Subsolidus evolution Mineral transformations Secondary minerals Fluids expulsion

Sub-solidus evolution

• Mineral transformations • Secondary minerals • Fluids expulsion and movement – Pegmatite/aplite veins – Mineralized veins • Hydrothermal alteration – Episyenites, endoskarns, greisens – Exoskarns

Mineral transformations • Polymorphs • Exsolutions (solvus)

Phase diagram for Si. O 2
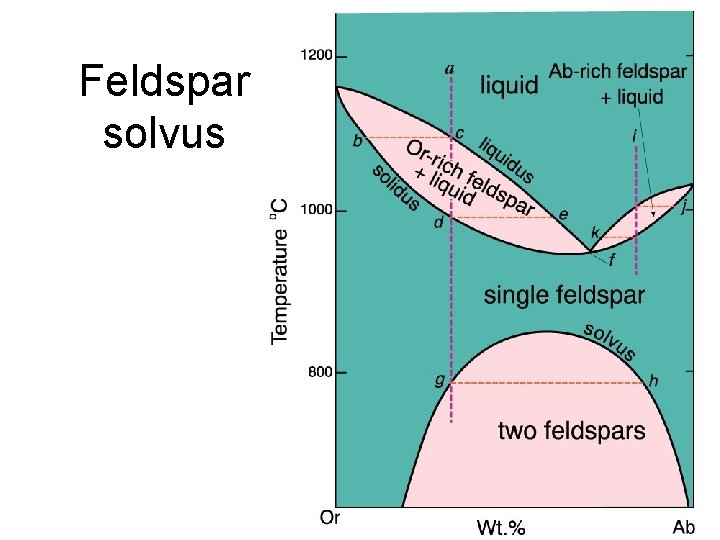
Feldspar solvus

Perthites

Opx-Cpx exsolution

Secondary minerals • « Autometamorphism »
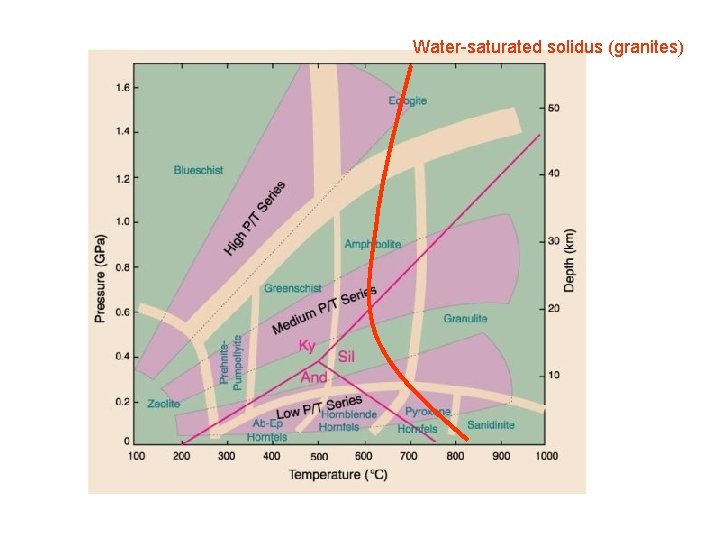
Water-saturated solidus (granites)

Secondary minerals • Px => Amp => Bt • Px, Amp, Bt => chlorite (phyllosilicate) • K-feldspar, feldspathoids => sericite (fine white mica) • Ca-plagioclase => saussurite (epidote) • Olivine => serpentine (complex phyllosilicate), iddingsite (a mixture of various Fe-Mg silicates)

Figure 3 -20. a. Pyroxene largely replaced by hornblende. Some pyroxene remains as light areas (Pyx) in the hornblende core. Width 1 mm. b. Chlorite (green) replaces biotite (dark brown) at the rim and along cleavages. Tonalite. San Diego, CA. Width 0. 3 mm. © John Winter and Prentice Hall. Pyx Hbl Chl Bt

