SUBLINGUAL IMMUNOTHERAPY Giovanni Passalacqua Allergy Respiratory Diseases Dept

SUBLINGUAL IMMUNOTHERAPY Giovanni Passalacqua Allergy & Respiratory Diseases Dept. Internal Medicine. University of Genoa ITALY

THE LITERATURE 66 RDBPC TRIALS 8 RANDOMIZED OPEN TRIALS 6 COMPARATIVE (SLIT vs SCIT) 6 TRIALS IN OTHER DISEASES
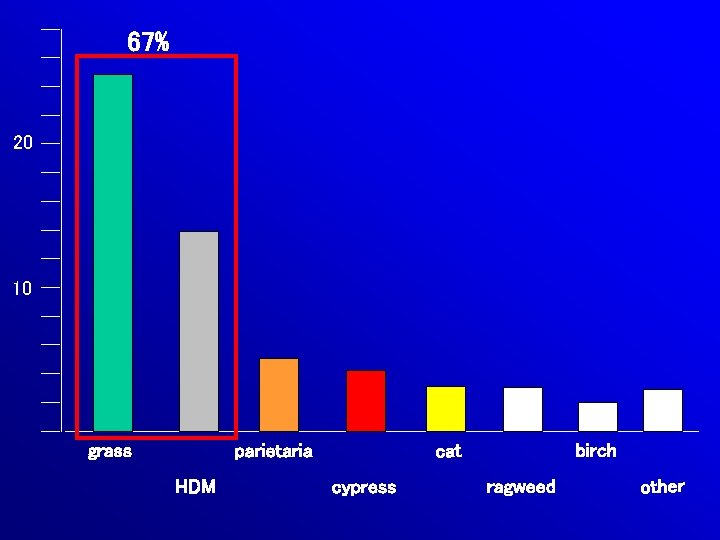
67% 20 10 grass HDM birch cat parietaria cypress ragweed other

TREATMENT DURATION Less or equal 6 months: 23 studies Less or equal 12 months: 15 studies More than 12 months: 20 studies PATIENTS ENROLLED < 20 : 4 studies < 150 : 11 studies < 50 : 21 studies > 150 : 8 studies < 100 : 16 studies
