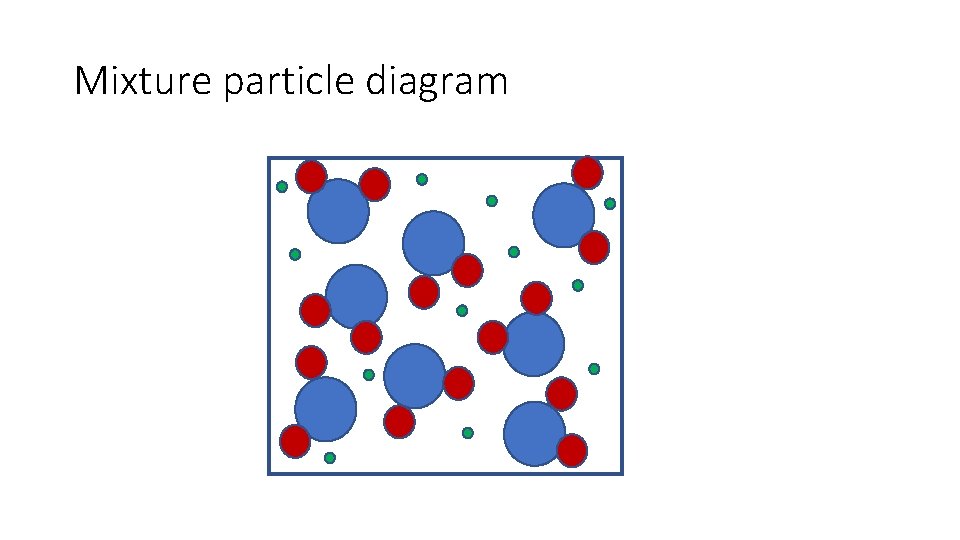
Mixture particle diagram
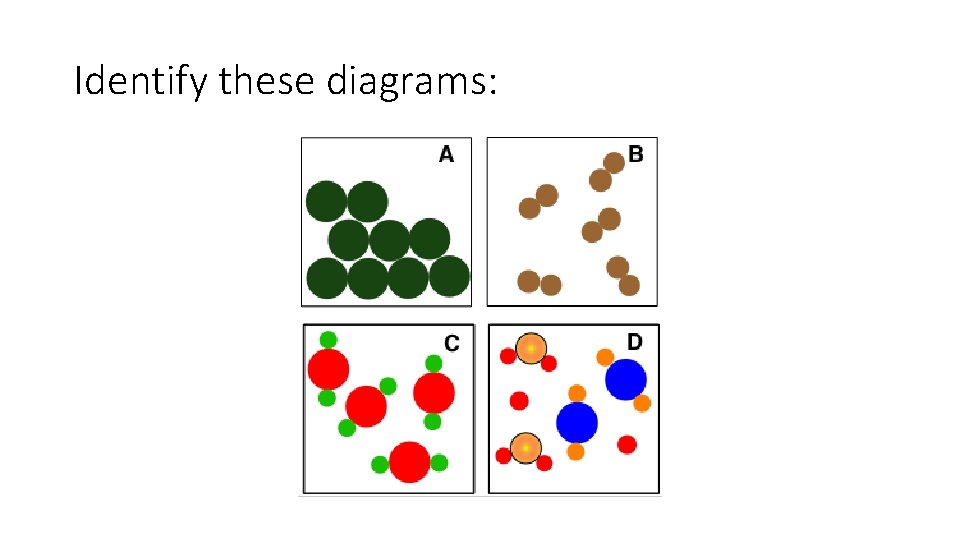
Identify these diagrams:
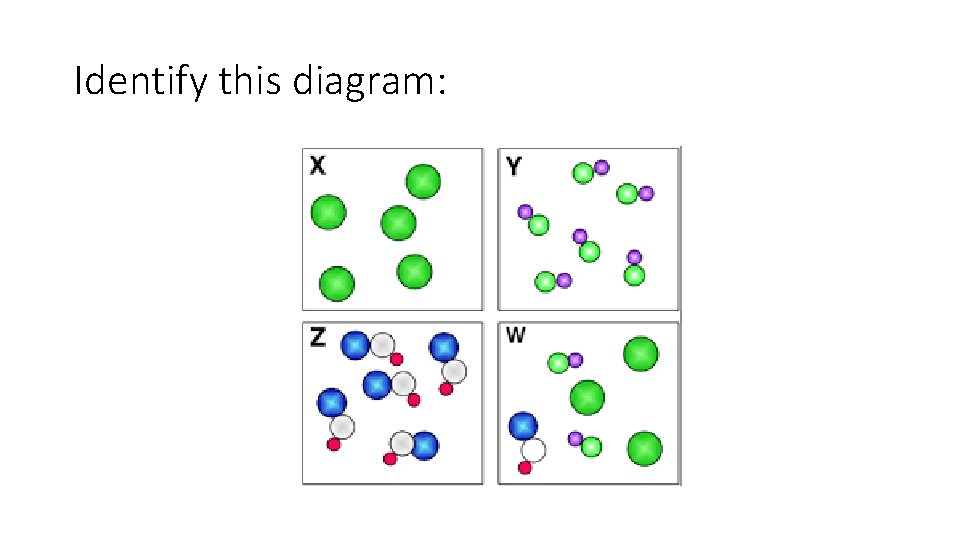
Identify this diagram:

Ex: #5 A compound differs from a mixture in that a compound always has a a) homogeneous composition b) maximum of two components c) minimum of three components d) heterogeneous composition Ex: #6 Which substance cannot be decomposed into simpler substances? a) ammonia (NH 3) b) aluminum (Al) c) methane (CH 4) d) methanol (CH 3 OH)

Physical Vs Chemical Properties Physical Properties- Properties that can be observed WITHOUT producing a new substance • Examples include: • • • Color Hardness Density Mass Melting point Volume Weight Malleability Ductility Odor Boiling point Chemical Properties- describes how a substance interacts (of fails to interact) with other substances to produce new substances • Combustibility • Failure to react with other substances • Tendency to corrode • Bubbles forming

Extensive physical Vs Intensive physical properties Extensive properties- physical properties that depends on the amount of matter in a sample Intensive properties- physical properties that do not depend on the amount of matter in a sample • Mass • Volume • Weight • Size • Length • Density • Melting point • Boiling point • Color • Hardness • Odor malleability
- Slides: 14