SubCommittee on DiseaseSpecific Methods And Strategies For Monitoring

Sub-Committee on Disease-Specific Methods And Strategies For Monitoring Relapse Following Allogeneic Stem Cell Transplantation Co-Chairs: Nicolaus Kröger, MD, Alan Wayne, MD Ulrike Bacher, MD Eduardo Olavarria, MD Peter Bader, MD Jerald Radich, MD Sebastian Böttcher, MD Wendy Stock, MD Michael Borowitz, MD, Ph. D Daniel Weisdorf, MD Peter Dreger, MD Andre Willasch, MD Issa Khouri, MD Julie Vose, MD

POINTS TO CONSIDER How can studies using different methods be compared, especially if we are going to change definitions of response? How do we deal with patients with uninformative markers in clinical trials? How do we deal with moving target of increasing sensitivity as techniques improve?

CML SUMMARY • MRD monitoring is well established with Q-PCR for BCR-ABL • PCR positivity predicts for relapse (all types) and disease progression • PCR monitoring can be used for assessing response to treatment of relapse (DLI +/- Imatinib) • Treatment of early relapse (molecular-cytogenetic) results in superior response rates and survival • There is a need for standardization of PCR methodology • Future clinical trials should focus on MRD monitoring after treatment with TKIs post allogeneic SCT

THE FUTURE § Why do some CP cases relapse? Why don’t all BC? § m. RNA signature of aggressive disease? § What is PCR negativity? § Sensitive detection of low abundance m. RNA. § What is the variation of BCR-ABL between cells? § Single cell PCR of BCR-ABL m. RNA.
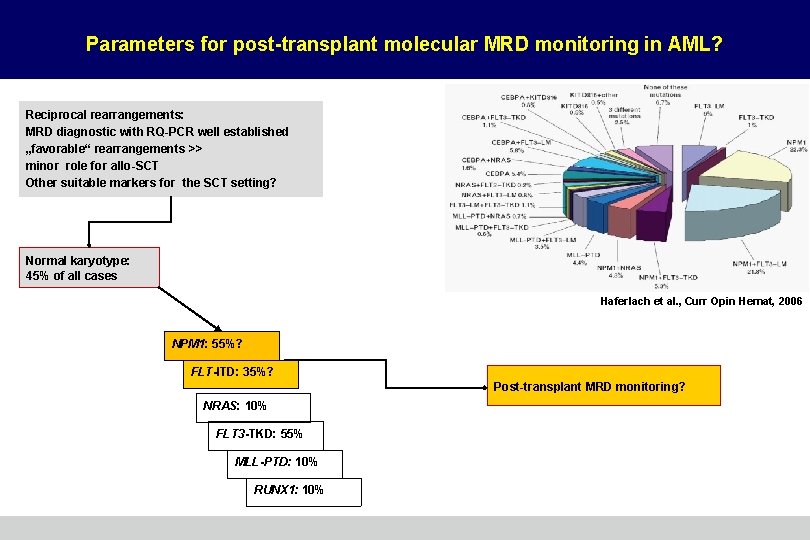
Parameters for post-transplant molecular MRD monitoring in AML? Reciprocal rearrangements: MRD diagnostic with RQ-PCR well established „favorable“ rearrangements >> minor role for allo-SCT Other suitable markers for the SCT setting? Normal karyotype: 45% of all cases Haferlach et al. , Curr Opin Hemat, 2006 NPM 1: 55%? FLT-ITD: 35%? Post-transplant MRD monitoring? NRAS: 10% FLT 3 -TKD: 55% MLL-PTD: 10% RUNX 1: 10%

Conclusions: Post-transplant monitoring in AML and MDS • So far, only very few studies focused on MRD monitoring in AML and MDS specifically in the post-transplant period. • In AML, post-transplant MRD monitoring with RQ-PCR for patients with NPM 1 or FLT 3 mutations should be further evaluated. In MDS, the RUNX 1/AML 1 mutations might represent a utile molecular MRD parameter. • First studies suggest that immunophenotyping with MFC contributes to posttransplant early detection of relapse in AML. The definition of thresholds of LAIPpositive cells for immunotherapeutic intervention, however, requiries further studies. • Monitoring of chimerism offers the possibility of post-transplant monitoring irrespective of the individual subgroup in myeloid malignancies. Interpretation of the results should always consider the kinetics of mixed chimerism. The potential of CD 34+ lineage specific chimerism should be further investigated for both entities. • The combination of chimerism and MRD techniques will improve safety of posttransplant monitoring. It remains to evaluated whether novel mutations - e. g. of TET 2 - might contribute to post-transplant MRD strategies in the future.

