Substance Use Disorders in Pregnancy Julianna Finelli MD

Substance Use Disorders in Pregnancy Julianna Finelli, MD Assistant Professor of Psychiatry, Tulane University School of Medicine Lead Psychiatrist, Louisiana Mental Health Perinatal Partnership (LAMHPP)

ECHO guidelines • Please remain on ‘mute’ except when speaking • Always maintain patient confidentiality (avoid use of names or other identifiers) • Please ask questions by raising your hand • Sessions are recorded • Please sign in using the chat function (name/title/e-mail address)

Acknowledgement This publication was produced by the U. S. Department of Health and Human Services, Health Resources and Services Administration, Maternal and Child Health Bureau under contract number UK 3 MC 32243. This publication lists nonfederal resources in order to provide additional information to consumers. The views and content in these resources have not been formally approved by the U. S. Department of Health and Human Services (HHS) or the Health Resources and Services Administration (HRSA). Listing these resources is not an endorsement by HHS or HRSA.
LAMHPP Team Principal Investigator Gina Easterly Ph. D CCC-SLP Medical Director Mary Margaret Gleason MD Perinatal Psychiatry Expert Program Manager Tracey Thomas MPA Mental Health Specialist Region 4 Jen Bordelon LMSW Renee Bruno, MD Mental Health Specialist Region 8 Ilana Wahlder MEd LPC Lead Psychiatrist Julianna Finelli, MD Yasaman Hemmat, MD Steven Stein, MD IEMH Consultant Anna Kelley, Psy. D Nithya Ravindran, DO

Goals of LAMHPP Among health care clinicians who care for pregnant and postpartum women… Increase universal screening for depression, anxiety, substance use disorders (SUDs), and intimate partner violence (IPV) Increase early detection, evidence-informed first line management, and referral to specialty evidence-based treatments and culturallyappropriate community supports as needed Support referral and connections to services, treatment and recovery support in underserved and rural areas of Louisiana. Develop plan for sustainability of consultation and care coordination beyond the duration of the funding.

Other services • Webinars • Web site – – Care guides Clinical handouts Screening tools Statewide resources/referral information • • • Mental health Substance use Basic needs Infant mental health Peer support • Limited onsite consultation/telepsychiatric consultation

Scope of the problem

Women and substance use • From 1999 through 2014, opioid overdose deaths increased more than 4 -fold among women of reproductive age (CDC, 2017) • Women are more likely than men to experience chronic pain and use prescription opioid pain medications for longer periods and in higher doses • Sex based differences in substance use – Hormonal changes (menstrual cycle, pregnancy, menopause) – Body fat percentages, metabolic rate • Few programs offer women-targeted treatment Centers for Disease Control and Prevention. Analysis of the National Vital Statistics System Multiple Cause of Death data, Wide-ranging On. Line Data for Epidemiologic Research (WONDER). 2017. Prescription Painkiller Overdoses. CDC Vital Signs; July 2013. http: //www. cdc. gov/vitalsigns/prescriptionpainkilleroverdoses/index. html.
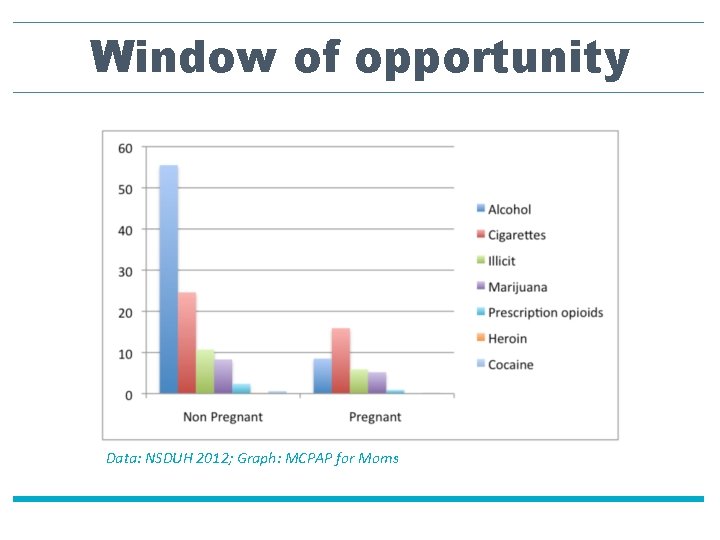
Window of opportunity Data: NSDUH 2012; Graph: MCPAP for Moms

Effects on the developing child • Estimated 12% of children in U. S. live with a parent with a substance use disorder (SAMHSA, Office of Applied Studies, 2009) – – – – Increased risk for maltreatment Increased risk for entering child welfare system Unpredictable environment and caregiving Estrangement from social supports Reduced capacity to respond to child’s cues and needs Difficulty regulating emotions Disrupted parent-child attachment • “Difficult regulatory partners” (Pajulo et al, 2008) Pajulo M, Suchman N, Kalland M, Sinkkonen J, Helenius H, Mayes L (2008). Role of maternal reflective ability for substance abusing mothers. J Prenat Perinat Psychol Health. 23(1): 13 -31.

