Study Designs in Epidemiology Gl Ergr Rescap Med






- Slides: 19

Study Designs in Epidemiology Gül Ergör Rescap. Med Epi 22 -26 Sept Çeşme, İzmir

Aims of medical research n Defining a disease or health condition n Assesing the prevalance or incidence of a disease n Biological mechanism of a disease n Etiology or risk factors n Prognosis n Comparing therapies n Assesing the validity of a diagnostic or screening test

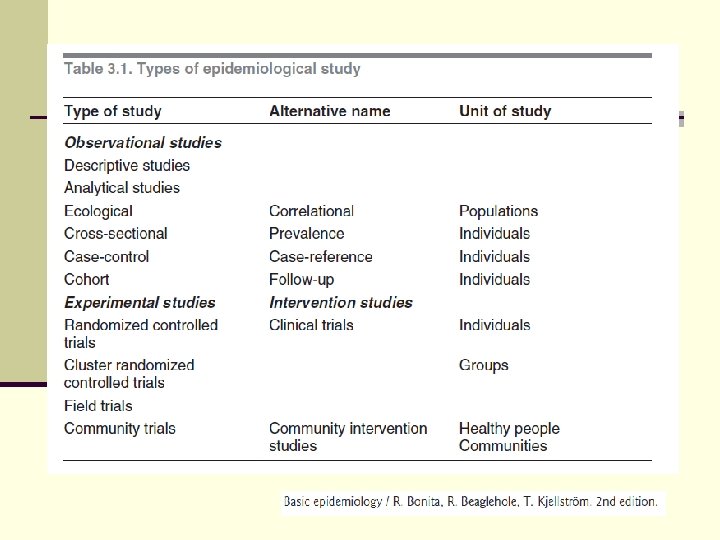
Study Designs OBSERVATIONAL n Descriptive Case report/series Analytic Ecologic studies Cross sectional Case control Cohort EXPERIMENTAL • Clinical Trial • Field Trial


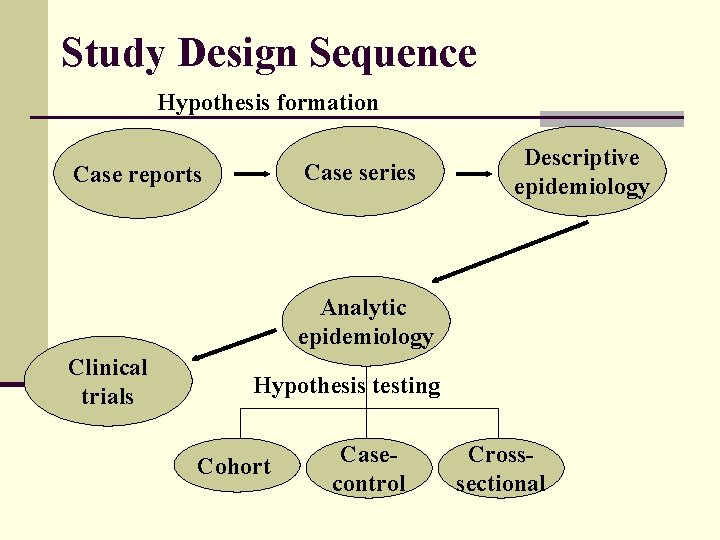
Study Design Sequence Hypothesis formation Case series Case reports Descriptive epidemiology Analytic epidemiology Clinical trials Hypothesis testing Cohort Casecontrol Crosssectional

