Submitting to the Human Research Review Committee HRRC

Submitting to the Human Research Review Committee (HRRC) Sarah Targownik, LMSW, CIP IRB “On-the-Go” Specialist Outreach, Education & Support to the Academic Research Community Human Research Protections Office University of New Mexico Health Sciences Center stargownik@salud. unm. edu (505) 272 -0949 (Direct Line) (505) 272 -1129 (Main Office) (505) 272 -0803 (Fax) Fitz Hall B-71

TOPICS TO BE COVERED: ■ THE HRRP, HRRC AND THE HRPO – Definitions & responsibilities ■ RISK AND REVIEW LEVEL – As one increases so does the other ■ TEMPLATES AND FORMS: Choosing wisely to save time ■ DOCUMENTS ISSUES: Spelling, formatting, titles, versions ■ ANCILLARY REVIEW ISSUES: DUA, MTA, HUS, SRC ■ STUDY TEAM TRAINING ISSUES: Big source of delays
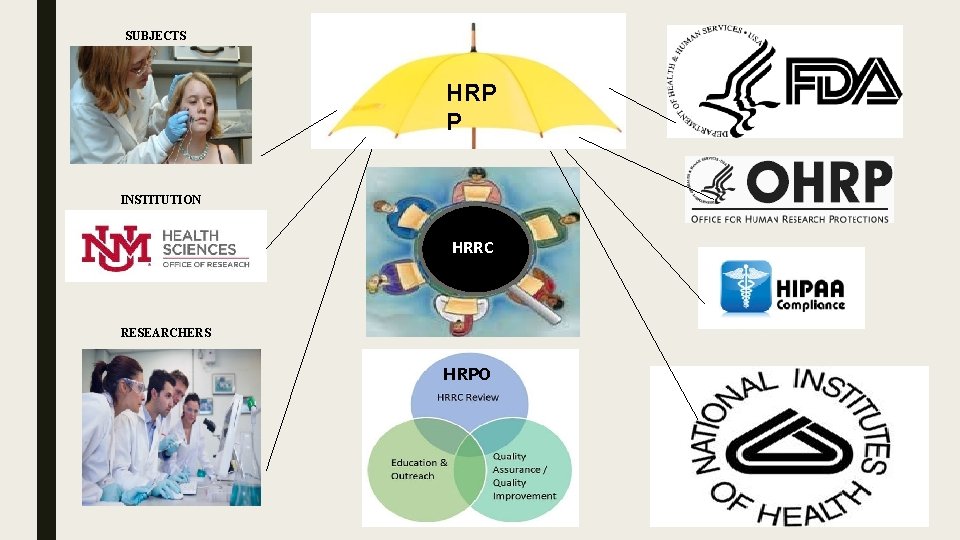
SUBJECTS HRP P INSTITUTION HRRC RESEARCHERS HRPO

The Human Research Review Committee = HRRC ■ 3 HRRC Committees ■ They meet the first 3 Tuesdays of each month ■ 10 -12 members per committee ■ Composition of committees ■ COMPLETE Submissions are due at least 10 days before committee meeting ■ Assignment of a study to a committee HRRC

Human Research Protections Office = HRPO • Provides Administrative support and regulatory reference for the HRRC chair and members • Provides education, outreach and support to investigators study teams, HSC Departments and UNM HSC as a whole • Quality Assurance and Quality Improvement completes routine and “for cause” audits and other related evaluations.

Human Subject Research ■ Research: “A systematic investigation designed to develop or contribute to generalizable knowledge” [45 CFR 46. 102(d)] ■ Human Subjects (DHHS): “A living individual about whom an investigator conducting research obtains (1) data through intervention or interaction with the individual, or (2) identifiable private information” [45 CFR 46. 102(f)].