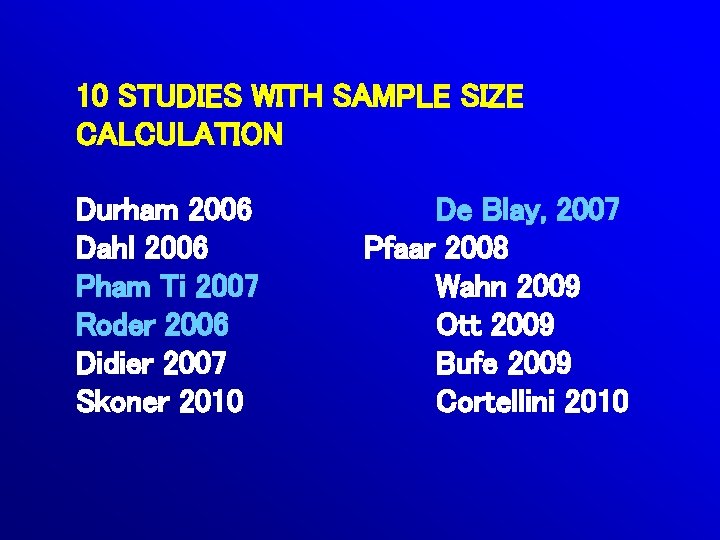
10 STUDIES WITH SAMPLE SIZE CALCULATION Durham 2006 Dahl 2006 Pham Ti 2007 Roder 2006 Didier 2007 Skoner 2010 De Blay, 2007 Pfaar 2008 Wahn 2009 Ott 2009 Bufe 2009 Cortellini 2010
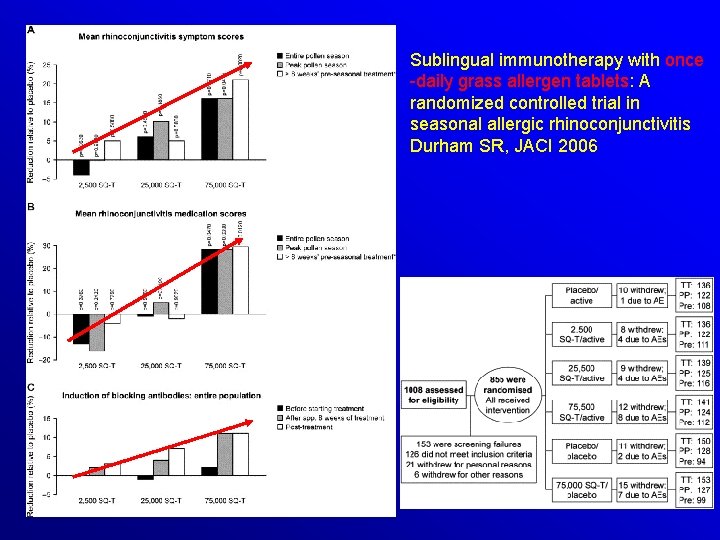
Sublingual immunotherapy with once -daily grass allergen tablets: A randomized controlled trial in seasonal allergic rhinoconjunctivitis Durham SR, JACI 2006
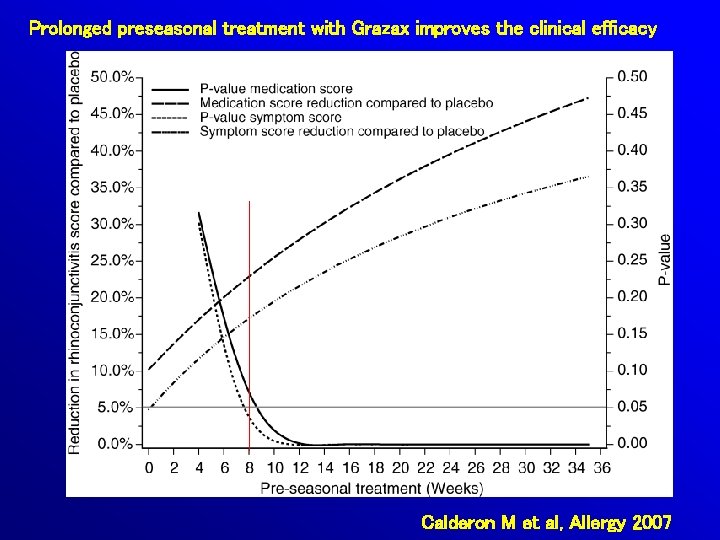
Prolonged preseasonal treatment with Grazax improves the clinical efficacy Calderon M et al, Allergy 2007
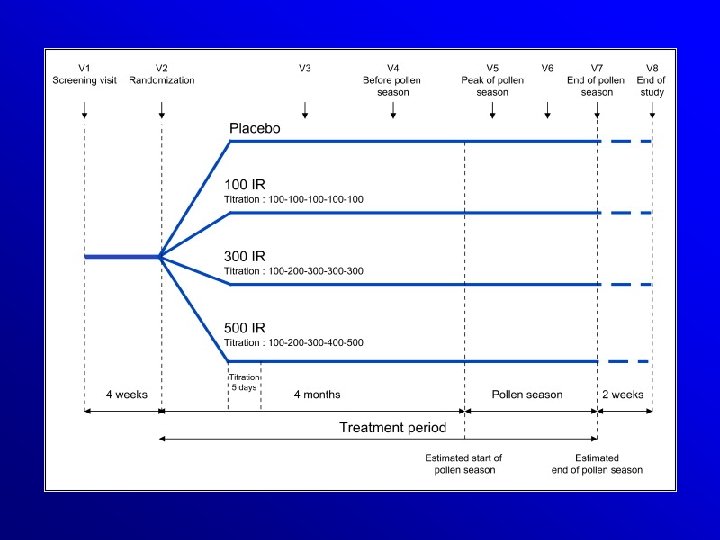
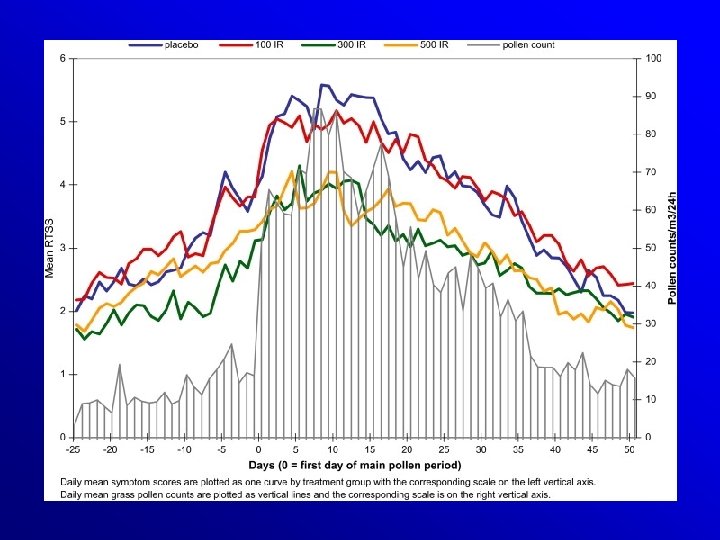
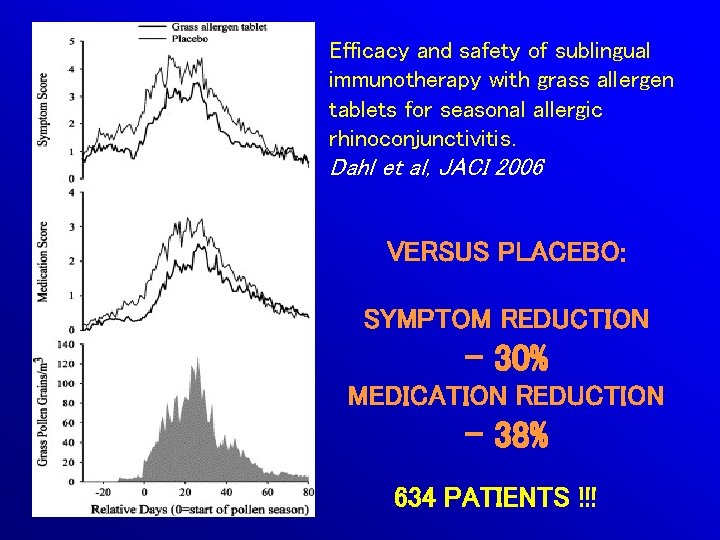