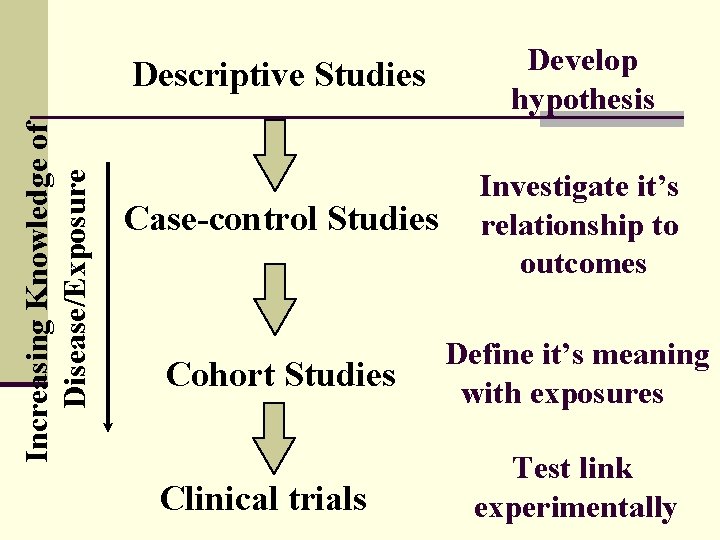
Increasing Knowledge of Disease/Exposure Descriptive Studies Develop hypothesis Case-control Studies Investigate it’s relationship to outcomes Cohort Studies Define it’s meaning with exposures Clinical trials Test link experimentally

Descriptive studies n Assemble morbidity or mortality statistics, usually n n n from routine sources Data on health - related variables (e. g. surveillance, health services utilisation) Report the information according to meaningful categories, such as: Time (when it occurs) Place (whereit is found) Person (who is affected)

Analytic studies n The analysis of descriptive data often leads to hypothesis formation based on observed correlations between diseases or health related states and possible determinants. n The hypotheses generated need to be tested by planned field studies designed to examine if: n people with disease have common attributes, or have been exposed to the same agent; n people with particular attributes who are exposed to a specific agent are more likely to develop disease than those who do not have those attributes.

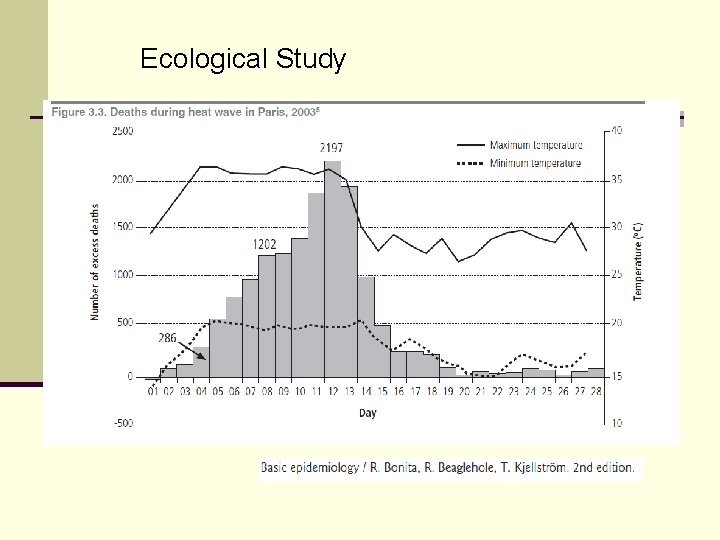
Ecological Study

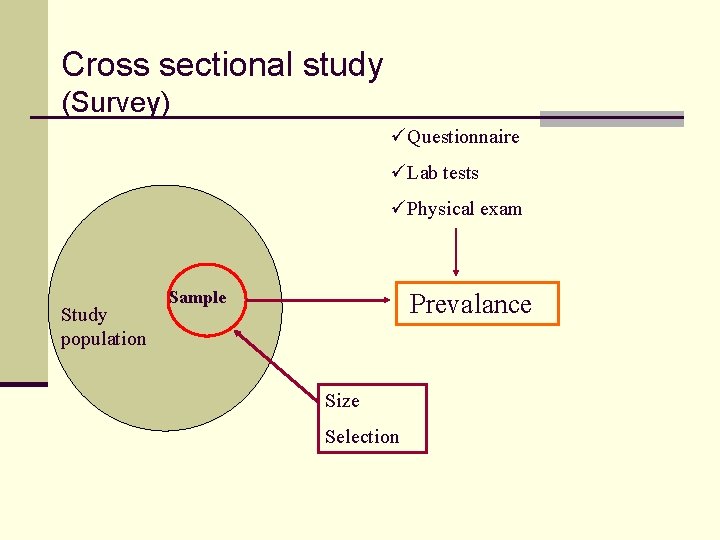
Cross sectional study (Survey) üQuestionnaire üLab tests üPhysical exam Study population Sample Prevalance Size Selection

