Subcutaneous ICD implants in a Pediatric center Srikant

Subcutaneous- ICD implants in a Pediatric center Srikant Das, MD Director, Electrophysiology and Pacing Arkansas Children’s Hospital arpediatrics. org uams. edu

Background • An entirely subcutaneous ICD system (S-ICD) avoids the need for the placement of electrodes within the heart and can provide clinical advantages especially in pediatric population. • Approved by the Food & Drug Administration (FDA) in 2012 • Gained Category 1 CPT Codes in January 2015 • Shown to be highly effective. • We describe the initial experience of S-ICD implants in four children in electrophysiology laboratory in Arkansas Children’s Hospital. arpediatrics. org uams. edu
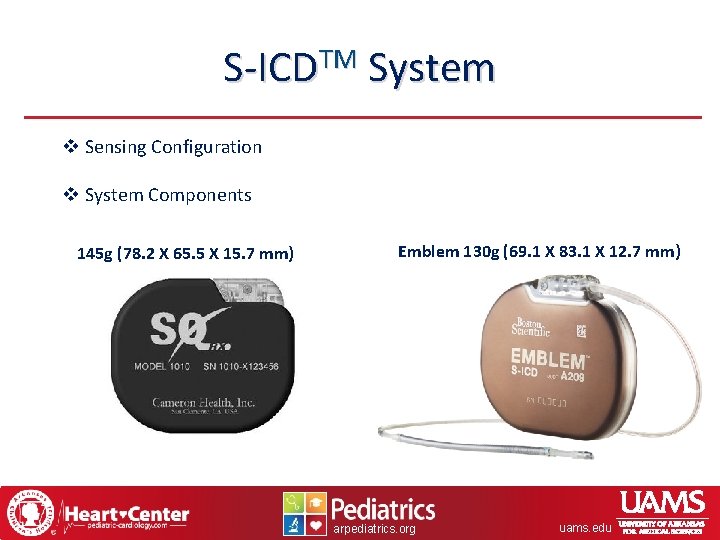
S-ICDTM System v Sensing Configuration v System Components 145 g (78. 2 X 65. 5 X 15. 7 mm) Emblem 130 g (69. 1 X 83. 1 X 12. 7 mm) arpediatrics. org uams. edu

S-ICD in children arpediatrics. org uams. edu
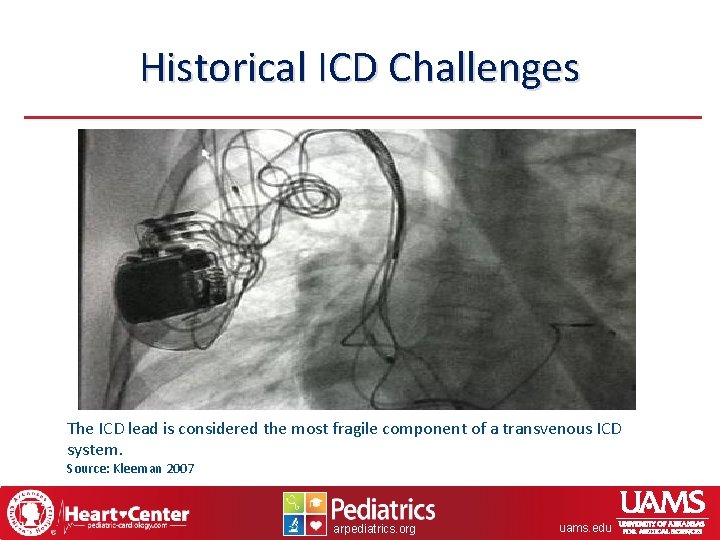
Historical ICD Challenges The ICD lead is considered the most fragile component of a transvenous ICD system. Source: Kleeman 2007 arpediatrics. org uams. edu
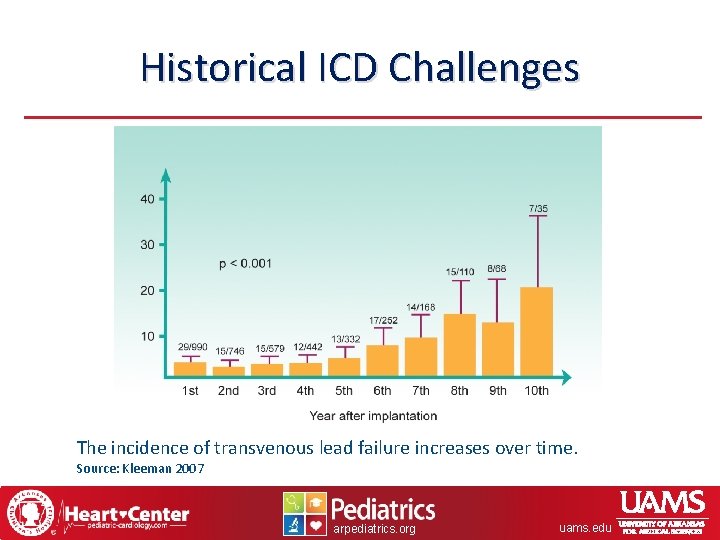
Historical ICD Challenges The incidence of transvenous lead failure increases over time. Source: Kleeman 2007 arpediatrics. org uams. edu
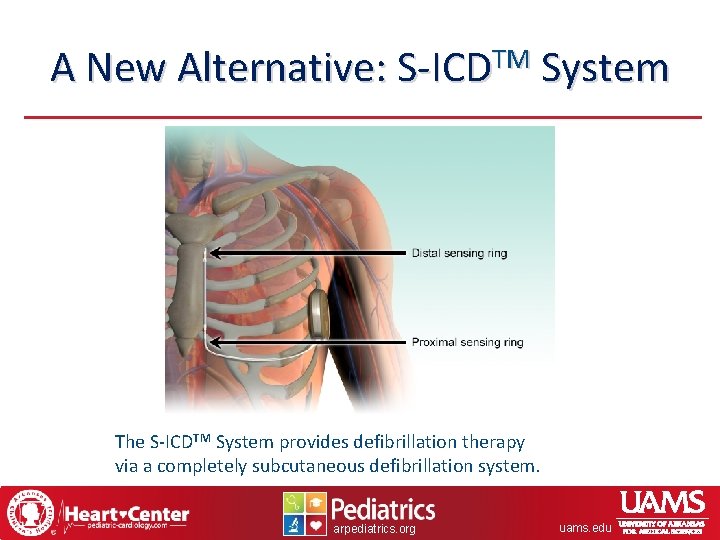
A New Alternative: S-ICDTM System The S-ICDTM System provides defibrillation therapy via a completely subcutaneous defibrillation system. arpediatrics. org uams. edu
Clinical Benefits Because the heart and vasculature remain untouched, the SICDTM System reduces the risks associated with TV-ICDs arpediatrics. org uams. edu