Efficacy and safety of sublingual immunotherapy with grass allergen tablets for seasonal allergic rhinoconjunctivitis. Dahl et al, JACI 2006 VERSUS PLACEBO: SYMPTOM REDUCTION - 30% MEDICATION REDUCTION - 38% 634 PATIENTS !!!
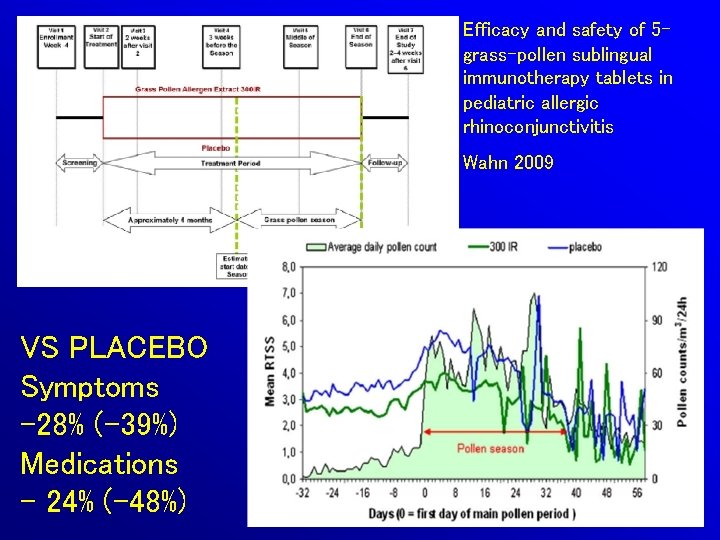
Efficacy and safety of 5 grass-pollen sublingual immunotherapy tablets in pediatric allergic rhinoconjunctivitis Wahn 2009 VS PLACEBO Symptoms -28% (-39%) Medications - 24% (-48%)

VS PLACEBO Symptoms -34% to – 44%
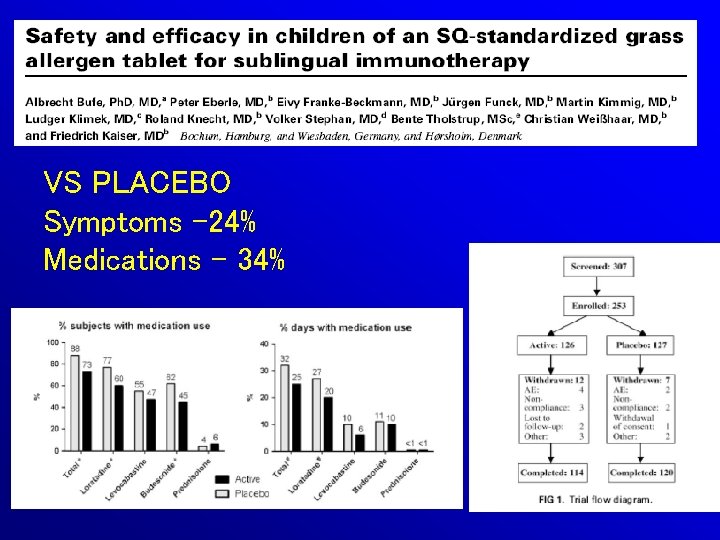
VS PLACEBO Symptoms -24% Medications - 34%

21 trials Sublingual immunotherapy for allergic rhinitis. Allergy, 2005. Wilson D. , Torres Lima I, Durham S.
