Subcutaneous Implantable Cardioverter Defibrillator SICD Tanner Barnes BME

Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) Tanner Barnes BME 281

The Problem: • 300, 000 people die each year in the United States due to sudden cardiac arrest (SCA) • 80 -90% due to ventricular tachyarrhythmias • People who have severe coronary heart disease are at a heightened risk for (SCA) • Research shows that an implantable cardioverter-defibrillator (ICD) can reduce the chance of dying from (SCA)
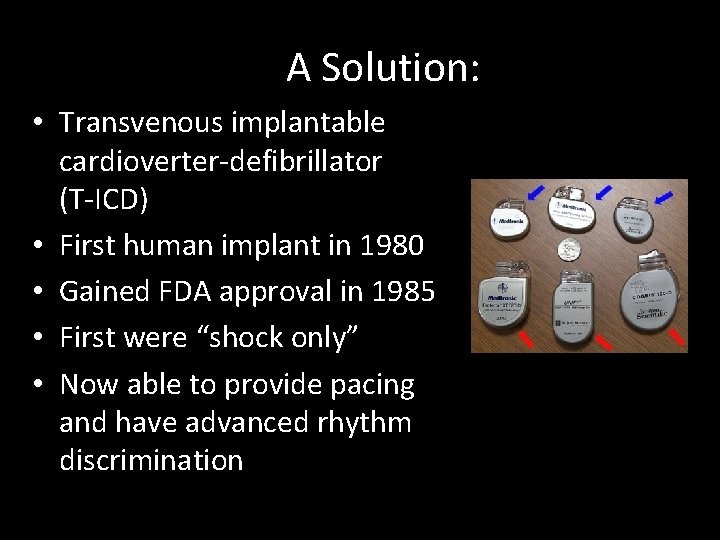
A Solution: • Transvenous implantable cardioverter-defibrillator (T-ICD) • First human implant in 1980 • Gained FDA approval in 1985 • First were “shock only” • Now able to provide pacing and have advanced rhythm discrimination
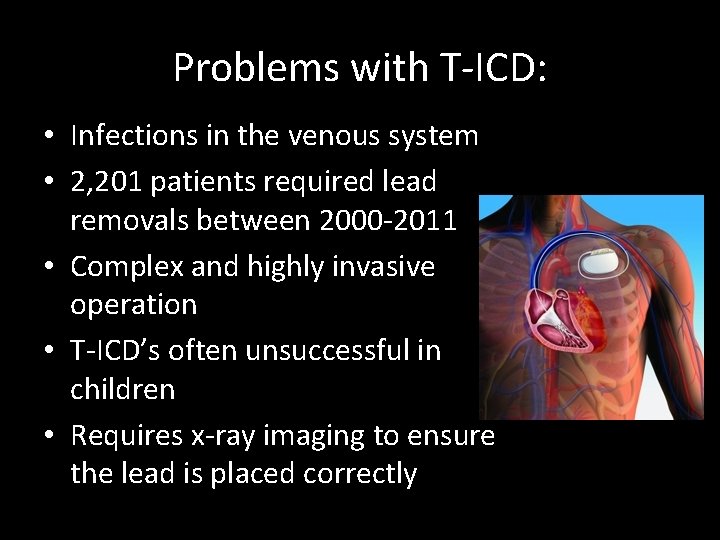
Problems with T-ICD: • Infections in the venous system • 2, 201 patients required lead removals between 2000 -2011 • Complex and highly invasive operation • T-ICD’s often unsuccessful in children • Requires x-ray imaging to ensure the lead is placed correctly

A New Alternative: • Cameron Health, recently purchased by Boston Scientific, pioneered the subcutaneous implantable cardioverter-defibrillator (S-ICD) and it was put into commercial use outside the U. S. in 2009 • The (S-ICD) was approved for observational study by FDA • Awaiting FDA approval for commercial use

