Structure of Materials Prepared by Dental Materials Department

Structure of Materials Prepared by: Dental Materials Department Yenepoya Dental College, Yenepoya University, Mangalore.

“ Properties of materials originate from their internal structures. ” 2
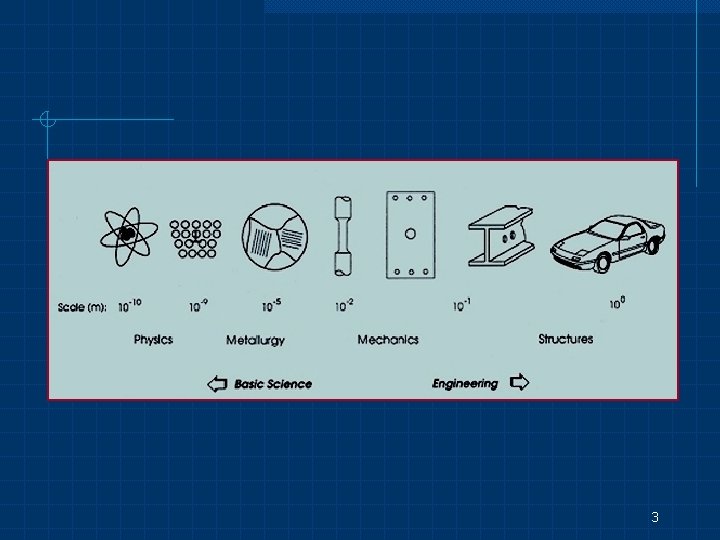
3

Classes of Dental Materials Polymers Metals Ceramics and Glasses Composites General Properties? 4

Polymers (Plastics( Low density Both thermal and electrical insulators Poor reflectors of light Some are flexible and deformable. WHY? 5

Metals High density High thermal and electrical conductivity Opaque Polished to high luster Relatively heavy and deformable WHY? 6

Ceramics and Glasses Hard and brittle Thermally resistant Inert (chemically resistant( WHY? 7

Structures Atomic level n n Atomic bonding Bonding distance/ energy Structural level n n Crystalline structure Noncrystalline structure (Amorphous) 8

Atomic Level

Bonding Primary bonds n n n Ionic, Covalent, Metallic Strong and stiff, do not easily melt with increasing temperature e. g. Metals, Ceramics Secondary bonds n n Hydrogen, van der Waals Weak 10

Ionic Bonding Involves the transfer of one or more electrons between atoms of different types The atom becomes charged ions (e. g. Na+ and Cl-) which attract one another and form a chemical bond due to their opposite electrostatic charges. 11

e. g. salts, oxides, Na. Cl, Mg. O, gypsum Tend to be hard and brittle 12
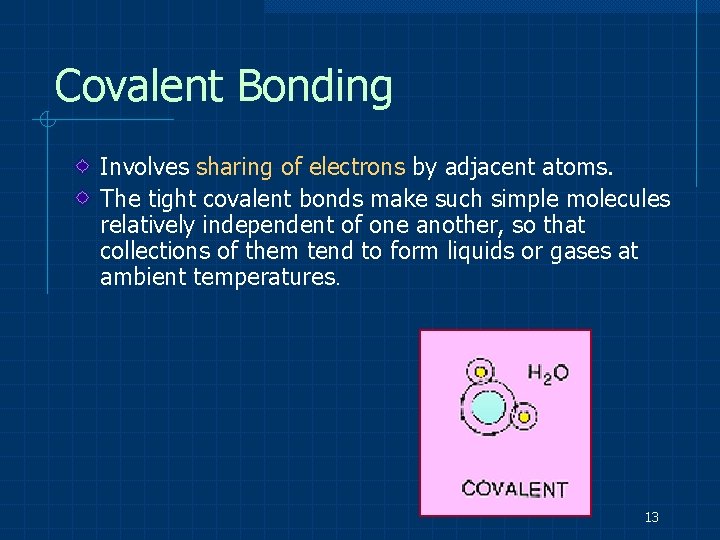
Covalent Bonding Involves sharing of electrons by adjacent atoms. The tight covalent bonds make such simple molecules relatively independent of one another, so that collections of them tend to form liquids or gases at ambient temperatures. 13