Design of S-ICD The S-ICD System is comprised of the following four devices: 1. Pulse Generator – 80 -J biphasic shock – Charge time to 80 -J ≤ 10 seconds – 5. 1 year longevity – 30 seconds post-shock pacing 2. Q-TRAK Subcutaneous Electrode 3. Q-GUIDE Electrode Insertion Tool (EIT) 4. Q-TECH Programmer arpediatrics. org uams. edu

System Components In addition to the pulse generator and subcutaneous electrode, the S-ICD TM System includes an electrode insertion tool and programmer. arpediatrics. org uams. edu
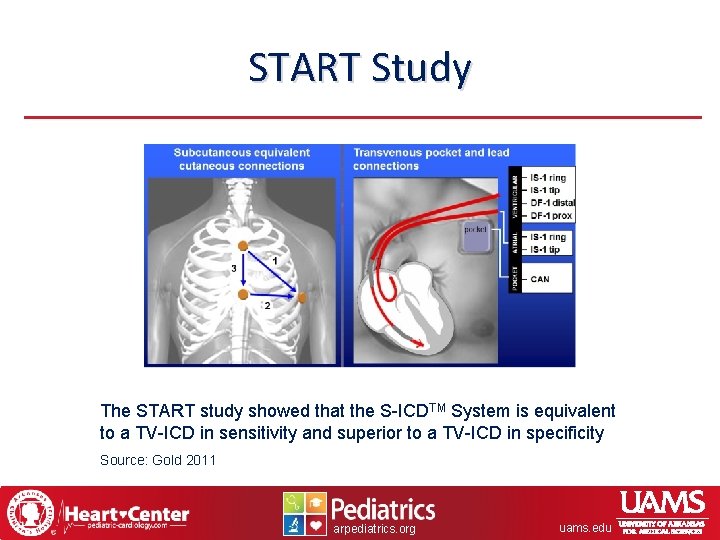
START Study The START study showed that the S-ICDTM System is equivalent to a TV-ICD in sensitivity and superior to a TV-ICD in specificity Source: Gold 2011 arpediatrics. org uams. edu
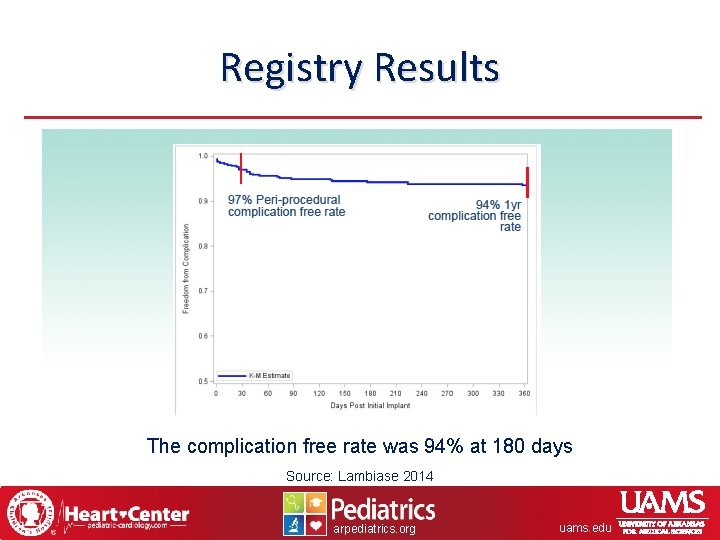
Registry Results The complication free rate was 94% at 180 days Source: Lambiase 2014 arpediatrics. org uams. edu
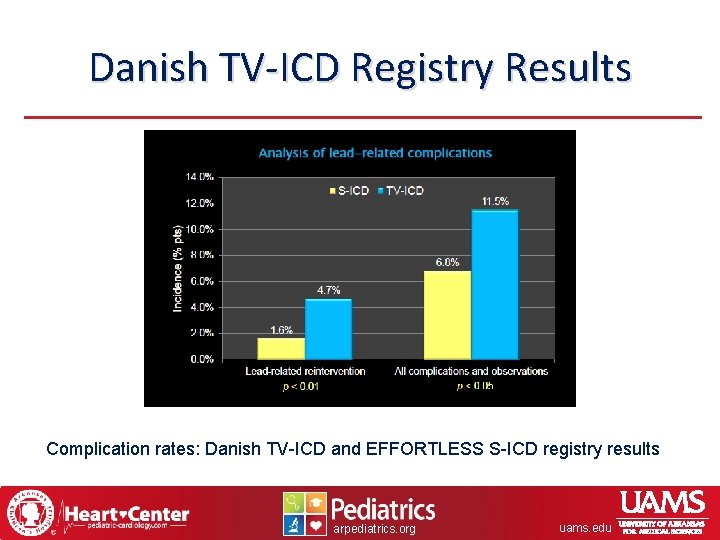
Danish TV-ICD Registry Results Complication rates: Danish TV-ICD and EFFORTLESS S-ICD registry results arpediatrics. org uams. edu
Patient Screening v ECG Screening Tool v ECG Screening Configuration v QRS Evaluation v Lead Acceptability arpediatrics. org uams. edu
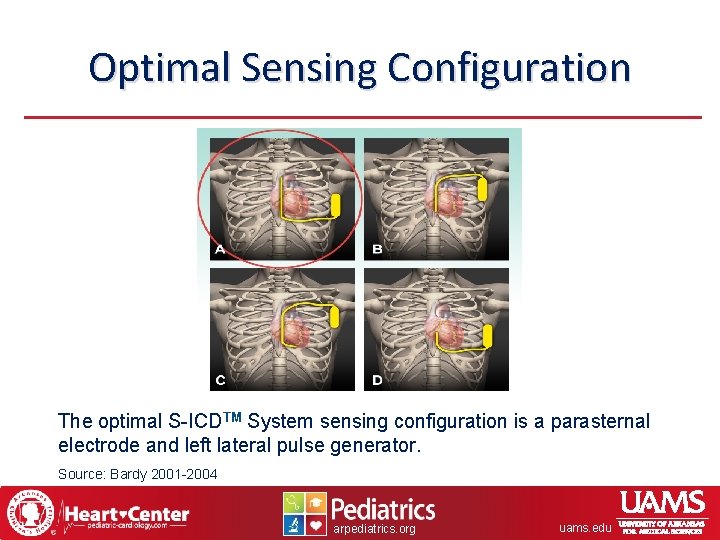
Optimal Sensing Configuration The optimal S-ICDTM System sensing configuration is a parasternal electrode and left lateral pulse generator. Source: Bardy 2001 -2004 arpediatrics. org uams. edu
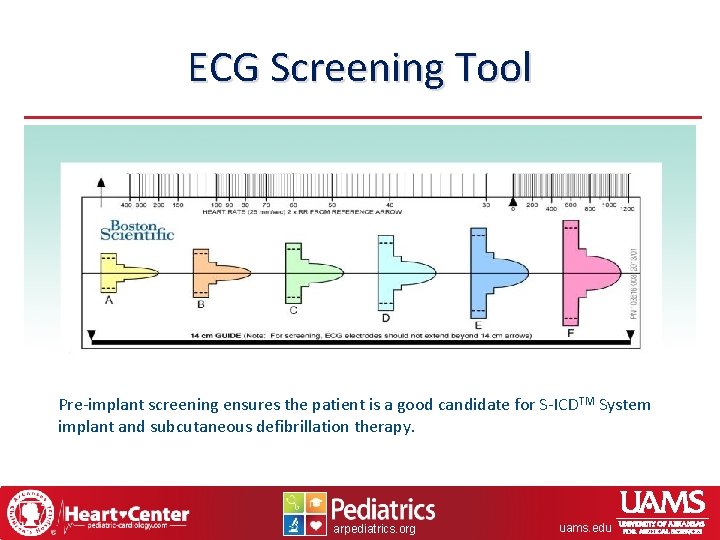
ECG Screening Tool Pre-implant screening ensures the patient is a good candidate for S-ICDTM System implant and subcutaneous defibrillation therapy. arpediatrics. org uams. edu