Sericitization K-feldspar to sericite: 3 KAl. Si 3 O 8 + 2 H+ > KAl 3 Si 3 O 10(OH)2 + 6 Si. O 2 + 2 K+
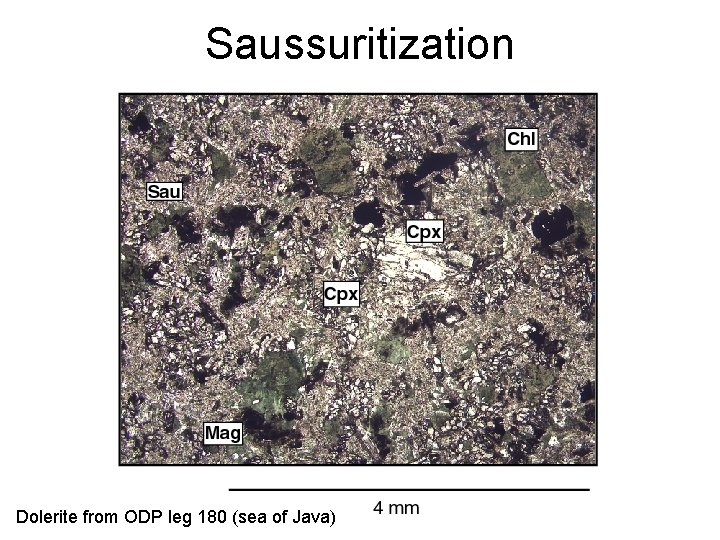
Saussuritization Dolerite from ODP leg 180 (sea of Java)
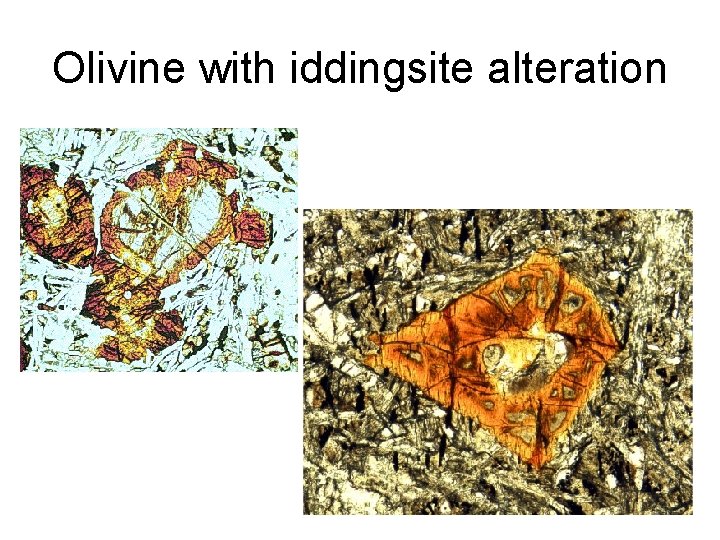
Olivine with iddingsite alteration

Calcite vein

Fluid expulsion • Typical water contents: 2 -4% in a granite • Water content of a biotite: ~2 % • Biotite: max. 5 -10 % of the rock Excess water = ? + meteoric water also feeding the hydrothermal system
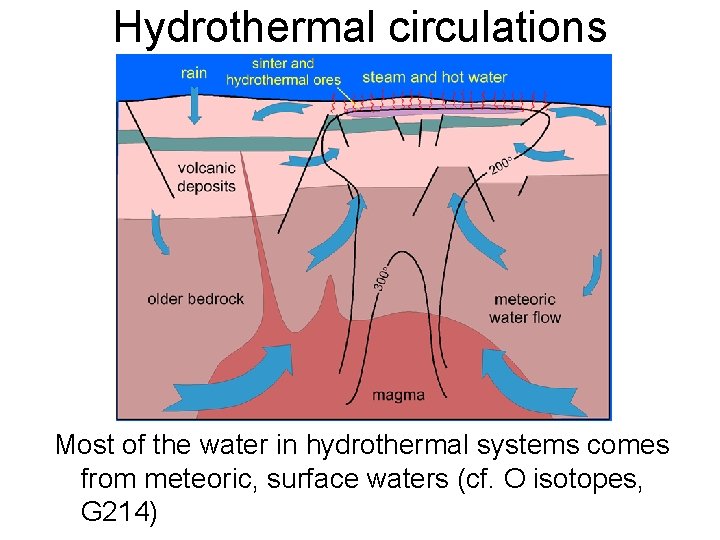
Hydrothermal circulations Most of the water in hydrothermal systems comes from meteoric, surface waters (cf. O isotopes, G 214)

Effect of free, hot water • Overpressure, fractures, etc. • Very aggressive solvent! • Aplite/pegmatite veins