Precise bond orientations e. g. H 2, , H 2 O, Cl 2, Methylmethacrylate 14

Metallic Bonding The outer shell of electrons of most metals is less than half full. Each atom donates one or more electrons to a “cloud” of electrons. These electrons are shared in common by all of the metal atoms, which have become positively charged ions as a result of giving up electrons. 15

The metal ions are thus held together by their mutual attraction to the electron cloud. n n Excellent electrical and thermal conductivity Ability to deform plastically 16
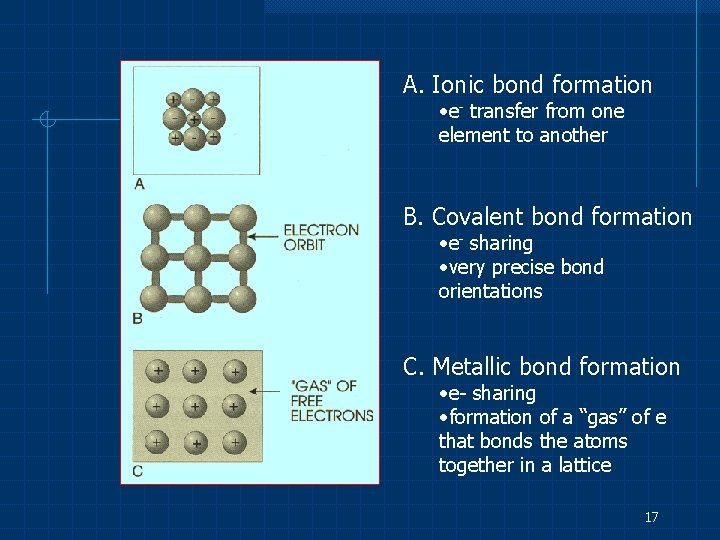
A. Ionic bond formation • e- transfer from one element to another B. Covalent bond formation • e- sharing • very precise bond orientations C. Metallic bond formation • e- sharing • formation of a “gas” of e that bonds the atoms together in a lattice 17

Discussion of Primary Bonds Covalent bonds vs. Ionic and Metallic bonds? n n n Covalent bonds Sharing of electrons with specific neighboring atoms Ionic and Metallic bonds electrostatic attraction involving all neighboring ions Covalent: Bonding being strongly directional to form a three-dimensional network to make a solid w The crystal is very hard and stiff e. g. carbon in the form of diamond. 18

Many solids, such as Si. O 2 and other ceramics have a mixed ionic-covalent character. (more common than pure bonding( 19

Secondary Bonds Occur due to the presence of an electrostatic dipole, which can be induced by a primary bond. n n Hydrogen bond van der Waals bond 20
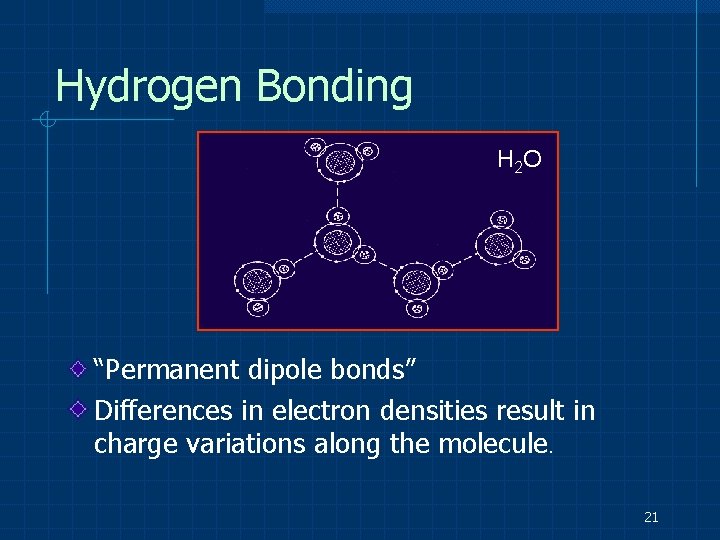
Hydrogen Bonding H 2 O “Permanent dipole bonds” Differences in electron densities result in charge variations along the molecule. 21