Design of S-ICD: The S-ICD System is comprised of the following four devices: 1. SQ-RX Pulse Generator – 80 -J biphasic shock – Charge time to 80 -J ≤ 10 seconds – 5. 1 year longevity – 30 seconds post-shock pacing 2. Q-TRAK Subcutaneous Electrode 3. Q-GUIDE Electrode Insertion Tool (EIT) 4. Q-TECH Programmer
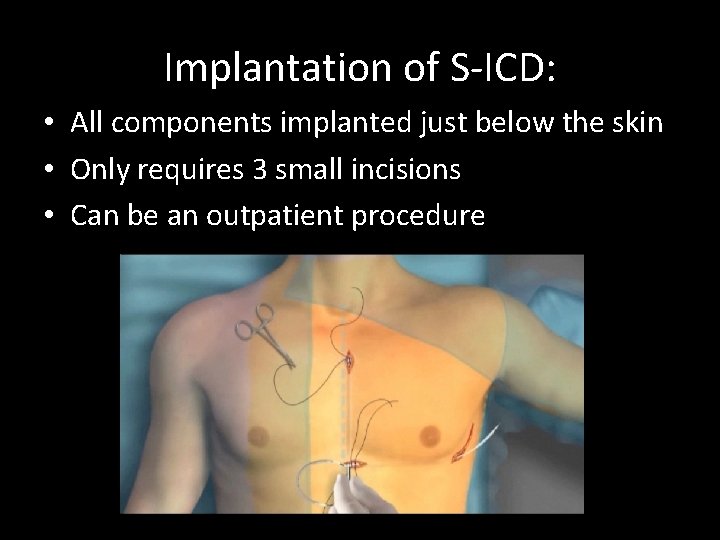
Implantation of S-ICD: • All components implanted just below the skin • Only requires 3 small incisions • Can be an outpatient procedure

Advantages: • Less invasive surgery • Eliminates potential for infection and damage to venous system • May be implanted using anatomical landmarks • Potential for less inappropriate shocks in children

Disadvantages: • Size – Twice that of current T-ICD • Battery life – 5 years as opposed to upwards of 10 with T-ICD • Does not provide anti-tachycardia pacing (ATP) or bradycardia pacing

S-ICD in Children: • Study conducted in UK with two boys, aged 10 and 12 • Early clinical trials suggest less inappropriate shocks than T-ICD • Can be implanted in children >30 kg – Possible for children <30 kg • Adapts to growth better than T-ICD

Latest Observational Study: • 330 patients in 33 different centers • 99% complication free after 180 days • 100% success rate in converting induced VT/VF to sinus rhythm • Only 1 out of 119 spontaneous VT/VF episodes required external defibrillation – Storm episode • 37 out of 38 discrete spontaneous VT/VF episodes were corrected with one or more 80 J shocks

Continued: • The one episode terminated spontaneously while device was charging for second shock • Unintentional therapy occurred in 13. 1% of patients over an 11 -month follow-up – Similar to inappropriate therapy with T-ICD • The mean time to therapy for all inductions was 14. 6 seconds

Conclusion: • The S-ICD system represents a viable alternative to conventional T-ICD therapy in patients at risk of death from VT/VF • Low rate of major complications thus far in European market and in clinical studies • FDA decision on approval should be announced in the near future

References • • • Mc. Leod, Dr. Karen, and Dr. Andrew Mc. Lean. "Implantation of a Fully Subcutaneous ICD in Children. " Authors. Journal compilation. 35. (2012): 20 -23. Web. 19 Oct 2013. Hauser, Dr. Robert. "The Subcutaneous Implantable Cardioverter-Defibrillator. " Journal of the American College of Cardiology. 61. 1 (2013): 20 -22. Web. 19 Oct 2013. "Safety and Efficacy of a Totally Subcutaneous Implantable-Cardioverter Defibrillator. " Circulation. 128. (2013): 944 -953. Web. 20 Oct. 2013. "Superior Vena Cava Defibrillator Coils Make Transvenous Lead Extraction More Challenging and Riskier. " Journal of the American College of Cardiology. 61. 9 (2013): 987 -989. Web. 20 Oct. 2013. Boston Scientific. Web. 20 Oct 2013. <http: //www. bostonscientific. com/cardiac-rhythmresources/cameron-health/sicd-system. html>. "What Causes Sudden Cardiac Arrest? . " National Heart, Lung, and Blood Institute. N. p. , 1 1 2011. Web. 20 Oct 2013. <http: //www. nhlbi. nih. gov/health-topics/scda/causes. html>. "Subcutaneous Implantable Defibrillator (S-ICD) System Advisory Panel Package Summary of Safety and Effectiveness Data. ". FDA. Web. 20 Oct 2013. <http: //www. fda. gov/downloads/Advisory. Committees/Committees. Meeting. Materials/Medical. Devices/M edical. Devices. Advisory. Committee/Circulatory. System. Devices. Panel/UCM 301237. pdf>. <http: //www. epdoc. net/professional/patientpages/Equipment-and-Device-Images. aspx> Boston Scientific. “Fundamentals of ICD Therapy”. <http: //www. columbia. edu/itc/hs/medical/hickey/docs/Evolution%20 of%20 ICD%20 Therapy%20010907. p df>
- Slides: 14