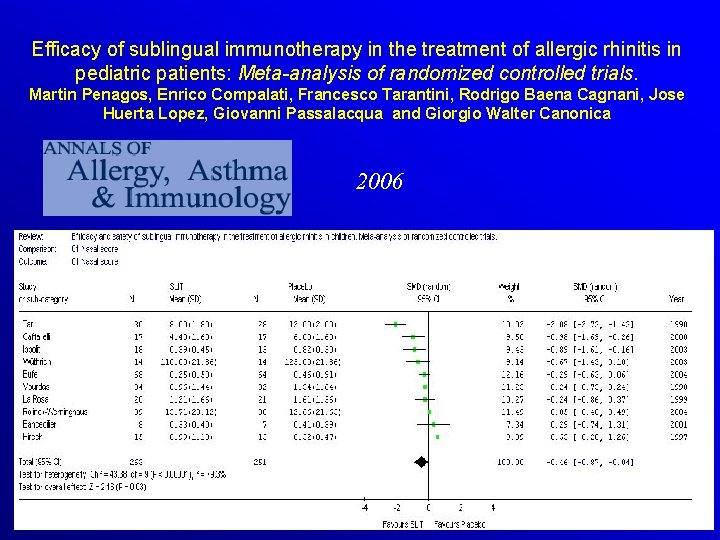
Efficacy of sublingual immunotherapy in the treatment of allergic rhinitis in pediatric patients: Meta-analysis of randomized controlled trials. Martin Penagos, Enrico Compalati, Francesco Tarantini, Rodrigo Baena Cagnani, Jose Huerta Lopez, Giovanni Passalacqua and Giorgio Walter Canonica 2006
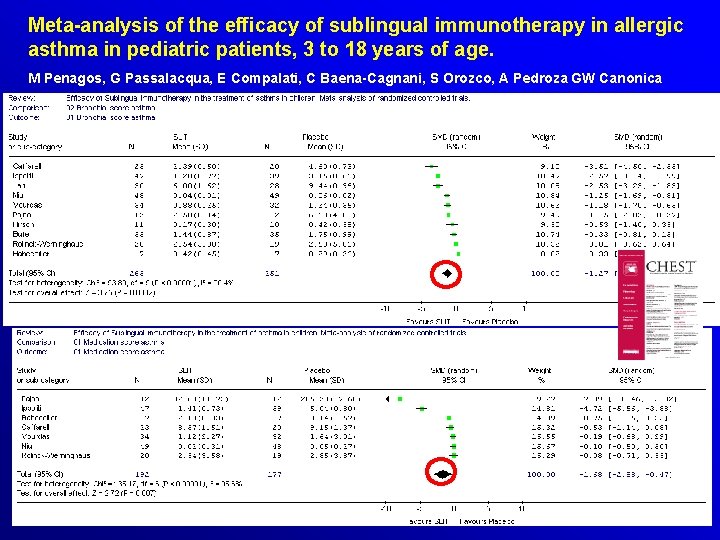
Meta-analysis of the efficacy of sublingual immunotherapy in allergic asthma in pediatric patients, 3 to 18 years of age. M Penagos, G Passalacqua, E Compalati, C Baena-Cagnani, S Orozco, A Pedroza GW Canonica

ARIA Update on immunotherapy SR Durham and G. Passalacqua JACI 2007 in press SCIT SLIT Clinical efficacy: Rhinitis Ia Ia Clinical Efficacy: Asthma Ia Ia Clinical efficacy: Children (rhinitis) Children (asthma) Ib Ib Ia Ia Prevention of new sensitizations Ib IIa Longterm effect Ib IIa Prevention of asthma IIb
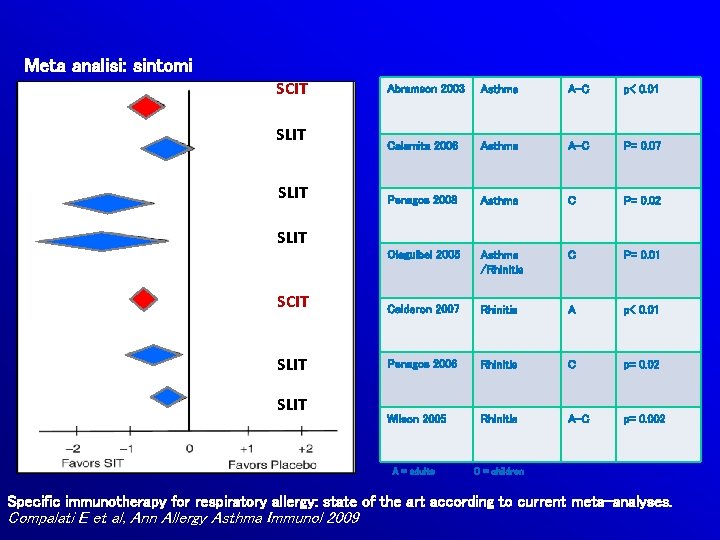