van der Waals Forces “Fluctuating dipole bonds” Fluctuating positions of electrons relative to an atom’s nucleus ( the dipole is not fixed in direction( 22
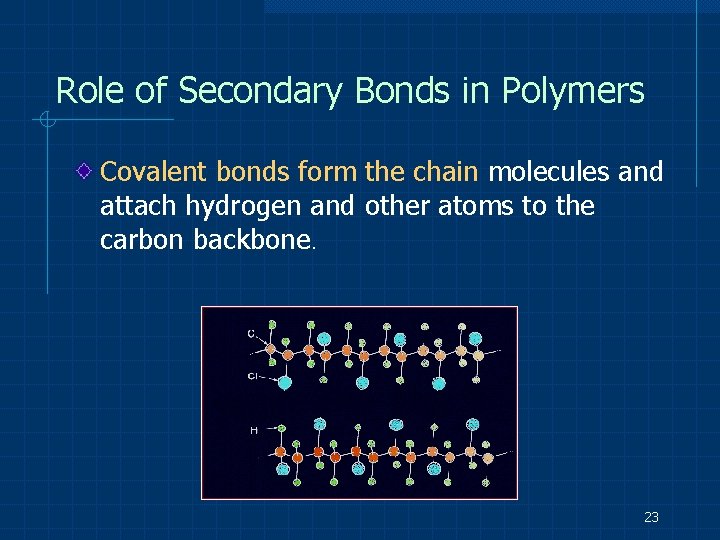
Role of Secondary Bonds in Polymers Covalent bonds form the chain molecules and attach hydrogen and other atoms to the carbon backbone. 23
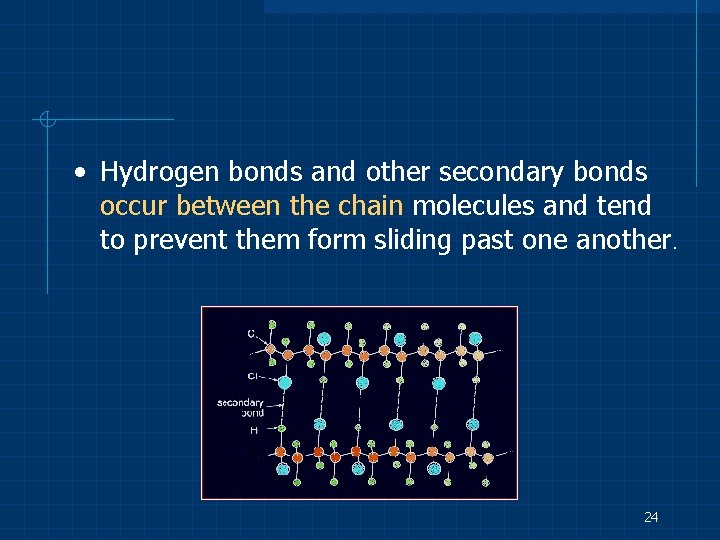
• Hydrogen bonds and other secondary bonds occur between the chain molecules and tend to prevent them form sliding past one another. 24
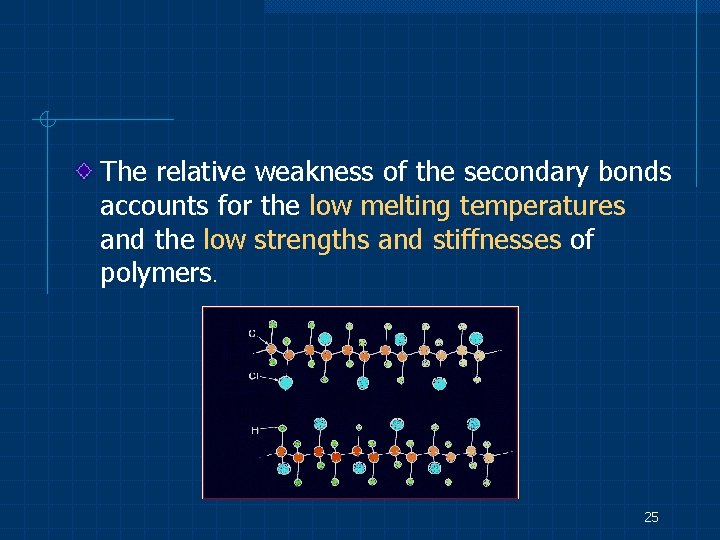
The relative weakness of the secondary bonds accounts for the low melting temperatures and the low strengths and stiffnesses of polymers. 25