RISK AND REVIEW

Its All About… Risk: the chance that a subject will experience harm. Minimal risk: the harm or discomfort anticipated in the research are not greater in and of themselves than those ordinarily encountered in daily life.
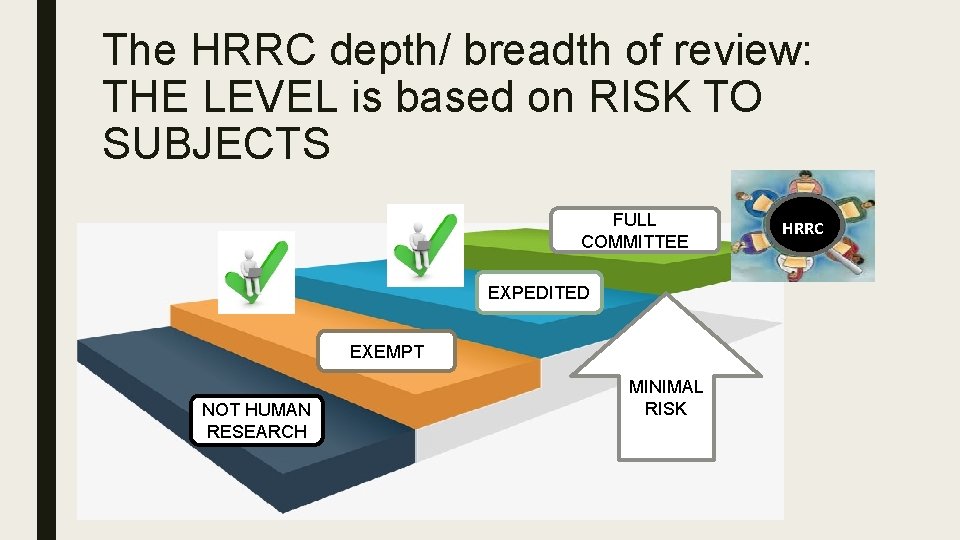
The HRRC depth/ breadth of review: THE LEVEL is based on RISK TO SUBJECTS FULL COMMITTEE EXPEDITED EXEMPT NOT HUMAN RESEARCH MINIMAL RISK HRRC

Possible examples of Not Human Research at UNM HSC? Case study Data or specimens from cadavers Public Health program evaluation GET Quality assurance/ Improvement project within a department A HRPO CONSULT A SUBMISSION TO REQUEST A NHSR DETERMINATION IS ALWAYS RECOMMENDED

The “Quality Improvement” Project

Not Human Subject Research: Request For Determination Submission HRP-585 NHSR TEMPLATE ■ Why Submit a NHSR request? “In Case…” ■ How is a NHSR request submitted? – New Study Submission made through Click IRB – NHSR is the protocol document and is the only document needed – Do not add other study team members

Exemptions under Title 45 CFR § 46 ■ EXEMPT (4) Secondary research for which consent is not required. Information is recorded by the investigator in such a manner that the identity of the human subjects cannot readily be ascertained directly or through identifiers linked to the subjects, the investigator does not contact the subjects, and the investigator will not re-identify subjects; ■ EXEMPT (2) Research that ONLY includes interactions involving educational tests (cognitive, diagnostic, aptitude, achievement), survey procedures, interview procedures AND The information obtained is recorded by the investigator in such a manner that the identity of the human subjects cannot readily be ascertained ■ EXEMPT (1) Research conducted in established or commonly accepted educational settings, involving normal educational practices, such as (i) research on regular and special education instructional strategies, or (ii) research on the effectiveness of or the comparison among instructional techniques, curricula, or classroom management methods.

Elements that increase risk to subjects Collection/ retention of PHI Experimental elements, drugs & devices Vulnerable Populations Sicker patients Invasive procedures Randomization schemes Biologics First in Humans Sensitive Topics

Choose Wisely: forms & templates Documents should fit your type of study Kind of Research + Review Level = Template & Form choice Current versions - from the Click IRB Library BE

Required Documents REQUIRED FOR ALL NEW STUDY SUBMISSIONS: ☐ New Study Submission (Click Smart. Form) ☐ Protocol ☐ Departmental Scientific Review – requires signature (not required for exempt) ☐ Curriculum Vitae (CV) for PI IF APPLICABLE: ☐ HIPAA Consent Form, Assent Form ☐ Participantand Recruitment Materials ☐ Surveys, Questionnaires, Interview Scripts ☐ Data Collection Tools ☐ Letters of support from an unaffiliated research location ☐ Agreements/ approvals from other IRBs ☐ Ancillary review forms and executed agreements