Meta analisi: sintomi SCIT Abramson 2003 Asthma A-C p< 0. 01 Calamita 2006 Asthma A-C P= 0. 07 Penagos 2008 Asthma C P= 0. 02 Olaguibel 2005 Asthma /Rhinitis C P= 0. 01 SCIT Calderon 2007 Rhinitis A p< 0. 01 SLIT Penagos 2006 Rhinitis C p= 0. 02 Wilson 2005 Rhinitis A-C p= 0. 002 SLIT A = adults C = children Specific immunotherapy for respiratory allergy: state of the art according to current meta-analyses. Compalati E et al, Ann Allergy Asthma Immunol 2009

49 (OUT OF 60) RDBPC TRIALS SUITABLE FOR POOLING: 2333 SLIT 2256 PLACEBO
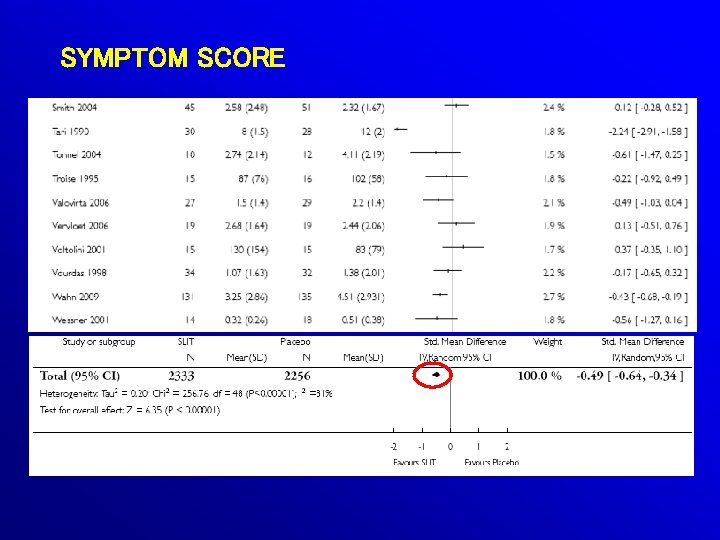
SYMPTOM SCORE
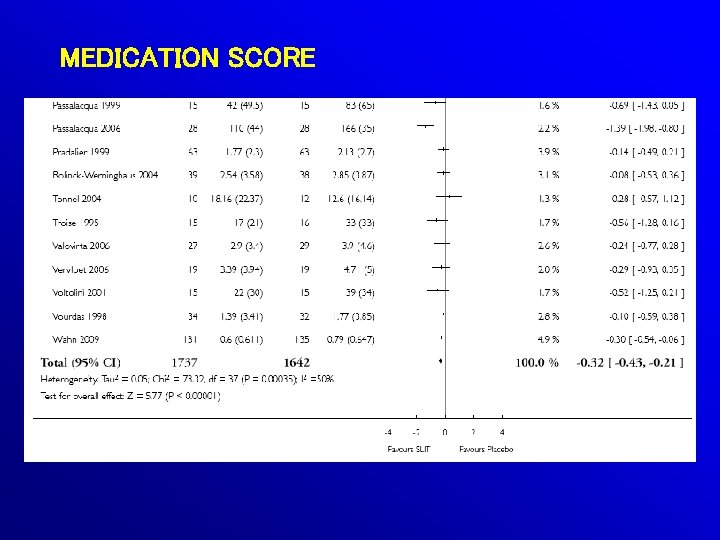
MEDICATION SCORE
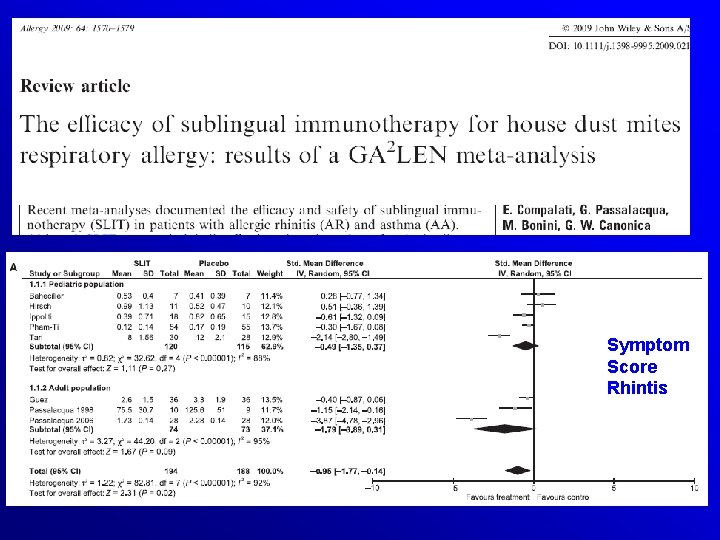
Symptom Score Rhintis
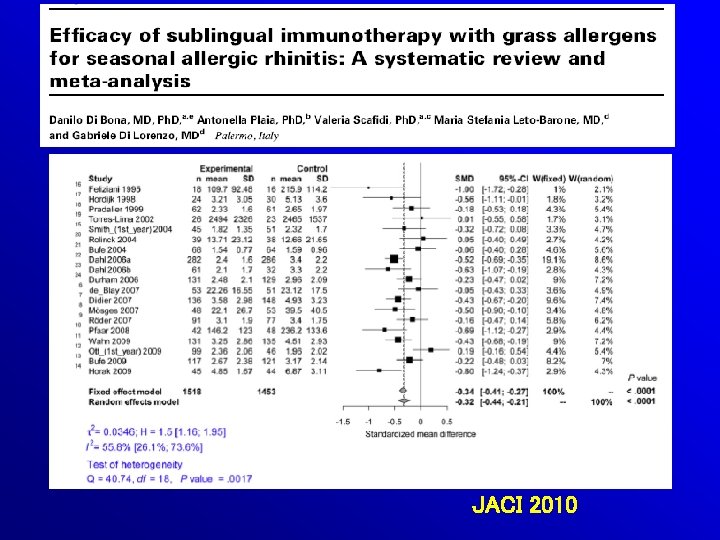
JACI 2010

SLIT No fatal event reported since 1986
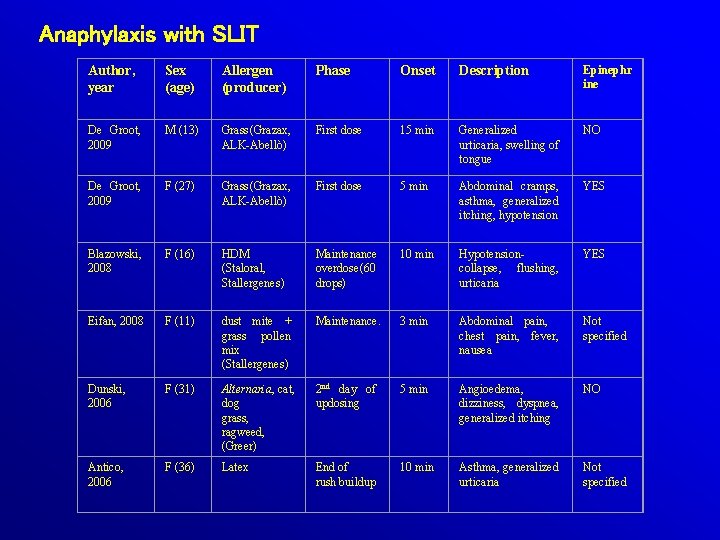
Anaphylaxis with SLIT Author, year Sex (age) Allergen (producer) Phase Onset Description Epinephr ine De Groot, 2009 M (13) Grass (Grazax, ALK-Abellò) First dose 15 min Generalized urticaria, swelling of tongue NO De Groot, 2009 F (27) Grass (Grazax, ALK-Abellò) First dose 5 min Abdominal cramps, asthma, generalized itching, hypotension YES Blazowski, 2008 F (16) HDM (Staloral, Stallergenes) Maintenance overdose (60 drops) 10 min Hypotensioncollapse, flushing, urticaria YES Eifan, 2008 F (11) dust mite + grass pollen mix (Stallergenes) Maintenance. 3 min Abdominal pain, chest pain, fever, nausea Not specified Alternaria, cat, dog grass, ragweed, (Greer) 2 nd day of updosing 5 min Angioedema, dizziness, dyspnea, generalized itching NO Latex End of rush buildup 10 min Asthma, generalized urticaria Not specified Dunski, 2006 Antico, 2006 F (31) F (36)

Cox L, Linneman D, Nolte H, Weldon D, Finegold I, Nelson HS. Sublingual immunotherapy: A comprehensive review. J Allergy Clin Immunol 2006; 117: 1021 -35 In 41 studies with information on the total number of AEs, 1047 adverse reactions in a total of 386, 149 doses were identified (2. 7 reactions/1, 000 doses). In 49 studies with information on the number of patients, the AEs were 529 per 4378 patients (12%). In the trials that specified the severity of reaction, the occurrence of severe AE was 0. 56 per 1, 000 doses. Overall, 14 serious AEs probably related to the treatment were found.