ECG Screening Configuration Adjust the gain as needed to ensure the peak of each R wave is completely visible…. not clipped as shown here. CRM-151903 -AC FEB 2015 arpediatrics. org uams. edu
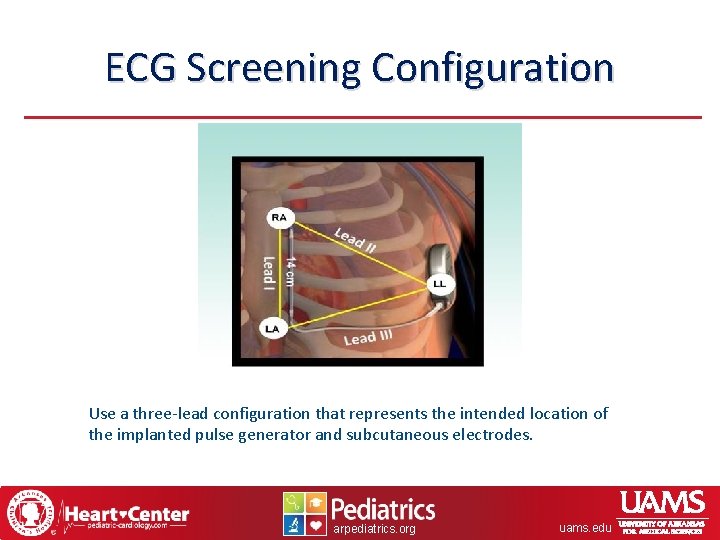
ECG Screening Configuration Use a three-lead configuration that represents the intended location of the implanted pulse generator and subcutaneous electrodes. arpediatrics. org uams. edu
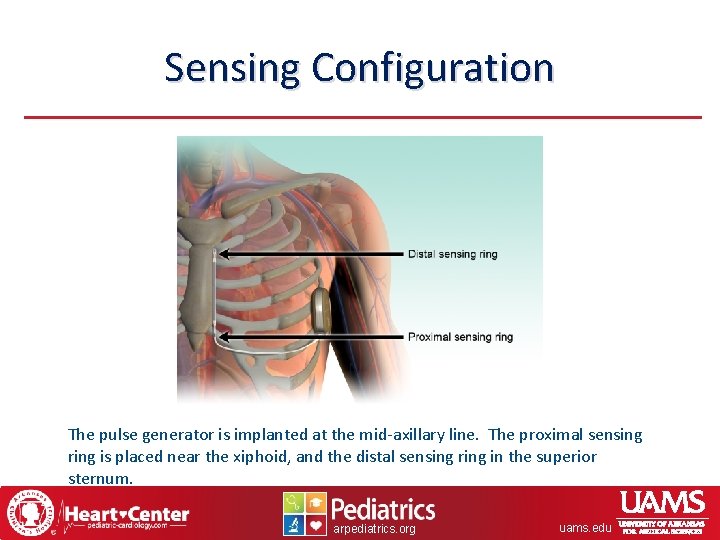
Sensing Configuration The pulse generator is implanted at the mid-axillary line. The proximal sensing ring is placed near the xiphoid, and the distal sensing ring in the superior sternum. arpediatrics. org uams. edu
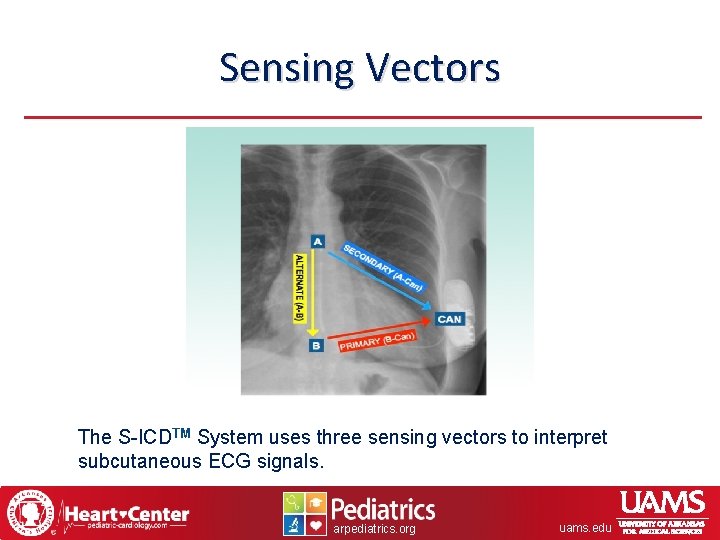
Sensing Vectors The S-ICDTM System uses three sensing vectors to interpret subcutaneous ECG signals. arpediatrics. org uams. edu