Pegmatites recording the same strain pattern as ductile structures Cape de Creus, Spain
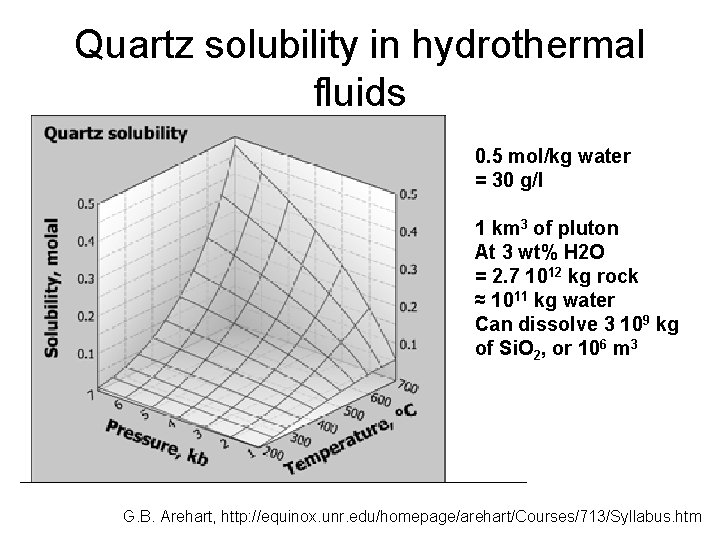
Quartz solubility in hydrothermal fluids 0. 5 mol/kg water = 30 g/l 1 km 3 of pluton At 3 wt% H 2 O = 2. 7 1012 kg rock ≈ 1011 kg water Can dissolve 3 109 kg of Si. O 2, or 106 m 3 G. B. Arehart, http: //equinox. unr. edu/homepage/arehart/Courses/713/Syllabus. htm

Evidence for Si-rich hydrothermal fluids Tatio hydrothermal field, Peru

Network of pegmatites/apl ite dykes

Mineralized veins • Very incompatible elements (large ions, typically) concentrated in last liquids, then in fluids • The same elements are leached from an already cooled rock (igneous intrusion or its wall-rock) • Precipitate with hydrothermal veins
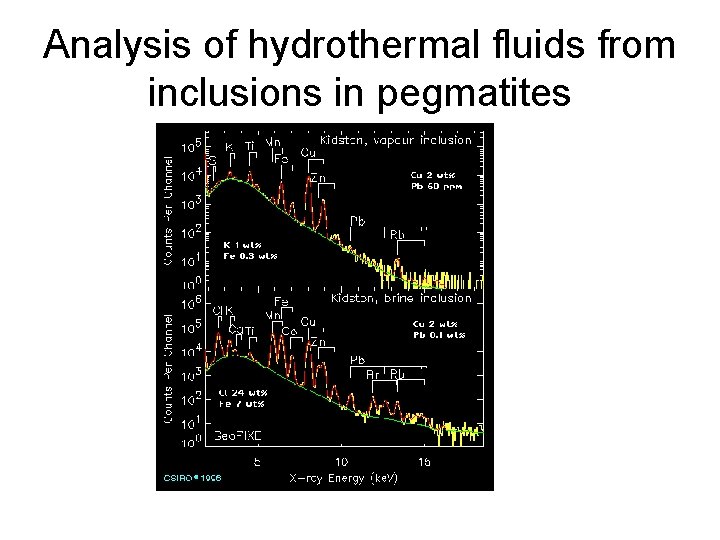
Analysis of hydrothermal fluids from inclusions in pegmatites
Gold-quartz veins • See economic geology (GEOL 344)
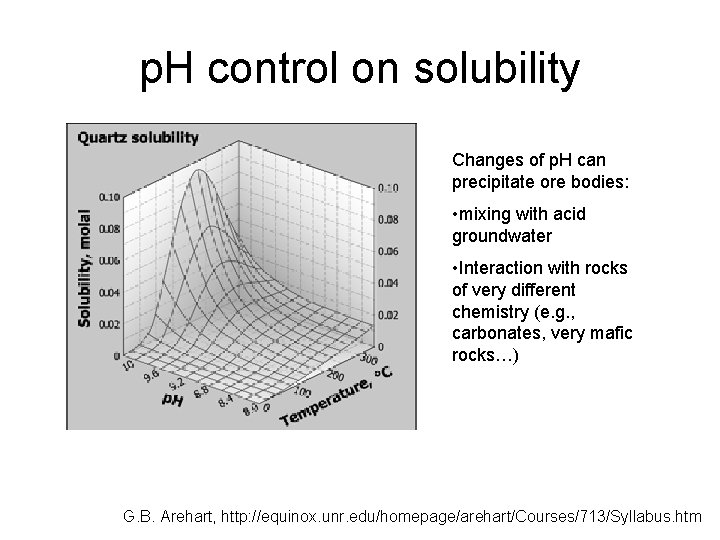
p. H control on solubility Changes of p. H can precipitate ore bodies: • mixing with acid groundwater • Interaction with rocks of very different chemistry (e. g. , carbonates, very mafic rocks…) G. B. Arehart, http: //equinox. unr. edu/homepage/arehart/Courses/713/Syllabus. htm
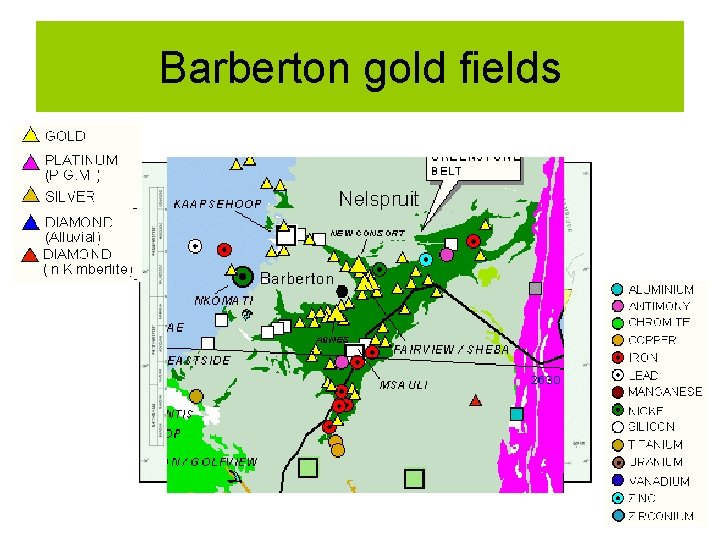
Barberton gold fields