Conclusions I n n Immunotherapy (WD of immunosuppression, DLI) is principally effective as pre-emptive treatment Chimerism can be used as surrogate marker for identifying patients at risk for impending relapse n However: n Not in all patients! Additional role for n MRD?

Conclusions II n n MRD prior to stem cell transplantation has a profound impact on post transplant outcome! What adds MRD post transplant?
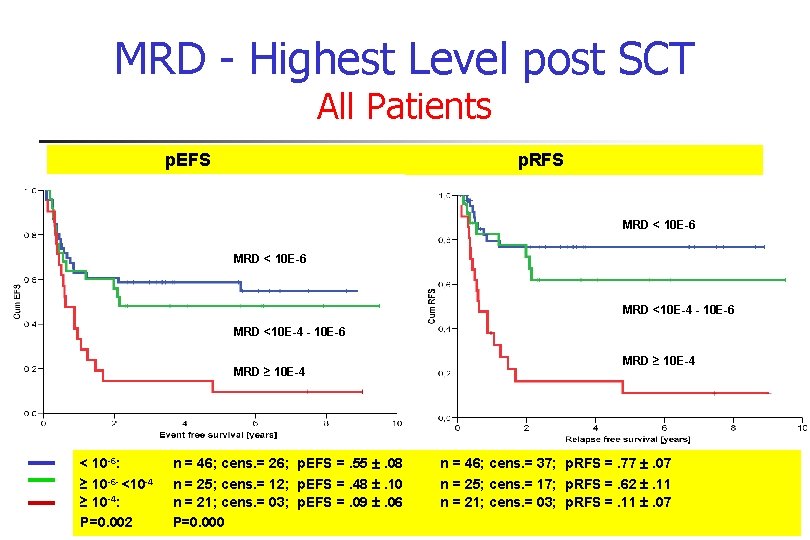
MRD - Highest Level post SCT All Patients p. EFS p. RFS MRD < 10 E-6 MRD <10 E-4 - 10 E-6 MRD ≥ 10 E-4 < 10 -6: ≥ 10 -6 - <10 -4 ≥ 10 -4: P=0. 002 n = 46; cens. = 26; p. EFS =. 55 . 08 n = 25; cens. = 12; p. EFS =. 48 . 10 n = 21; cens. = 03; p. EFS =. 09 . 06 P=0. 000 MRD ≥ 10 E-4 n = 46; cens. = 37; p. RFS =. 77 . 07 n = 25; cens. = 17; p. RFS =. 62 . 11 n = 21; cens. = 03; p. RFS =. 11 . 07

Conclusions III and Summary n MRD assessment in BM post transplant is predictive for relapse n n n Serial BM investigations are warranted. Current working recommendations of the BFM: days 30, 60, 100, 200, 365, at 18 months and 24 months. Summary: n n Patients with mixed chimerism have a high risk for relapse Patients, who become/remain MRD positive >10 -4, have a very high risk to develop relapse n Additional treatment in these patients is warranted

Summary n MRD detection both prior to and following allo. SCT for adults with ALL is associated with poor DFS n Clinical interventions based on MRD measurements suggest utility but data are very limited: n n Allocation to allo. SCT in CR 1 Post-transplant intervention to prevent relapse n n Targeted therapy (e. g. imatinib) following transplant Challenge: implementation of standardized MRD assays that can be done in “real-time” n n Ig. H/TCR q. PCR assays are laborious Data on flow cytometric measurements of MRD in adults with ALL are lacking PB-04/06 tk 05. 06

Summary: MRD after allo. SCT • Techniques: have to be quantitative & sensitive ( 10 -4) • MRD flow • ASO Ig. H q. PCR • Retrospective analyses show that: • delayed, likely GVL-mediated MRD clearance occurs • MRD clearance: • predicts of very low relapse risk • is durable • might serve as surrogate marker for cure • MRD persistence after Cs. A tapering can be used as trigger for preemptive immun-therapy (DLI) Ø Treatment aim to be tested prospectively : MRD negativity (< 10 -4) 12 months after allo. SCT