QRS Evaluation Select the color profile that best matches the QRS complexes on the ECG strip. Align left edge of color profile to QRS onset. arpediatrics. org uams. edu

QRS Evaluation Ensure the entire QRS complex and T wave fit within the color profile. arpediatrics. org uams. edu

Lead Acceptability A patient is considered suitable for an S-ICD® System implant if at least one ECG lead is acceptable for each tested posture. arpediatrics. org uams. edu
S-ICDTM System Implant v X-ray Landmarks v Patient Preparation v Initial Incisions v Electrode Placement v Pulse Generator Placement v X-ray Assessment arpediatrics. org uams. edu
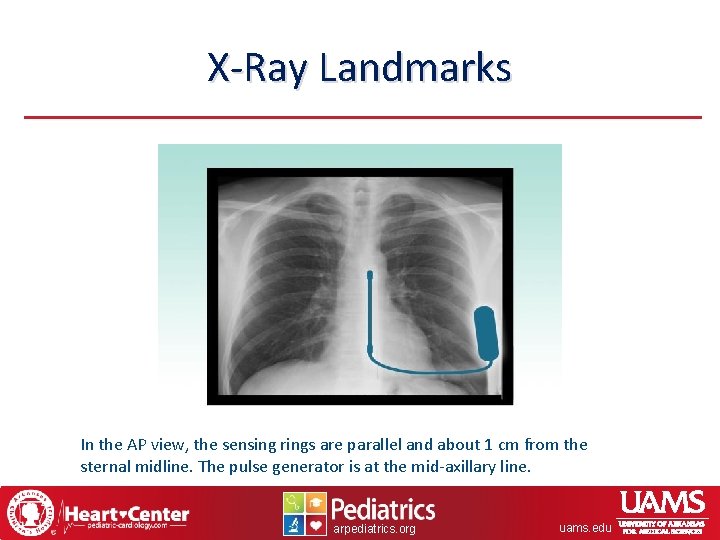
X-Ray Landmarks In the AP view, the sensing rings are parallel and about 1 cm from the sternal midline. The pulse generator is at the mid-axillary line. arpediatrics. org uams. edu
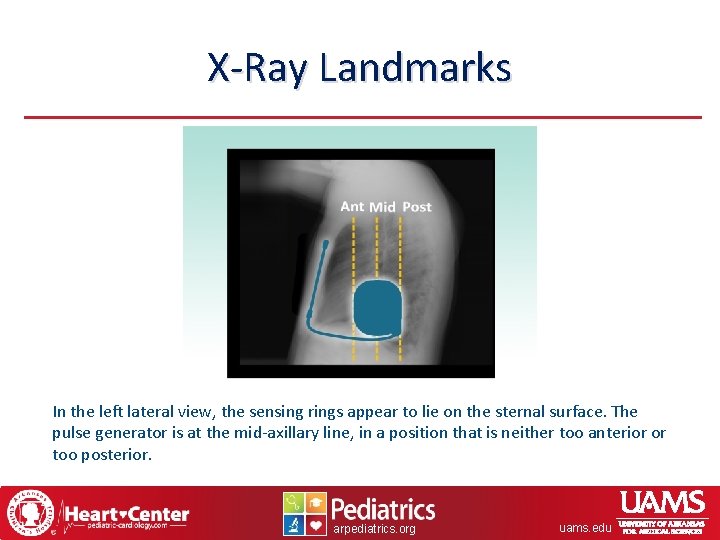
X-Ray Landmarks In the left lateral view, the sensing rings appear to lie on the sternal surface. The pulse generator is at the mid-axillary line, in a position that is neither too anterior or too posterior. arpediatrics. org uams. edu