Hydrothermal modifications of rocks • Around the intrusion – Exoskarns, etc. • In the intrusive rocks – Episyenites – Endoskarns, greisens

Around the pluton
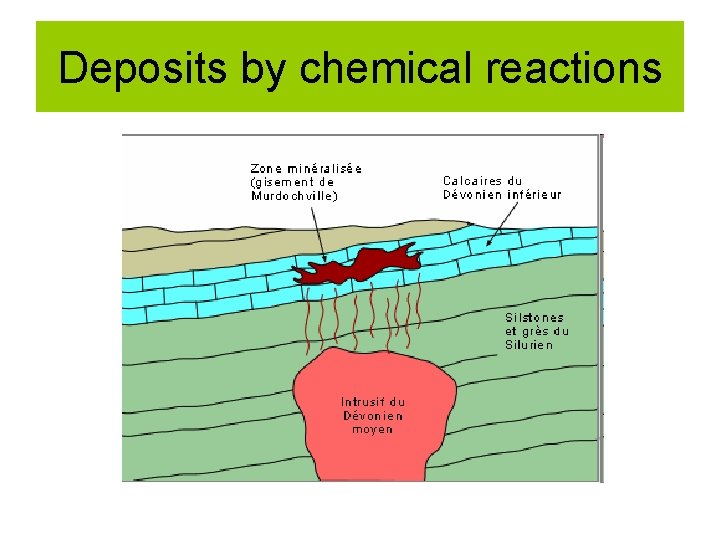
Deposits by chemical reactions
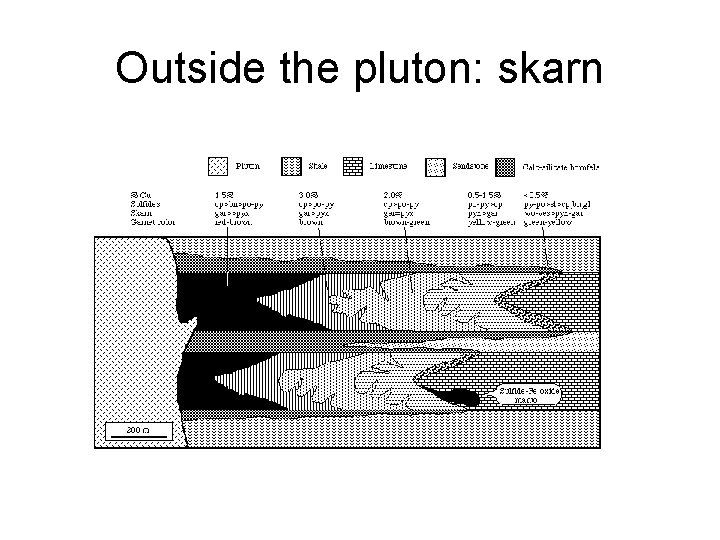
Outside the pluton: skarn