Bond Distance Intermolecular Force

Bond distance Factors that control bond distance n n n Diameter of the atoms involved Repulsion and attraction forces “Disturbing” force e. g. mechanical, thermal, or electrical force 27
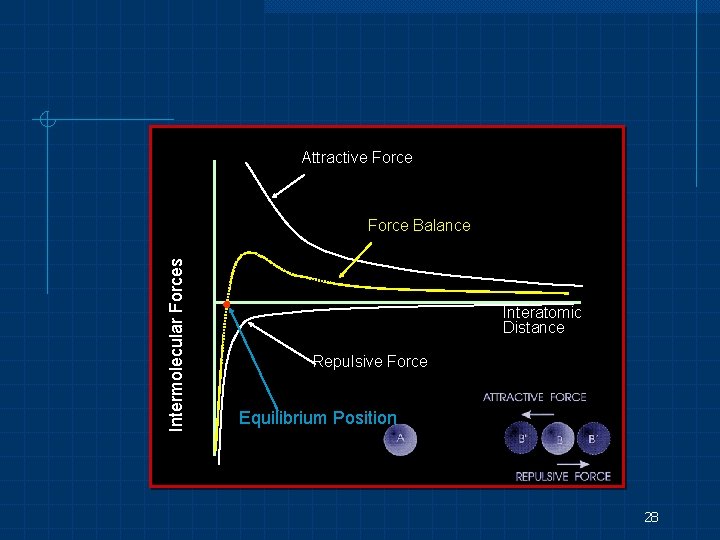
Attractive Force Intermolecular Forces Force Balance Interatomic Distance Repulsive Force Equilibrium Position 28
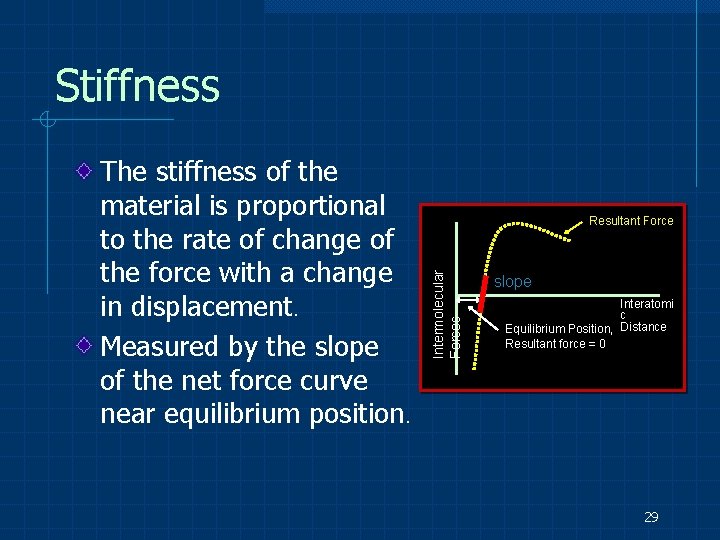
Stiffness Resultant Force Intermolecular Forces The stiffness of the material is proportional to the rate of change of the force with a change in displacement. Measured by the slope of the net force curve near equilibrium position. slope Interatomi c Equilibrium Position, Distance Resultant force = 0 29