Implantation Procedure arpediatrics. org uams. edu
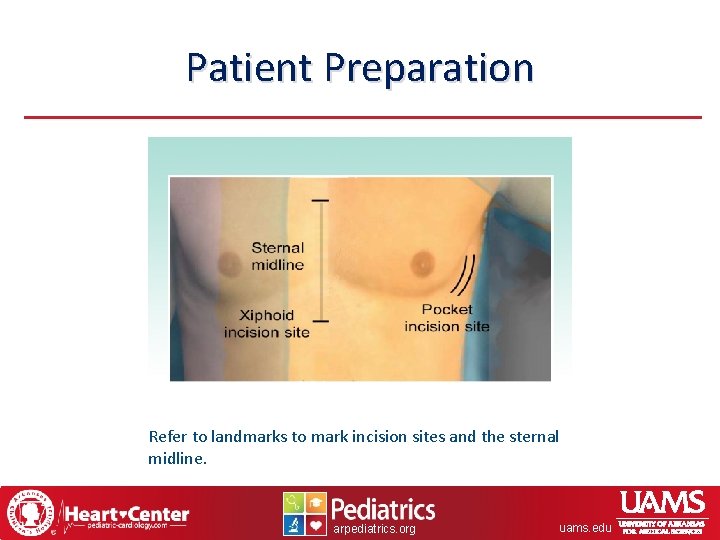
Patient Preparation Refer to landmarks to mark incision sites and the sternal midline. arpediatrics. org uams. edu
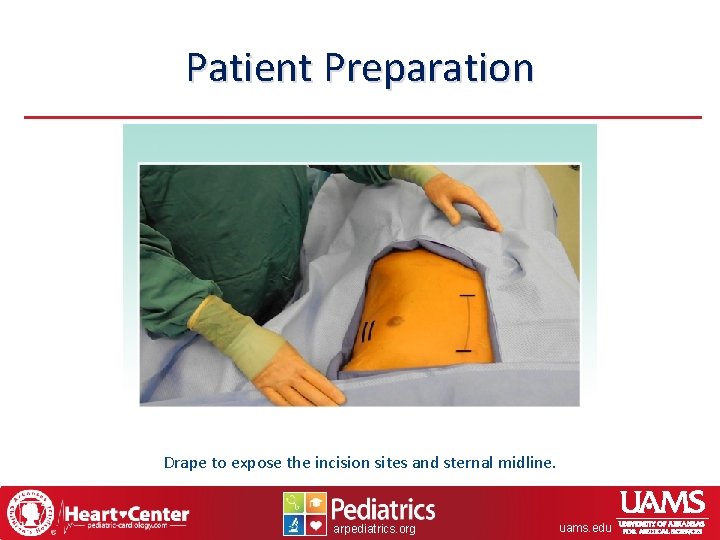
Patient Preparation Drape to expose the incision sites and sternal midline. arpediatrics. org uams. edu
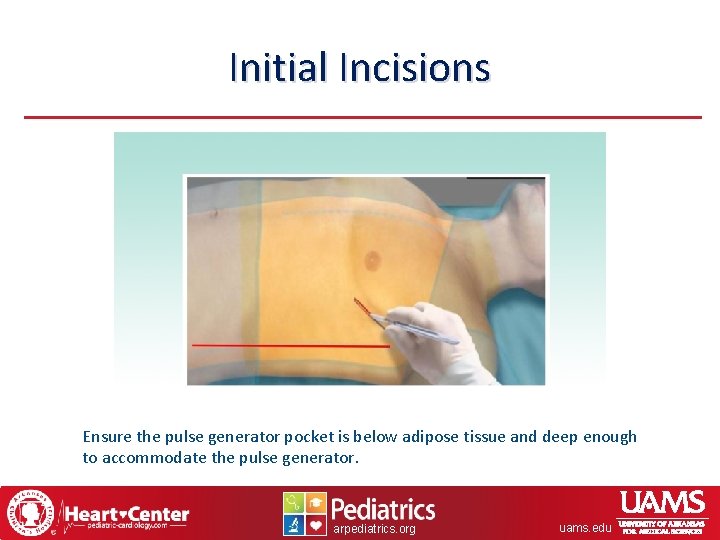
Initial Incisions Ensure the pulse generator pocket is below adipose tissue and deep enough to accommodate the pulse generator. arpediatrics. org uams. edu
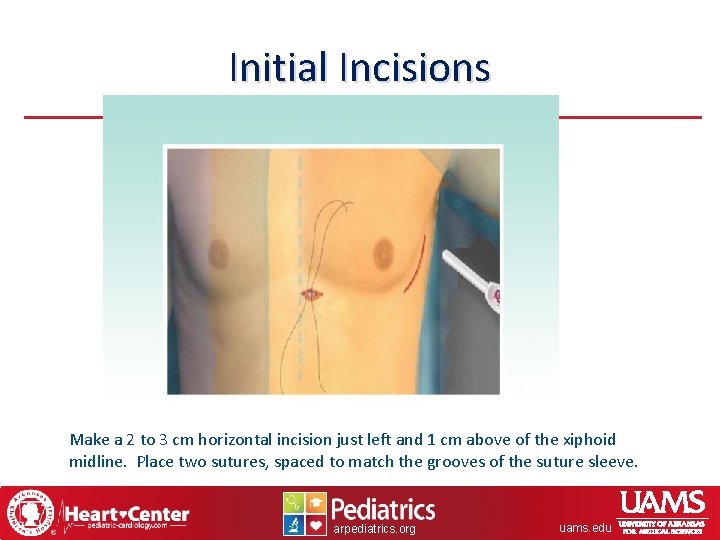
Initial Incisions Make a 2 to 3 cm horizontal incision just left and 1 cm above of the xiphoid midline. Place two sutures, spaced to match the grooves of the suture sleeve. arpediatrics. org uams. edu
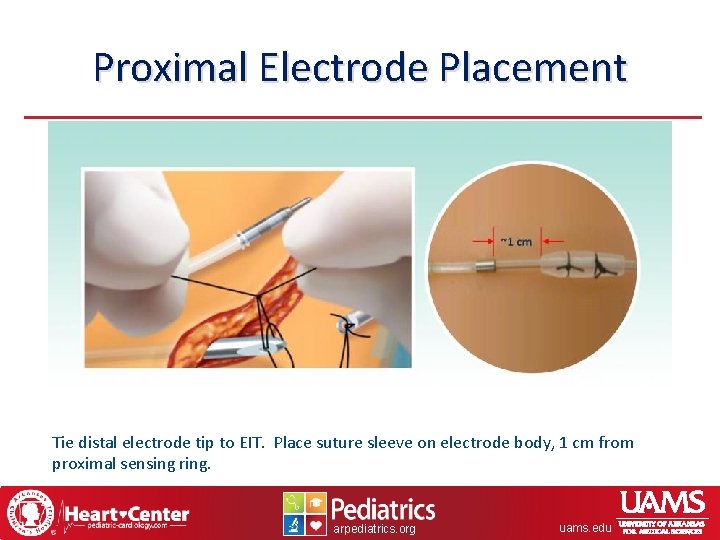
Proximal Electrode Placement Tie distal electrode tip to EIT. Place suture sleeve on electrode body, 1 cm from proximal sensing ring. arpediatrics. org uams. edu
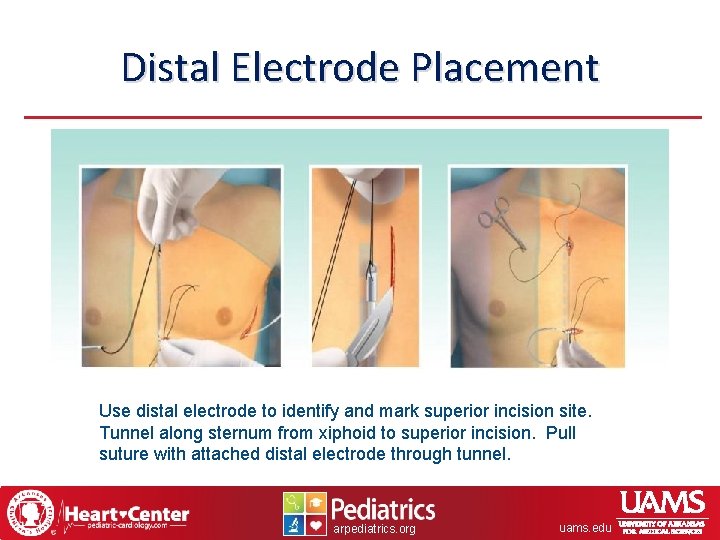
Distal Electrode Placement Use distal electrode to identify and mark superior incision site. Tunnel along sternum from xiphoid to superior incision. Pull suture with attached distal electrode through tunnel. arpediatrics. org uams. edu
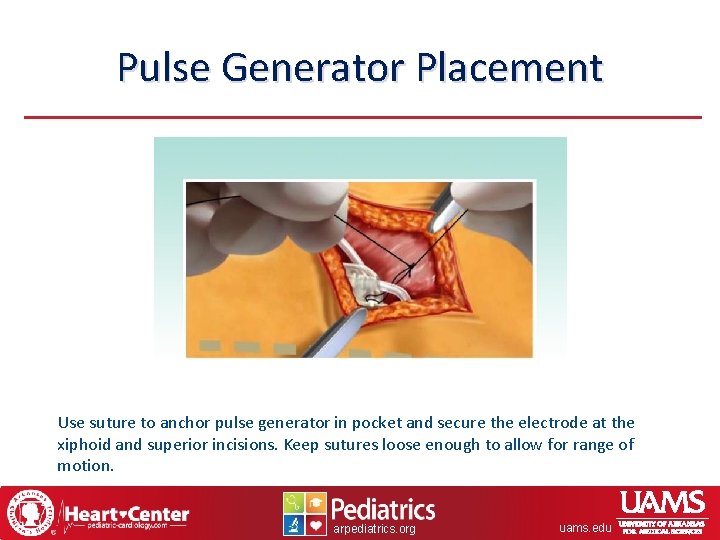
Pulse Generator Placement Use suture to anchor pulse generator in pocket and secure the electrode at the xiphoid and superior incisions. Keep sutures loose enough to allow for range of motion. arpediatrics. org uams. edu