In the pluton
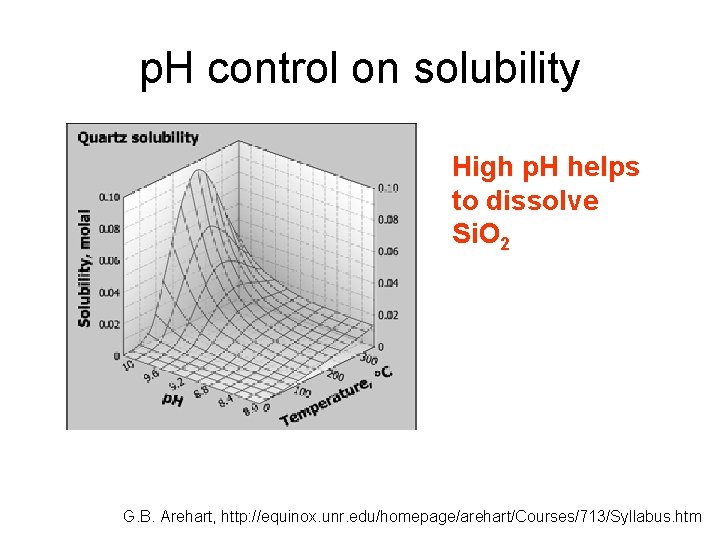
p. H control on solubility High p. H helps to dissolve Si. O 2 G. B. Arehart, http: //equinox. unr. edu/homepage/arehart/Courses/713/Syllabus. htm
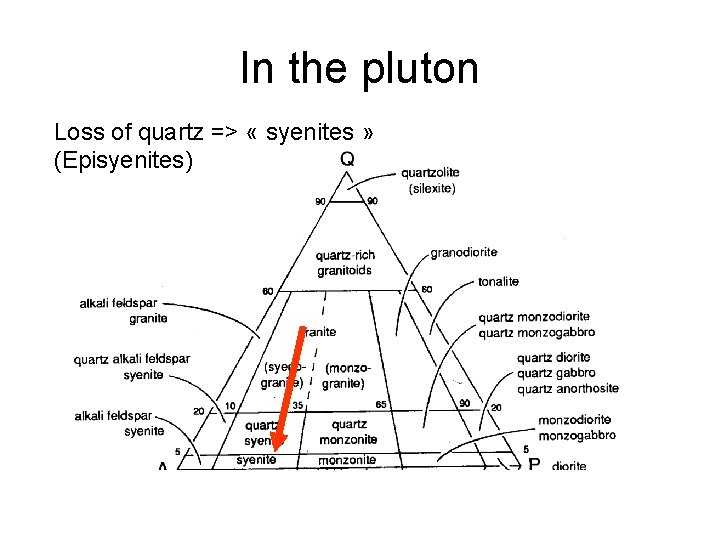
In the pluton Loss of quartz => « syenites » (Episyenites)

Fedlspar alteration in the pluton • K-feldspar to sericite: 3 KAl. Si 3 O 8 + 2 H+ > KAl 3 Si 3 O 10(OH)2 + 6 Si. O 2 + 2 K+ • Sericite to kaolin: 2 KAl 3 Si 3 O 10(OH)2 + 2 H+ + 3 H 20 > 3 Al 2 Si 2 O 5(OH)4 + 2 K+ Requires acidic fluids!

In the pluton • Episyenites are plutonic rocks from which the quartz has been dissolved away (therefore, they become syenites) (high p. H) • Greisens are plutonic rocks where the feldspar has been transformed into clays (kaolinite) by hydrothermal reactions (low p. H)
- Slides: 38