Silos SUD OB/GYN Pediatrics Mental Health Treatment

Social determinants of health Intimate partner violence

How we talk to our partners about screening

Screening recommendations • Screening ≠ urine toxicologic screen! • Importance of consent • Maintain nonjudgmental stance – Can help to first ask about behaviors before pregnancy • Ask about every substance – – Amount used Frequency Last Use Route of administration • Screen for co-occurring mood anxiety disorders

Legal considerations

ACOG Committee Opinion “Drug enforcement policies that deter women from seeking prenatal care contrary to the welfare of the mother and fetus. Incarceration and the threat of incarceration have proved to be ineffective in reducing the incidence of alcohol or drug abuse. OB-GYNs should be aware of the reporting requirements related to alcohol and drug abuse within their states. They are encouraged to work with state legislators to retract legislation that punishes women for substance abuse during pregnancy. ” ACOG Committee on Health Care for Underserved Women, Number 473, January 2011

LA mandatory reporting • Prenatal neglect definition, Children's Code Article 603(16. 1) – ”…the unlawful use by a mother during pregnancy of a controlled dangerous substance, as defined by R. S. 40: 961 et seq. , which results in symptoms of withdrawal in the infant or the presence of a controlled substance in the infant's body. ” (emphasis added)

Physician mandatory reporting Cause to believe newborn exposed inutero to unlawfully used controlled dangerous substance? Must order toxicology test on newborn If positive, must make report of suspected prenatal neglect Newborn has symptoms of withdrawal or other observable effects due to chronic or severe use of alcohol or FAS? Prenatal neglect suspected but test results cannot confirm exposure prior to discharge from hospital? Newborn has withdrawal sx believed due to lawful use of controlled dangerous substance? Must make report of suspected prenatal neglect Must submit Physician Notification Form

Risks and Treatment

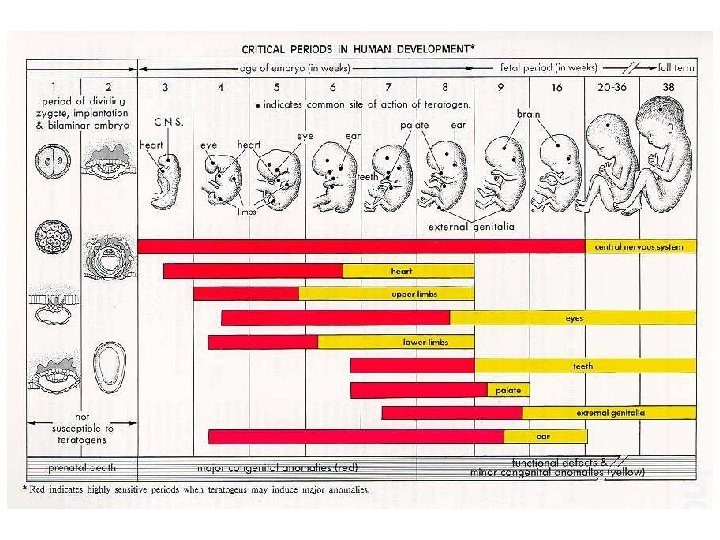
Teratogenic vs Adverse Fetal Effects What? When? Examples Teratogenic effects Cause alteration of Early in pregnancy tissue development or organ formation Neural tube defects; congenital heart defects; cleft lip/palate Adverse fetal effects Result in Later in pregnancy dysfunction of organ or tissue after it has been formed Difficult postnatal adaptation; withdrawal; electrolyte abnormalities; altered glucose metabolism