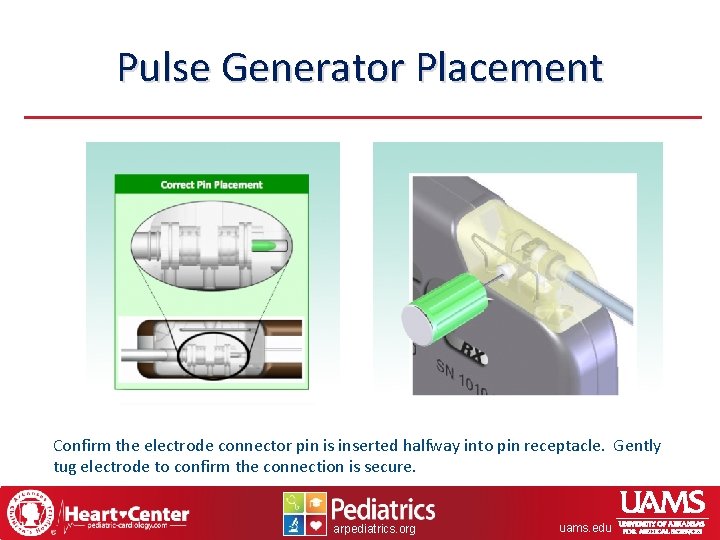
Pulse Generator Placement Confirm the electrode connector pin is inserted halfway into pin receptacle. Gently tug electrode to confirm the connection is secure. arpediatrics. org uams. edu
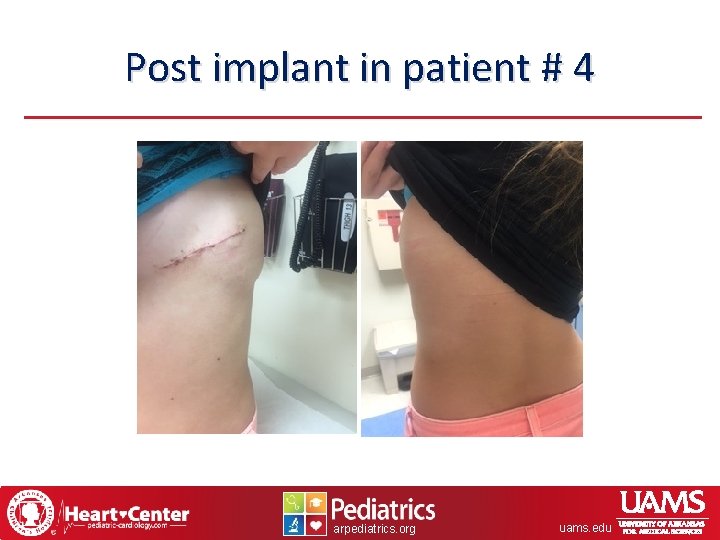
Post implant in patient # 4 arpediatrics. org uams. edu
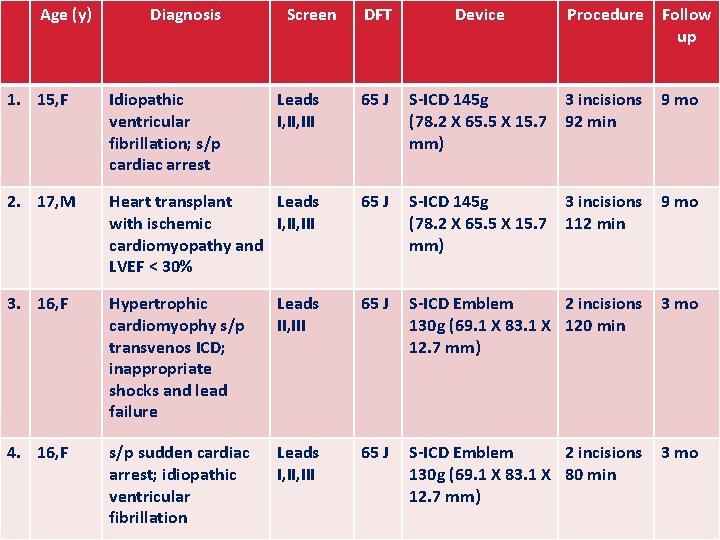
Age (y) Diagnosis Screen DFT Device Procedure Follow up 1. 15, F Idiopathic ventricular fibrillation; s/p cardiac arrest Leads I, III 65 J S-ICD 145 g 3 incisions (78. 2 X 65. 5 X 15. 7 92 min mm) 9 mo 2. 17, M Heart transplant Leads with ischemic I, III cardiomyopathy and LVEF < 30% 65 J S-ICD 145 g 3 incisions (78. 2 X 65. 5 X 15. 7 112 min mm) 9 mo 3. 16, F Hypertrophic cardiomyophy s/p transvenos ICD; inappropriate shocks and lead failure Leads II, III 65 J S-ICD Emblem 2 incisions 130 g (69. 1 X 83. 1 X 120 min 12. 7 mm) 3 mo 4. 16, F s/p sudden cardiac arrest; idiopathic ventricular fibrillation Leads I, III 65 J S-ICD Emblem 2 incisions 130 g (69. 1 X 83. 1 X 80 min 12. 7 mm) 3 mo arpediatrics. org uams. edu
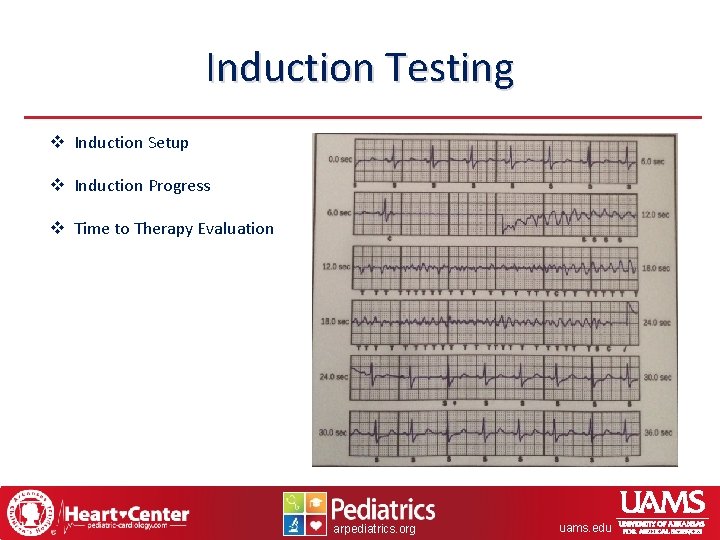
Induction Testing v Induction Setup v Induction Progress v Time to Therapy Evaluation arpediatrics. org uams. edu
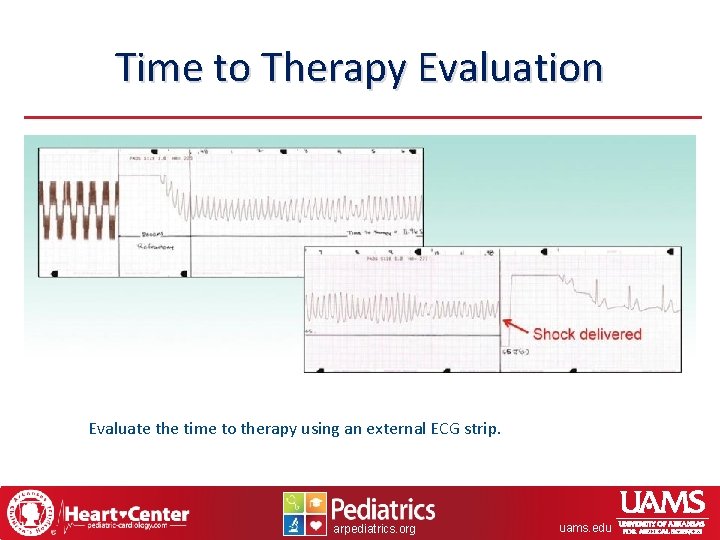
Time to Therapy Evaluation Evaluate the time to therapy using an external ECG strip. arpediatrics. org uams. edu