Protocol Templates CLICK > IRB > LIBRARY TEMPLATES • • • HRP – 503 ORIGINAL TEMPLATE: COMPLEX STUDIES, > MINIMAL RISK HRP – 509 LOCAL ADDENDUM: LOCAL PROCESS FOR SPONSORED PROTOCOLS HRP – 580 CHART REVIEW: RETROSPECTIVE AND PROSPECTIVE HRP – 581 SOCIAL-BEHAVIORAL: FOCUS GROUPS, SURVEYS HRP – 582, 583, 584 RESEARCH EXEMPT REQUESTS HRP-585 TEMPLATE-Not Human Subject Research

Consent Templates TEMPLATES ***FOR NEW STUDIES, ONLY CONSENT FORMS WITH ‘KEY INFORMATION’ ARE ACCEPTED HRP 507 T STANDARD CONSENT WITH HIPAA HRP 507 A SURVEY CONSENT (WAIVER OF CONSENT DOCUMENTATION) HRP 507 B CHILD AGES 7 -11 ASSENT HRP 507 R UNMHSC HIPAA AUTHORIZATION

Document Issues Word document Spelling & formatting Remove instructions Version dates x 2 Remove comments & TCs

Document Requirements New document - protocol, consent, questionnaire, poster, etc. Word Doc. CLEAN (delete comments, track changes, etc) Remove instructions Version dates x 2 within the document (header, footer) and in the e. File title “Protocol ver. 7 -8 -19” Spelling and formatting matter Revised document Word Doc. with THE NEW revisions in track changes only. If this document was revised before, accept the old previously approved track changes before you start to add the new ones Remember to update the version dates (both places) Use the “Update” button instead of “Add” to upload the document into Click IRB

Ancillary Reviews Be Pro-active Submit Forms early required Sub-committee Contracts reviews executed

Required Ancillary Review Processes & Documentation ☐ Radiation Safety Attachment HRP-223 The PI must describe the use of ionizing radiation and assign it “standard of care” or for research use assign “patient dose” for each procedure. Research Use must be reviewed by the “Human Use Subcommittee (HUS)” ☐ Biological Specimens Attachment HRP-224 If obtaining specimens from UNM Pathology, approval from Nancy Joste, Division Chief of Surgical Pathology is required. Please have Dr. Joste sign bottom section of the HRP-224 ☐ Banked Specimens already stored in the HTR and/or UNM Pathology, submit protocol to the Tissue Research Oversight Committee (TROC). Contact Karen Capobianco for guidance in this process. Once the SRC/TROC approves proposal, upload a copy of the approval letter into Click ‘Other attachments’ section of the Local Site Documents smart page. ☐ Materials Transfer Agreement are short contracts governing the transfer of tangible research property for research use. Contact the Sponsored Projects Office for information

Data Use Agreements (DUA) may be required when there will be an exchange of data with recipients outside the university. Data may be solely incoming, solely outgoing, or both (incoming and outgoing) – Verification that the technical method of transfer uses a known secure encryption protocol – Record that the transfer was completed – Record of who sent the data and who received the data – Record of where the data is stored and how it is secured – Record of who has been granted access to the data and the authorizing individual. – Record of who owns the data and what happens to it when the study is closed

Retention of Specimens ■ Use and storage during the Life of an approved study vs. intent to store for future use. ■ Clear language in the protocol to describe exactly what will happen to samples at the time of study closure: Central HTR, Destruction, Transfer (materials transfer agreement), Satellite HTR ■ Independent submission to establish a SATELLITE HTR (s. HTR): – The first step is to submit to the HTOC. – HTOC approval > HRRC submission

TRAINING & Study Team Issues ■ PI Eligibility ■ All courses ■ Correct & current courses BE PREPARED !! BEbe

UNM HSC Principal Investigator Eligibility Must be paid UNM HSC Faculty Member . 50 FTE or Greater Tenure Track or Non-Tenure Track Researcher, Clinical Educator, Lecturer, Probationary, Visiting- Flex Track, Visiting Research Adjunct Faculty, Retired Faculty, Volunteer Faculty Postdoctoral Students Fellows, Visiting, Residents,

Required Training: CITI Human Subjects Protection Course (Current for 3 years) FCOI course “Financial Conflicts of Interest” (Current for 4 years) Click COI/ ERA “Electronic Research Administration COI Disclosures (Completed Once to obtain an account) Click IRB Training (Completed Once to obtain an account). It is only required for the PI; however, any study team member wanting/needing to get into the Click IRB study record must get an account