Perspective: MRD after allo. SCT • Test MRD negativity (< 10 -4) 12 months after allo. SCT prospectively • Treat MRD after allo. SCT using • DLI • alternative treatment options (e. g. Rituximab) • Delineate mechanisms of MRD clearance

Relapse Definition NCI Workshop 1. /2. 11. 2009 Nicolaus Kröger Dept. of Stem Cell Transplantation, University Hospital Hamburg, Germany

CML Standard Definition Molecular relapse (The date of molecular relapse is the date of the first positive RT-PCR assay. ) Is said to be present in a CML patient lacking any other evidence of the disease (i. e. patient in hematological remission and cytogenetic remission) at least 4 months after SCT when any of the following apply: Three samples over a minimum of 4 weeks show a BCR-ABL/ABL ratio higher than 0. 02% as measured by quantitative RT-PCR tests. Three samples over a minimum of 4 weeks show clearly rising levels of BCR-ABL/ABL ratio with the last two higher than 0. 02% as measured by quantitative RT-PCR tests. Two samples over a minimum of 4 weeks show a BCR-ABL/ABL ratio higher than 0. 05% as measured by quantitative RT-PCR tests. Cytogenetic relapse Any of the following in a patient lacking any clinical or hematological evidence of the disease (i. e. patient in hematological remission): Presence of one or more Ph-positive metaphases with standard cytogenetics or hypermetaphase FISH; >2% cells with the BCR-ABL fusion gene by interphase FISH Hematological relapse All of the following: Abnormal blood or marrow counts or morphology consistent with CML. Cytogenetic and/or molecular confirmation of the presence of the disease. Hematological relapse is sub-classified into chronic phase, accelerated phase or blastic phase according to WHO criteria

CML Proposal Disease Definition of CR Definition of Relapse Molecular marker Chromosome Chimerism CML Hematologic Cytogenetic Molecular BCR-ABL RT-PCR Cytogenetic (incl FISH ) PCR or VNTR/STR applicable All patients All patients q. PCR identifies relapse risk groups Not as sensitive as q. PCR for MRD detection Comment: Imaging Flow cytometry 4 -6 color flow Not applicable subgroups Only helpful in identifying aberrant blasts in advanced phase disease

Myelofibrosis Standard Definition Progressive Disease: Requires one of the following: • Progressive splenomegaly that is defined by the appearance of a previous absent splenomegaly that is palpable at greater than 5 cm below the left costal margin or a minimum of 100% increase in palpable distance for baseline splenomegaly of 5 -10 cm or a minimum of 50% increase in palpable distance for baseline splenomegaly of greater than 10 cm. • Leukemic transformation confirmed by bone marrow blast count of at least 20% • Increase in peripheral blood blast percentage of at least 20% that lasts for 8 weeks • Relapse: Changes from CR to PR or CR/PR to Clinical improvement

Myelofibrosis Proposal Disease Definition of CR Relapse Molecular marker Chromosome Chimerism Imaging Flow cytometry Myelofibrosis IWG-MRT JAK 2/MPL Cytogenetic (incl FISH) PCR/VNTR MRT Flow-cytometry applicable All pts Subgroups All pts Comment: Not fully applicable High sensitivity and Not investigated Correlates with Circulating CD 34+ predictive for molecular marker, fibrosis cells may be useful relapse but less specific regression

AML Standard Definition (Cheson et al. , 2003) Parameters Complete remission Morphological/ BM blasts < 5%; hematological criteria thrombocytes ≥ 100 x 109/L; neutrophils ≥ 1. 0 x 109/L Cytogenetic criteria Major cytogenetic remission: Disappearance of cytogenetic alteration Molecular remission Flow cytometry Minor cytogenetic remission: > 50% reduction of abnormal metaphases Disappearance of molecular mutation Disappearance of cells with previously determined LAIP Relapse Reappearance of blasts post CR (BM: > 5%; PB) Reappearance of cytogenetic alteration Reappearance of molecular mutation Reappearance of cells with LAIP

