Structure of the Atom Subatomic Particles Proton Electron

Structure of the Atom
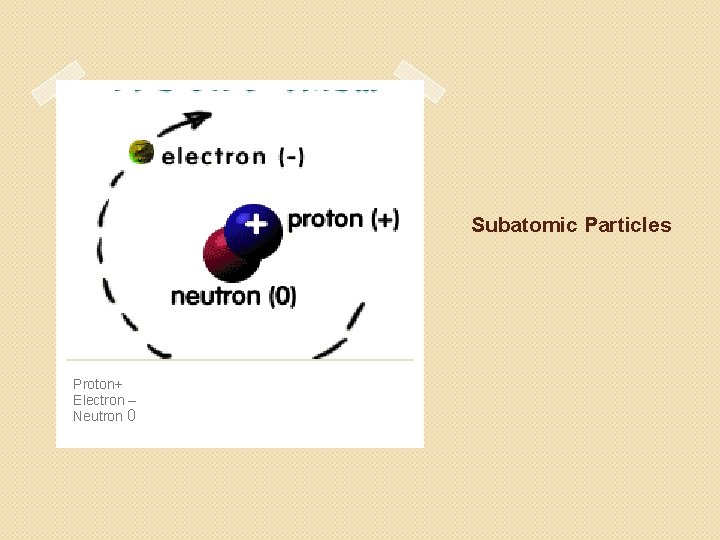
Subatomic Particles Proton+ Electron – Neutron 0

Protons �Positively charged subatomic particle that is found in the nucleus of an atom. �Each proton has a 1+ charge.

Electrons �Negatively charged subatomic particle found in the space outside the nucleus. �Each Electron has a 1 - charge. �Comes from the Greek word meaning “amber”.

Neutrons �A neutral subatomic particle found in the nucleus of an atom. �Mass almost equal to mass of a proton. �Conclude of existence based on an experiment by James Chadwick where the charged particles did not deflect from their paths/

Comparing Subatomic Particles �Protons, electrons and neutrons can be distinguished by mass, charge and location in an atom. �Scientists know about the nucleus and subatomic particles based on hpw particles behave.
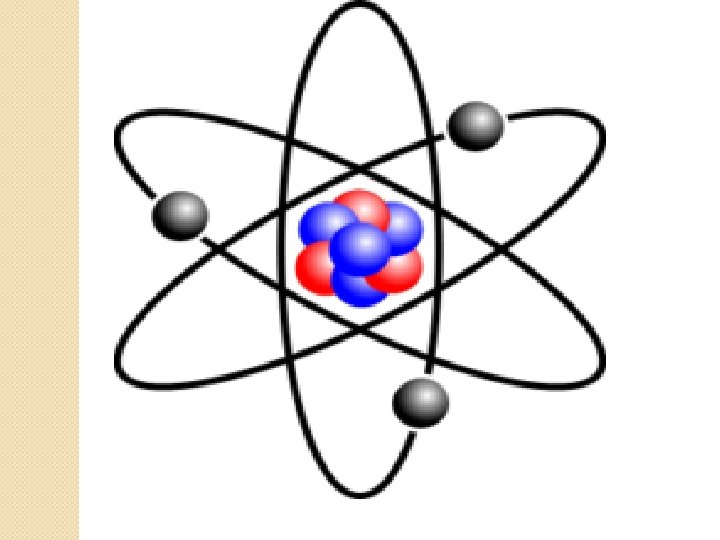

Mass �Protons and Neutrons have similar masses. � 2000 electrons would equal the weight of one proton. �Electron – charge, proton + charge. �Proton and Neutron found in atom nucleus.

Atomic Number �An atom of any given element always have the same amount of protons. �Different elements have different numbers of protons. �Ex: Sulfur (S), atomic number 16=16 protons. Copper (Cu) Atomic number 29 = 29 protons.

Mass Number �Number of Neutrons= Mass Number – Atomic Number. �Ex: Sulfur, Neutrons = 32. 06 -16. Neutrons = 16

Isotopes �Atoms of the same element that have different number of neutrons and different atomic masses. �Ex: Cu 63; Cu 65. or O 16, O 17, O 18

Modern Atomic Theory

Bohr’s Model �Model looks like a solar system. �Focused on the electron.

Energy Levels �Bohr’s model the electrons move in fixed orbits. �Energy of an atom can change if the energy of is gained or lost. �Stair steps…… �An electron in an atom can move from one energy level to another when the atom gains or loses energy.

Energy Levels �Movement of electrons between energy levels explains the light as a firework explodes.

Electronic Cloud �Visual model of the most likely locations for electrons in an atom. �Scientists use the electronic cloud model to describe locations of electrons around the nucleus.

Atomic Orbitals �Region pf space outside of the nucleus where an electron is likely too be found.

Electrons Configuration �Arrangement of electrons in the orbital’s of an atom. �The most stable electronic configuration is the one in which the electrons are in orbital's with the lowest possible energies. �Lowest energy is known as “ground state”

Electron Configurations �Ex: Lithium: silvery-white metal with an atomic number of 3. Has 3 electrons. �In the “ground state”, two electrons are in the orbital of the first energy level. The third electron is in the second energy level. �If lithium atom absorbs enough energy, one electron can move to an orbital with a higher energy.

- Slides: 20