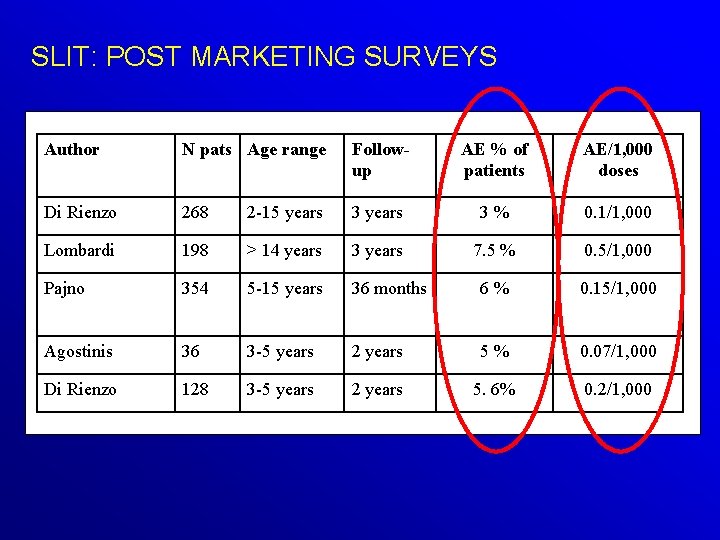
SLIT: POST MARKETING SURVEYS Author N pats Age range Followup AE % of patients AE/1, 000 doses Di Rienzo 268 2 -15 years 3 % 0. 1/1, 000 Lombardi 198 > 14 years 3 years 7. 5 % 0. 5/1, 000 Pajno 354 5 -15 years 36 months 6 % 0. 15/1, 000 Agostinis 36 3 -5 years 2 years 5 % 0. 07/1, 000 Di Rienzo 128 3 -5 years 2 years 5. 6% 0. 2/1, 000
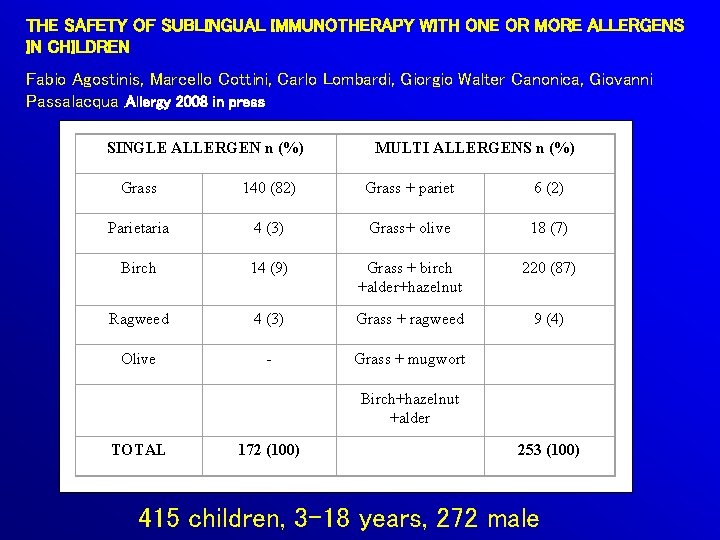
THE SAFETY OF SUBLINGUAL IMMUNOTHERAPY WITH ONE OR MORE ALLERGENS IN CHILDREN Fabio Agostinis, Marcello Cottini, Carlo Lombardi, Giorgio Walter Canonica, Giovanni Passalacqua , Allergy 2008 in press SINGLE ALLERGEN n (%) MULTI ALLERGENS n (%) Grass 140 (82) Grass + pariet 6 (2) Parietaria 4 (3) Grass+ olive 18 (7) Birch 14 (9) Grass + birch +alder+hazelnut 220 (87) Ragweed 4 (3) Grass + ragweed 9 (4) Olive - Grass + mugwort Birch+hazelnut +alder TOTAL 172 (100) 253 (100) 415 children, 3 -18 years, 272 male
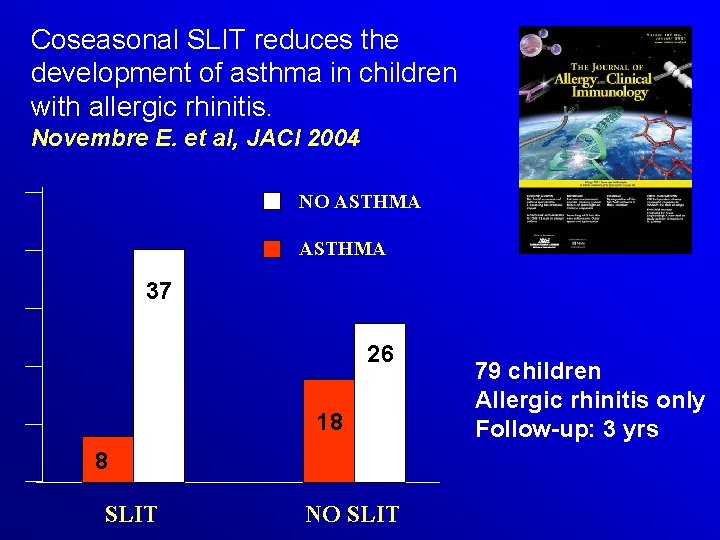
Coseasonal SLIT reduces the development of asthma in children with allergic rhinitis. Novembre E. et al, JACI 2004 NO ASTHMA 37 26 18 8 SLIT NO SLIT 79 children Allergic rhinitis only Follow-up: 3 yrs