Appropriate vs Inappropriate Shocks Dual-zone programming enhances AF/SVT vs VT/VF discrimination to determine the appropriateness of shock therapy. Source: Weiss 2013 arpediatrics. org uams. edu

Rhythm Discrimination The S-ICDTM System analyzes static morphology, dynamic morphology, and QRS width to classify the subcutaneous ECG signal in the Conditional zone. arpediatrics. org uams. edu
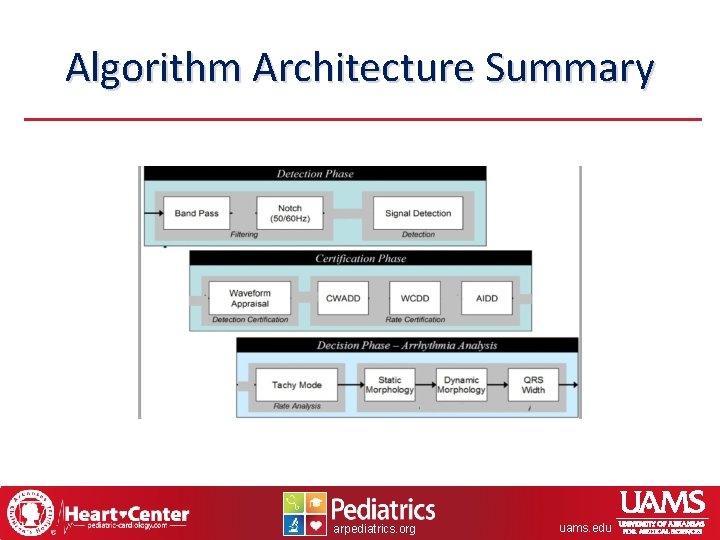
Algorithm Architecture Summary arpediatrics. org uams. edu