MDS Standard Definition (Cheson et al. , 2006) Criteria of remission Parameters Morphologic and Complete remission (CR): bone marrow blasts <5% hematological response without dysplasia, hemoglobin ≥ 11 g/d. L, platelets ≥ 100 x 109/L, neutrophils ≥ 1. 5 x 109/L Partial remission (PR): reduction of blasts by at least 50% or achievement of lower risk category than prior to treatment Cytogenetic response Major cytogenetic response: disappearance of a cytogenetic abnomality Minor cytogenetic response: ≥ 50% reduction of abnormal metaphases

AML / MDS Proposal Disease Definition of CR Definition of Relapse Molecular marker Chromosome Chimerism AML/MDS IWG Mol. Marker Cytogenetic (incl FISH ) PCR or VNTR/STR applicable All pts Subgroups All pts Comment: Well established, but less sensitive Expansion of MRD marker panel for posttransplant monitoring in AML (e. g. NPM 1 –mutations) or MDS (e. g. RUNX 1/AML 1 mutations) No standardization for MRD monitoring, useful for specific aberrations Well established, lack of specificity: investigation of lineage specific chimerism (e. g. CD 34+ cells); and standardization of techniques Imaging Flow cytometry 4 -8 color flow Not applicable All pts Few studies

ALL Standard Definition Progressive Disease: An increase of at least 25% in the absolute number of circulating or bone marrow leukemic blasts or extramedullary disease burden; or Development of new extramedullary disease. Relapsed Disease: The reappearance of leukemia blast cells in the blood or the bone marrow (≥ 25%) or in any other extramedullary site after a CR with confirmation of lymphoid blasts by morphology and flow cytometry, PCR for antigen receptor loci or fusion genes, or cytogenetics/FISH; or Progression to > 25% leukemia blasts in the marrow after a PR. Importantly, isolated extramedullary relapses (e. g. , CNS) are considered relapse from a diagnostic standpoint, although these are commonly approached differently in terms of therapy.
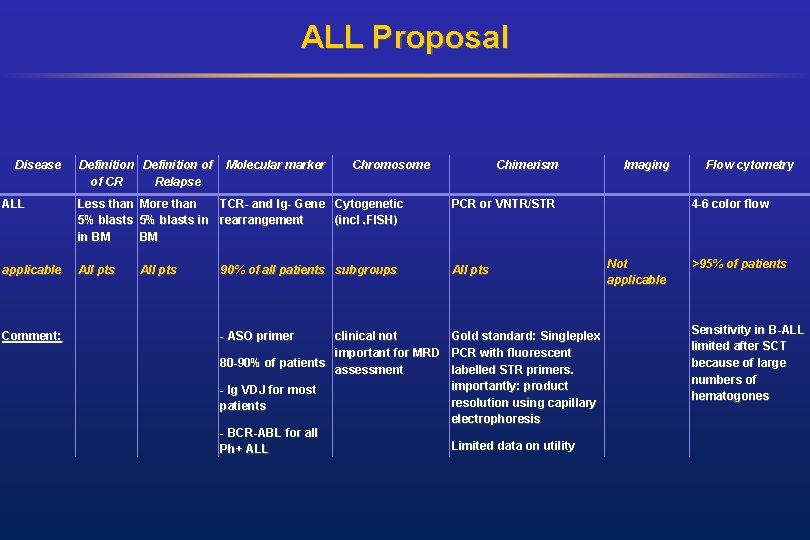
ALL Proposal Disease Definition of of CR Relapse Molecular marker Chromosome Chimerism ALL Less than 5% blasts in BM More than TCR- and Ig. Ig- Gene Cytogenetic 5% blasts in rearrangement (incl. FISH) BM PCR or VNTR/STR applicable All pts Comment: 90% of all patients subgroups - ASO primer clinical not Gold standard: Singleplex important for MRD PCR with fluorescent 80 -90% of patients assessment labelled STR primers. importantly: product - Ig VDJ for most resolution using capillary patients electrophoresis - BCR-ABL for all Limited data on utility Ph+ ALL Imaging Flow cytometry 4 -6 color flow Not applicable >95% of patients Sensitivity in B-ALL limited after SCT because of large numbers of hematogones