PREVENTIVE EFFECTS OF SUBLINGUAL IMMUNOTHERAPY IN CHILDHOOD. AN OPEN RANDOMIZED CONTROLLED STUDY MAURIZIO MAROGNA MD 1 , D. TOMASSETTI 1, A. BERNASCONI 1, F. COLOMBO 1, ALESSANDRO MASSOLO BS 2, A. DI RIENZO BUSINCO 4, GIORGIO W CANONICA 3 AND SALVATORE TRIPODI MD 4 MD 3, GIOVANNI PASSALACQUA MD 1 Pneumology Unit, Cuasso al Monte, Macchi Hospital Foundation, Varese 2 Department of Animal Biology, University of Pavia, Pavia 3 Allergy & Respiratory Diseases, Department of Internal Medicine, Genoa University 4 Pediatric Allergy Unit, S. Pertini Hospital, Rome AAAI 2008, 101: 261
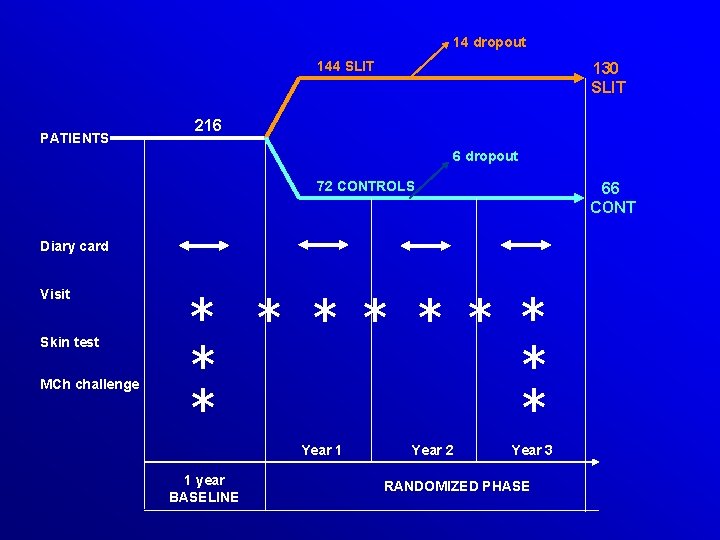
14 dropout 144 SLIT PATIENTS 130 SLIT 216 6 dropout 72 CONTROLS 66 CONT Diary card Visit Skin test MCh challenge * * * Year 1 1 year BASELINE Year 2 Year 3 RANDOMIZED PHASE
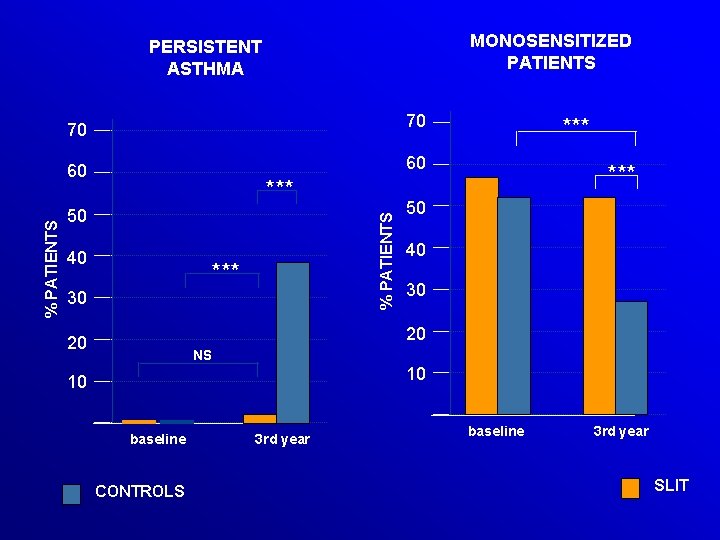
MONOSENSITIZED PATIENTS 70 70 60 60 *** 50 40 % PATIENTS PERSISTENT ASTHMA *** 30 *** 50 40 30 20 20 NS 10 10 baseline CONTROLS 3 rd year baseline 3 rd year SLIT

- Slides: 34