Opioid Use • Not likely direct structural teratogens • Opioid Use Disorder in pregnancy associated with: – Intrauterine growth restriction (IGR) – Intrauterine fetal demise and stillbirth – Preterm labor – Placental abruption – Postpartum hemorrhage – Subtle cognitive deficits in exposed child Maeda, A, Bateman BT, Clancy CR, Creanga AA, Leffert LR (2014). Opioid abuse and dependence during pregnancy: temporal trends and obstetrical outcomes. Perioperative Medicine, 121: 1158 -1165. Yeoh SL, Eastwood J, Wright IM, et al. Cognitive and Motor Outcomes of Children With Prenatal Opioid Exposure: A Systematic Review and Meta-analysis. JAMA Netw Open. 2019; 2(7): e 197025.
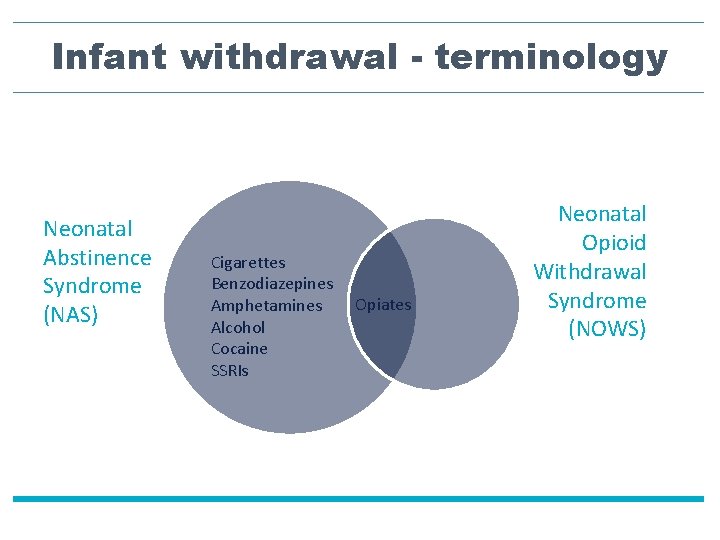
Infant withdrawal - terminology Neonatal Abstinence Syndrome (NAS) Cigarettes Benzodiazepines Amphetamines Alcohol Cocaine SSRIs Opiates Neonatal Opioid Withdrawal Syndrome (NOWS)

Neonatal Opioid Withdrawal Syndrome (NOWS) • Illicit opioids – Heroin • Prescription opioids – Oxycodone, fentanyl, hydromorphone • Exposure to medications used to treat maternal opioid use disorder – Methadone – Buprenorphine

Effects of withdrawal on infant • • • Sleep and wake cycle disturbances Altered tone or movement Autonomic dysfunction Easy overstimulation, sensitivity, hyperarousal Feeding difficulties

Maternal Opioid Use – Major concerns • Peaks/troughs and intermittent withdrawal – Fetus experiences the stress of cyclical withdrawal states, which compromises growth, may have long -term epigenetic consequences – Uterus may become hyperactive miscarriage, preterm labor/delivery • Cravings may make it difficult to avoid illicit use – Risk of overdose, of infection, unsafe behaviors, etc.

First-line treatment • MAT with an opioid agonist (methadone or buprenorphine) is considered first-line treatment for pregnant women with opioid use disorders – Suppress withdrawal symptoms and craving – Block the acute effects of other opioids

MAT and Pregnancy – Historical Context • Research in late 1970’s suggested relationship between maternal methadone dose and severity of neonatal withdrawal • Decades of research since: no compelling evidence to reduce maternal dose to avoid NOWS • Only within the last couple of decades that the norm has become medicating pregnant women according to same principles as non-pregnant women

Exposure to medication Exposure to untreated mental illness/ substance use disorder

Pregnancy pharmacokinetics • Pregnant women on methadone maintenance require dosage increases, particularly in third trimester – Increases in progesterone and estrogen concentrations – Alterations in plasma volume – Hepatic induction of CYP 3 A 4 – Often need for dosage reduction postpartum, but recommendation is to titrate dose to signs and symptoms of sedation • Pregnant women on buprenorphine may require modest dose increase

MOTHER Study • Multicenter, double-blind RCT • N=175 pregnant women – Buprenorphine (N=86, 58 completed study) – Methadone (N=89, 73 completed study) • Compared to neonates of mothers treated with methadone, neonates of mothers treated with buprenorphine in pregnancy: – Required less morphine – Had shorter hospital stays – Had shorter duration of treatment for NAS Jones HE, et al. "Neonatal abstinence syndrome after methadone or buprenorphine exposure". New England Journal of Medicine. 2010. 363(24): 2320 -2331.

MAT - Benefits Maternal • 70% reduction in overdose related deaths • Decreased risks of HIV, HBV, HCV • Increased engagement in prenatal care and SUD treatment Fetal • Reduced fluctuations in opioid levels, reducing fetal stress • Reduced exposure to adulterated heroin • Decrease in intrauterine fetal demise • Decrease in IUGR • Decrease in preterm delivery MCPAP for Moms, mcpapformoms. org

NOWS is expected and treatable • Babies of mothers on MAT were already at-risk for NOWS • If NOWS does occur, it is far safer to treat it in a fully grown, term baby with an appropriate pharmacologic agent – Versus a small, incompletely developed fetus withdrawing under blind conditions in utero • Non-pharmacologic treatments (breastfeeding, low stim environment, swaddling, sucking)