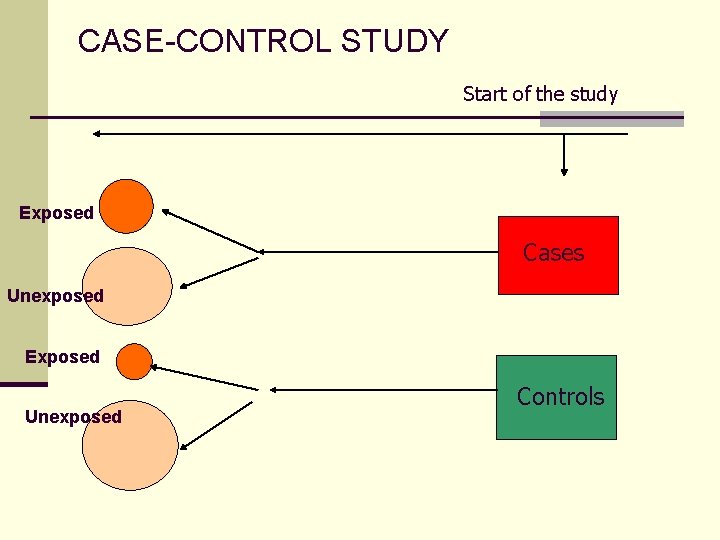
CASE-CONTROL STUDY Start of the study Exposed Cases Unexposed Exposed Unexposed Controls

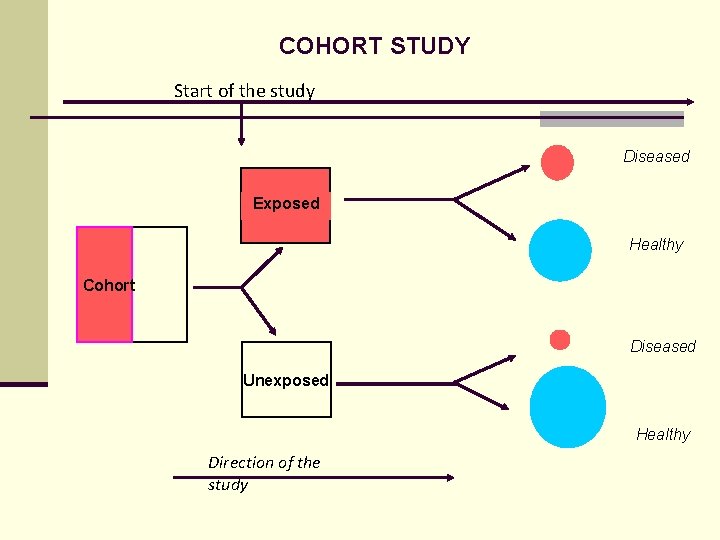
COHORT STUDY Start of the study Diseased Exposed Healthy Cohort Diseased Unexposed Healthy Direction of the study