HRPO Website: Training links and lookups HRPO page > Investigators > Getting Started > Training (https: //hsc. unm. edu/research/hrpo/) CITI completion CLICK Look-up List COI FCOI completion FCOI Look-up List

CITI: THE REQUIRED COURSES Create an account * UNM HSC affiliated Step 1: Select Your Organization Affiliation: University of New Mexico Health Sciences Center Step 7: Select Curriculum: – Question 1 Human Subjects Research – Select ONLY either Group 1 Biomedical OR Group 2 Social/Behavioral *

Conflict of Interest Process: UNM/ UNM HSC affiliates 1 The required COI courses in Learning Central: • Financial Conflicts of Interest (FCOI) Course HSC 104 -002. • Click ERA (COI) Course – HSC 001 AFTER BEING ADDED TO A NEW STUDY: 2 All study team members not listed on the grant in the ERA system will receive emails asking them to complete their COI process in the Click COI/ERA system. These emails come from "HSC-COI@salud. unm. edu" and the subject line is "HSC-Initiated Certification" ***** STAFF MUST LOOK FOR THESE EMAILS AND RESPOND AS QUICKLY AS POSSIBLE. IF ANY REMAIN INCOMPLETE AFTER 5 DAYS, YOUR NEW STUDY SUBMISSION WILL BE WITHDRAWN.

A Couple of Click IRB Tips ■ PI PROXY & Primary Contact ■ Failure to respond = withdrawal

CLICK IRB Main Study Page: Assigning Roles ■ PI Proxy – must be assigned by the PI – The PI Proxy can perform any function in Click that the PI can ■ Primary Contact will receive all Click IRB notifications ■ Guests can view the study, but can make no changes
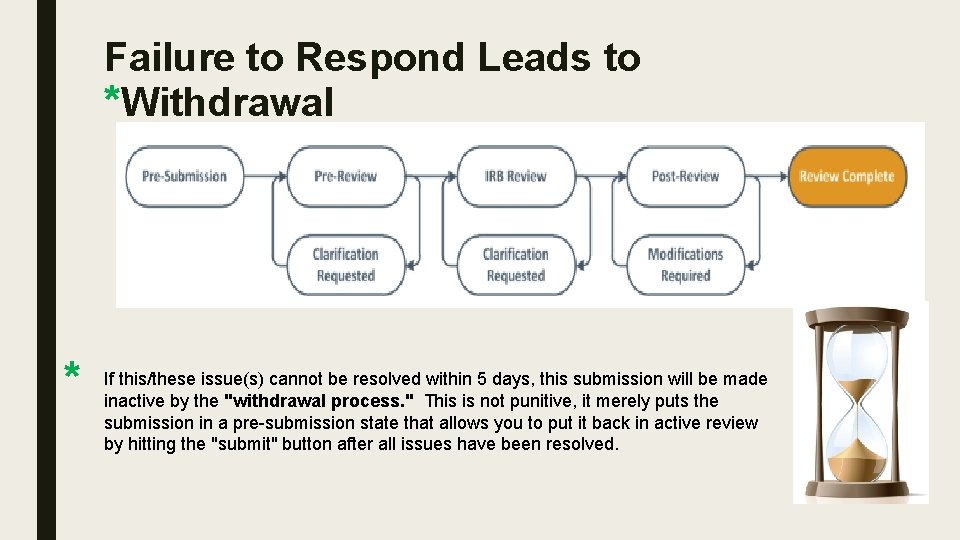
Failure to Respond Leads to *Withdrawal HRPO POLICY * If this/these issue(s) cannot be resolved within 5 days, this submission will be made inactive by the "withdrawal process. " This is not punitive, it merely puts the submission in a pre-submission state that allows you to put it back in active review by hitting the "submit" button after all issues have been resolved.

Contact the HRPO for a Consult HRPO CONSULTATIONS IRB ON THE GO • Individualized IRB & Click training is available with an appointment ( for one person or a whole department) • Bring your documents • Bring your questions Sarah Targownik, LMSW Human Protections Specialist / IRB-ON-THE-GO stargownik@salud. unm. edu (505) 272 -0949 (Direct Line) (505) 272 -1129 (Main Office) Fitz Hall B-71
- Slides: 35