CLL Standard Definition • Relapse: progression occurring 6 months or later after having achieved CR or PR • Progression: IW-CLL/NCI-WG criteria for CLL progression (at least one must apply) • Appearance of any new lesion such as enlarged lymph nodes (> 1. 5 cm), splenomegaly, hepatomegaly or other organ infiltrates; • increase of lymphadenopathy by 50% or more in greatest determined diameter of any previous site, or an increase of 50% or more in the sum of the product of diameters of multiple nodes; • increase in the liver or spleen size by 50% or more or the de novo appearance of hepatomegaly or splenomegaly; • increase in the number of blood lymphocytes by 50% or more with at least 5/n. L B cells; • transformation to a more aggressive histology (e. g. Richter's syndrome). • occurrence of cytopenia (neutropenia, anemia or thrombocytopenia) attributable to CLL. • Complete MRD response: clinical remission in the absence of one CLL cell per 10, 000 leukocytes in the peripheral blood or bone marrow • MRD relapse: Tumor cell recurrence or increases at the MRD level that does not exceed 5 B cells/n. L in the peripheral blood.

CLL Proposal Disease Definition of CR Definition of Relapse Molecular marker Chromosome Chimerism Imaging Flow cytometry CLL iw. CLL/NCI iw. CLL/NC ASO-primer IGH q. PCR Cytogenetic PCR/VNTR (incl FISH) CT MRD flow applicable All pts ~90% subgroup All pts > 95% Comment: iw. CLL definition of MRD negativity: predictive for No role in sustained remission relapse if < 10 -4 1 year post monitoring SCT. Only to be used if CR by clinical methods or in clinical trials predictive for sustained remission if < 10 -4 1 year post SCT. MRD < 10 -4 by q. PCR or MRD Flow More sensitive than MRD flow below Complete donor chimerism usually prerequisite for MRD negativity, but not suitable as MRD marker 10 -4 Equally sensitive and specific as q. PCR up to 10 -4
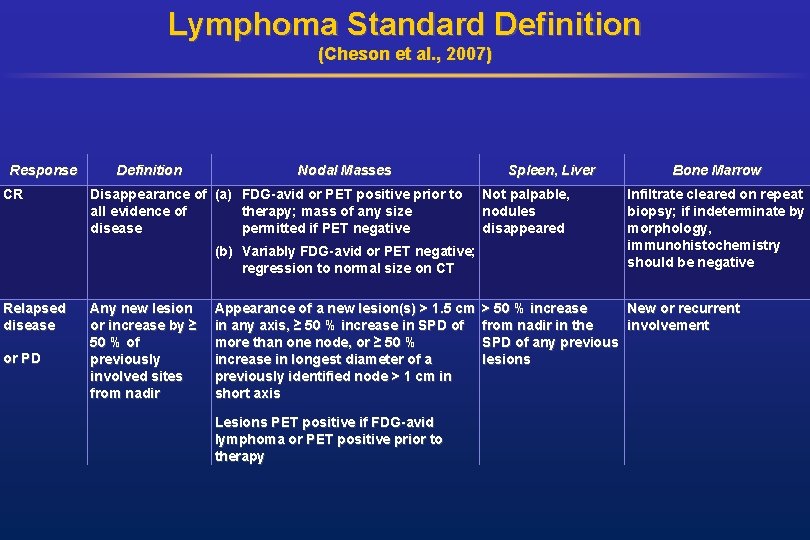
Lymphoma Standard Definition (Cheson et al. , 2007) Response CR Definition Nodal Masses Disappearance of (a) FDG-avid or PET positive prior to all evidence of therapy; mass of any size disease permitted if PET negative (b) Variably FDG-avid or PET negative; regression to normal size on CT Relapsed disease or PD Any new lesion or increase by ≥ 50 % of previously involved sites from nadir Appearance of a new lesion(s) > 1. 5 cm in any axis, ≥ 50 % increase in SPD of more than one node, or ≥ 50 % increase in longest diameter of a previously identified node > 1 cm in short axis Lesions PET positive if FDG-avid lymphoma or PET positive prior to therapy Spleen, Liver Not palpable, nodules disappeared Bone Marrow Infiltrate cleared on repeat biopsy; if indeterminate by morphology, immunohistochemistry should be negative > 50 % increase New or recurrent from nadir in the involvement SPD of any previous lesions