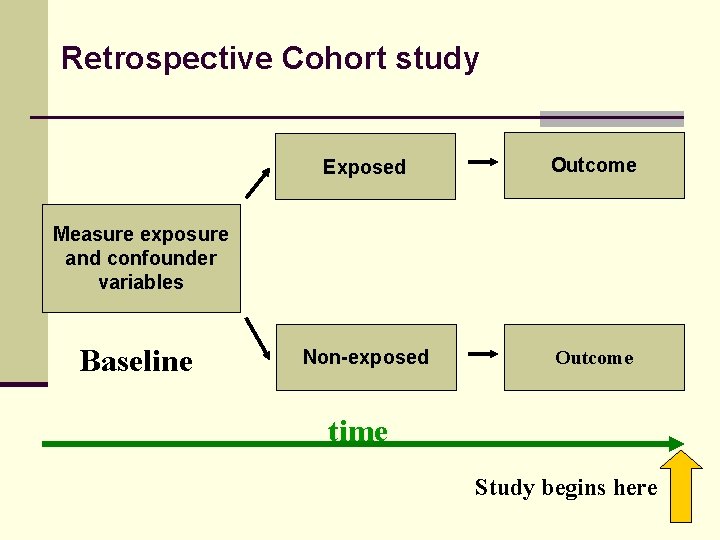
Retrospective Cohort study Exposed Outcome Non-exposed Outcome Measure exposure and confounder variables Baseline time Study begins here

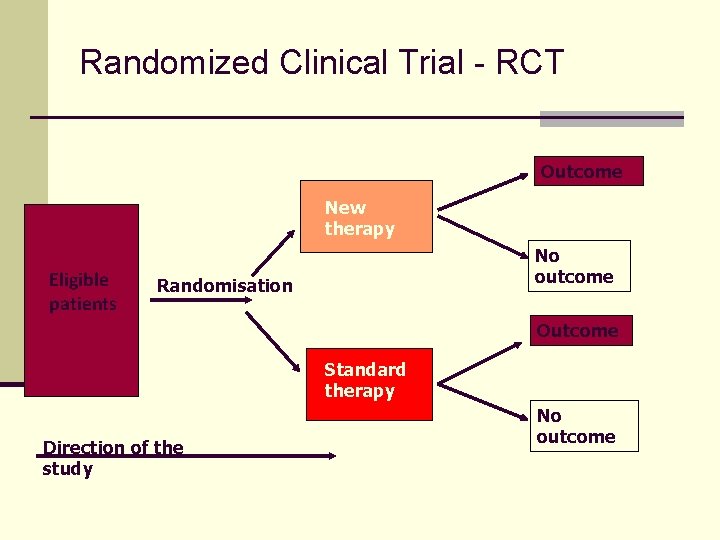
Randomized Clinical Trial - RCT Outcome New therapy Eligible patients No outcome Randomisation Outcome Standard therapy Direction of the study No outcome

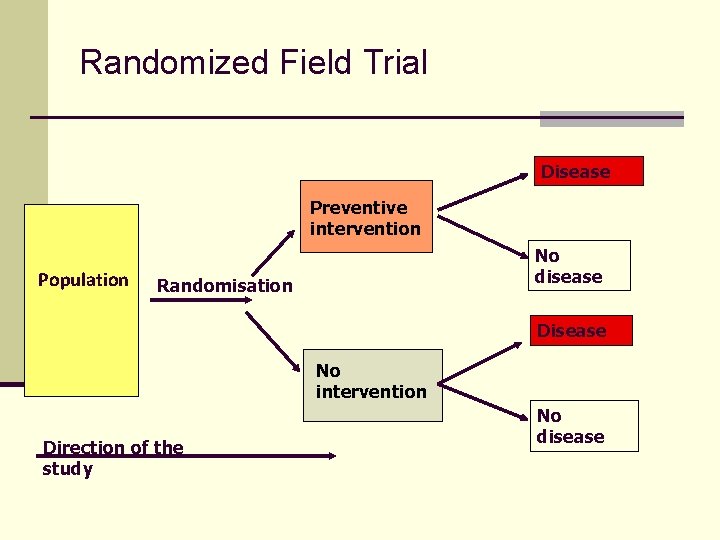
Randomized Field Trial Disease Preventive intervention Population No disease Randomisation Disease No intervention Direction of the study No disease