Advantages: Advantages • Eliminates potential for infection and damage to venous system • May be implanted using anatomical landmarks without fluoroscopy • Potential for less inappropriate shocks in children arpediatrics. org uams. edu

Disadvantages: • Size – Twice that of current T-ICD • Battery life – 5 to 6 years as opposed to >8 -10 with TV-ICD • Does not provide anti-tachycardia pacing (ATP) or bradycardia pacing arpediatrics. org uams. edu
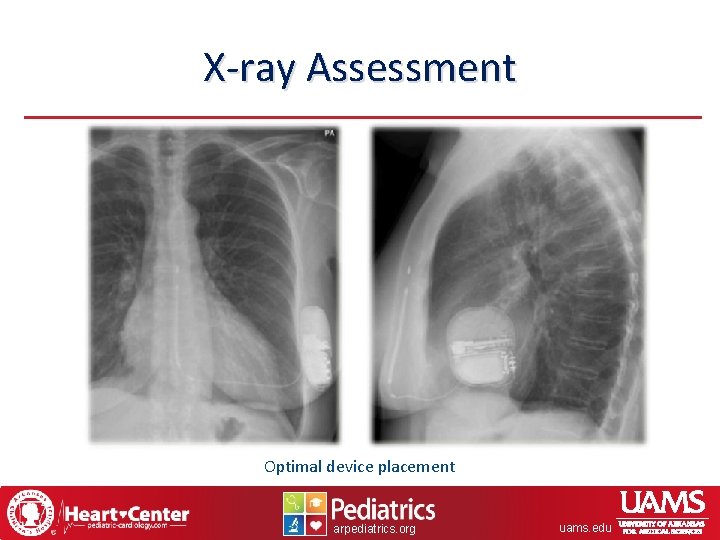
X-ray Assessment Optimal device placement arpediatrics. org uams. edu

Reimbursement • Currently, the S-ICD System is covered nationally by Medicare, Aetna, Cigna and others, and regionally by numerous private and Medicaid plans. arpediatrics. org uams. edu

Conclusion • The S-ICD system represents a viable alternative to conventional TV-ICD therapy in patients at risk of death from VT/VF • Low rate of major complications thus far in clinical studies • Young patients could benefit the most from this system. • The implantation can be safely performed in catheterization laboratory in children. arpediatrics. org uams. edu
- Slides: 47