Alcohol Use • Alcohol is a known teratogen • Also causes placental dysfunction • No safe amount, no safe time, no safe type of alcohol during pregnancy • Women who are pregnant or who may become pregnant should abstain from drinking alcohol; never too late to stop drinking • Fetal effects: Spontaneous abortion, preterm labor, stillbirth, IUGR • Neonatal effects: FASD, SIDS • Can increase other health risks: – – – Injuries Violence Heart disease, cancer Unintended pregnancy Sexual transmitted infections

Alcohol use in pregnancy • 10. 2% any alcohol use in past 30 days • 3. 1% four or more drinks on one occasion • Highest prevalence: – Unmarried college graduates (13. 0%) – Women age 35 -44 (18. 6%) • Cross demographic and socioeconomic boundaries CDC Behavioral Risk Factor Surveillance System (BRFSS), United states 2011 -2013

FASDs • Fetal alcohol syndrome (FAS) • Partial fetal alcohol syndrome (p. FAS) • Alcohol-related neurodevelopmental disorder (ARND) • Alcohol-related birth defects (ARBD) • Neurobehavioral disorder associated with prenatal alcohol exposure (ND-PAE)

Cocaine Use Disorder • Unlikely to be a structural teratogen • Adverse effects are due to vasoconstriction: – Placental insufficiency, placental abruption, spontaneous abortion • Increases risk for LBW, SGA, and preterm birth • Evidence of subtle later cognitive deficits Ackerman JP, Riggins T, Black MM. A review of the effects of prenatal cocaine exposure among school-aged children. Pediatrics. 2010; 125(3): 554 -565. Buckingham-Howes S, Berger SS, Scaletti LA, Black MM. Systematic review of prenatal cocaine exposure and adolescent development. Pediatrics. 2013; 131(6): e 1917 -e 1936.

Marijuana • Most commonly used illicit substance in pregnancy • THC crosses the blood-brain barrier and placenta – THC deposited in fetal fat (even if used infrequently, storage in fat persistent exposure) • Difficult to assess effects due to confounders – – – More likely to use alcohol and tobacco THC decreases fetal folic acid uptake Intrapartum stillbirth (may be modest increase) Low birth weight? Neural development? • No evidence that marijuana is more effective for nausea than FDA-approved medications Marijuana use during pregnancy and lactation. Committee Opinion No. 637. American College of Obstetricians and Gynecologists. Obstet Gynecol 2015; 126: 234– 8.

ACOG Committee Opinion “Because of concerns regarding impaired neurodevelopment, as well as maternal and fetal exposure to the adverse effects of smoking, women who are pregnant or contemplating pregnancy should be encouraged to discontinue marijuana use. Obstetrician–gynecologists should be discouraged from prescribing or suggesting the use of marijuana for medicinal purposes during preconception, pregnancy, and lactation. Pregnant women or women contemplating pregnancy should be encouraged to discontinue use of marijuana for medicinal purposes in favor of an alternative therapy for which there are better pregnancy-specific safety data. There are insufficient data to evaluate the effects of marijuana use on infants during lactation and breastfeeding, and in the absence of such data, marijuana use is discouraged. ” —ACOG Committee Opinion Number 722, October 2017 (Reaffirmed 2019)
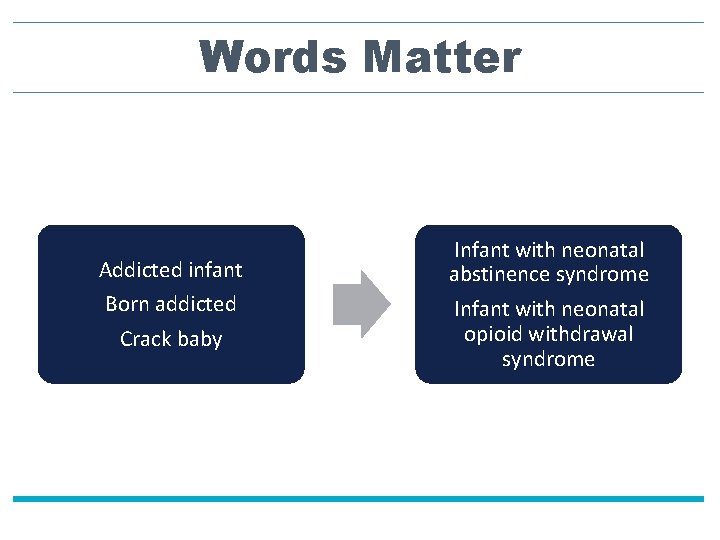
Words Matter Addicted infant Born addicted Crack baby Infant with neonatal abstinence syndrome Infant with neonatal opioid withdrawal syndrome

Thank you! Louisiana Mental Health Perinatal Partnership (LAMHPP) www. lamhpp. org perinatalpsych@tulane. edu 504 -988 -9171
- Slides: 41