Lymphoma Proposal Disease Definition of CR Definition of Relapse Molecular marker Chromosome Chimerism Imaging Lymphoma Cheson criteria ASO-primer (Ig. H ) for Cytogenetic B-cell NHL (incl FISH) PCR or VNTR/STR CT/PET applicable All patient Comment: Well established Well Bcl-2 for FL t(14; 18) for FL Monitoring T-cell for all established for by PCR useful in Bcl-1 for about 30% of t(11, 14) for MCL lymphomas all lymphomas NHL. Role not MCL established in HD T cell receptor for TNHL subgroups Flow cytometry 4 -6 color flow All patient Subgroups Well established Could be helpful for FL and MCL in all lymphomas

Multiple Myeloma Standard Definition Relapse: EBMT criteria (Bladè et al) requires at least one of the following: • Reappearance of serum or urinary paraprotein on immunofixation or routine electrophoresis, confirmed by at least on further investigation and excluding oligoclonal immune reconstitution. • ≥ 5 % plasma cells in a bone marrow aspirate or on trephine bone biopsy. • Development of new lytic bone lesions or soft tissue plasmacytomas or definite increase in the size of residual bone lesions (development of a compression fracture does not exclude continued response and may not indicate progression). • Development of hypercalcaemia (corrected serum calcium > 11. 5 mg/dl or 2. 8 mmol/l) not attributable to any other cause. IWG Criteria (Durie et al): Relapse from CR requires at least one of the following: • Reappearance of serum or urinary M-protein by immunofixation or electrophoresis • ≥ 5 % plasma cells in a bone marrow. • Appearance of any other sign of progression (i. e new lytic bone lesions or soft tissue plasmacytomas or hypercalcemia).

Multiple Myeloma Proposal Disease Definition of CR Definition of Relapse Molecular marker Chromosome Chimerism Imaging Multiple Myeloma 1) EBMT 2) IWG ASO-primer (Ig. H) Cytogenetic (incl FISH) PCR or VNTR/STR MRI PET-CT 4 -8 color flow applicable All pts 40 -80% subgroups All pts Comment: Accepted but less sensitive Important, but May be useful* not included in EBMT and IWG definition MNC-donor chimerism not useful, lineage specific donor chimerism (CD 138+ plasma cells) predicts relapse Not established, but useful for extramedull ary More sensitive than EBMT/IWG in predicting relapse disease Flow cytometry Other methods Free light chain assay subgroups Proposed by IWG: no valid data

Sub-Committee on Disease-Specific Methods And Strategies For Monitoring Relapse Following Allogeneic Stem Cell Transplantation Panel Discussion

Relapse and Response Definitions After SCT Standard diagnostic criteria used to define response and relapse Ø Well validated in upfront clinical trials Ø Utility after allogeneic SCT is limited for most hematologic malignancies Sensitive disease-specific detection methods Ø Methodologic standardization and validation Ø Highly sensitive monitoring possible Ø Prognostic value in predicting continuous remission vs. relapse Ø Facilitate early intervention Ø Utility “pre-emptive” initiation of therapy prior to overt relapse Proposed incorporation of sensitive detection methods to augment standard response/relapse definitions for use in allogeneic SCT trials Ø Response endpoints Ø Relapse prediction Ø Relapse prevention Ø Relapse treatment

Discussion Points 1. Are the standard diagnostic criteria for relapse and response adequate for use after allogeneic SCT? 2. Proposed incorporation of sensitive detection methods to augment disease-specific definitions after allogeneic SCT A. Methods included for specific diseases B. Value of chimerism C. Discordant results D. Frequency of monitoring 3. Should achievement of molecular remission be the goal of allogeneic SCT? 4. When does molecular relapse or residual disease justify therapeutic intervention?

Research Priorities 1. Harmonization and standardization of molecular monitoring and flow cytometry 2. Define the kinetics of molecular remission and molecular relapse after allogeneic SCT 3. Determine the predictive value of MRD and chimerism (incl lineage-specific) for clinical relapse 4. Apply and assess proposed definitions in studies designed to change the natural history of relapse after SCT 5. Apply and assess proposed definitions in trials of new treatments for prevention and treatment of relapse after SCT
- Slides: 33