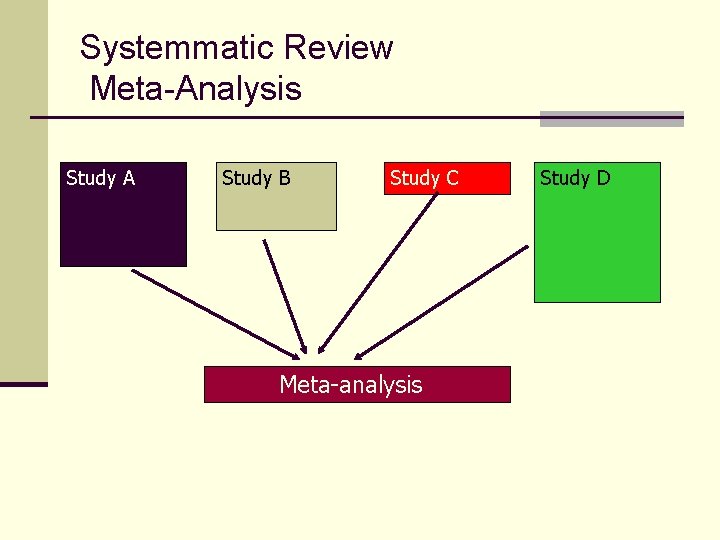
Systemmatic Review Meta-Analysis Study A Study B Study C Meta-analysis Study D

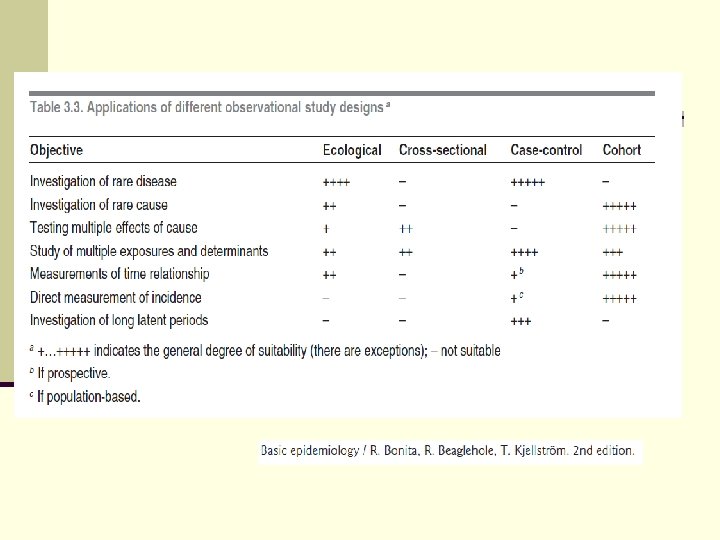
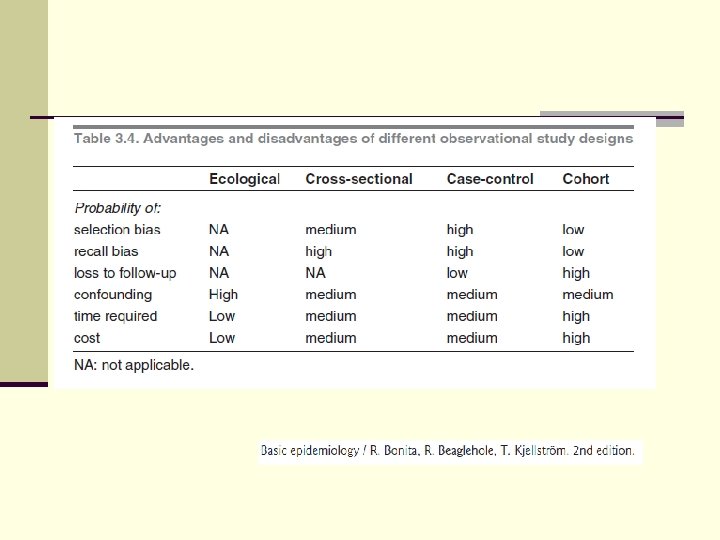
Summary of epidmiologic studies