Generalizations based on atomic bonding Properties Factors Density Atomic wt, radius, packing Stiffness Interatomic forces Thermal expansion Bond strength and Tm Conductivity Freedom of electron movement 30

Structure Level Crystalline structure Noncrystalline structure (Amorphous)

Materials can be amorphous or crystalline (or mixtures of both, as in some plastics) in solid state. Crystallinity, or the lack of it, often determines the use properties of a material. 32

Metals and ceramics are composed of aggregations of small grains, each of which is an individual crystal. 33

POLYMER Glasses have an amorphous or noncrystalline structure. Polymers are composed of chain-like molecules, which are sometimes arranged in regular arrays in a crystalline manner. Amorphous structure Crystalline structure 34
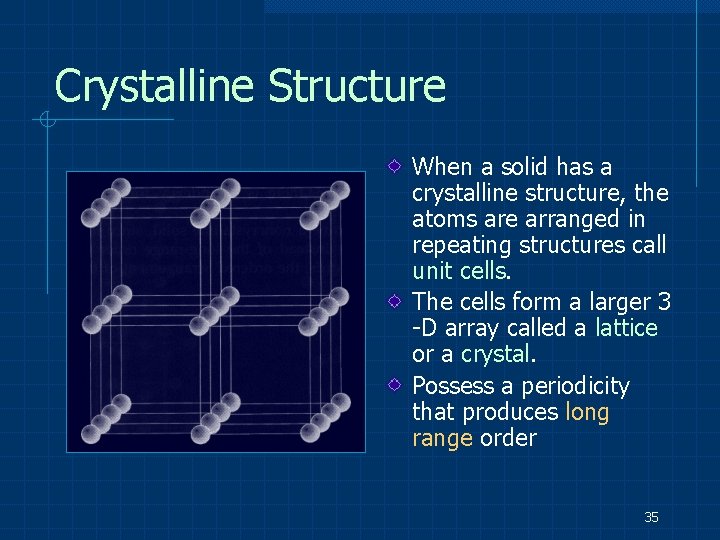
Crystalline Structure When a solid has a crystalline structure, the atoms are arranged in repeating structures call unit cells. The cells form a larger 3 -D array called a lattice or a crystal. Possess a periodicity that produces long range order 35
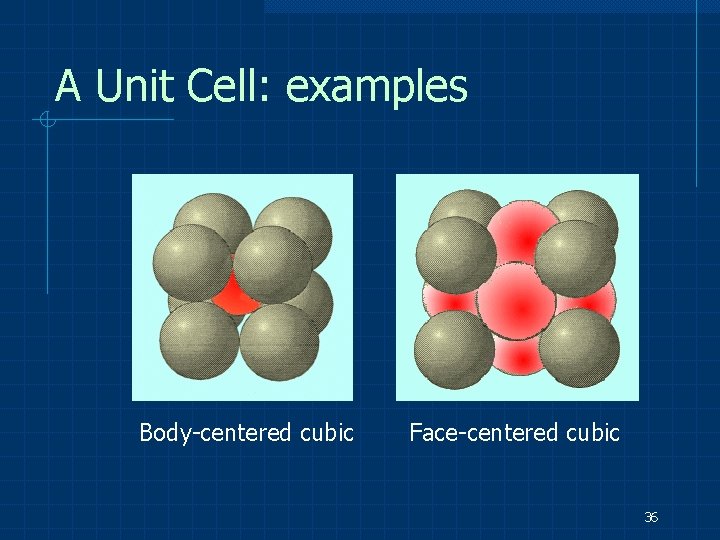
A Unit Cell: examples Body-centered cubic Face-centered cubic 36
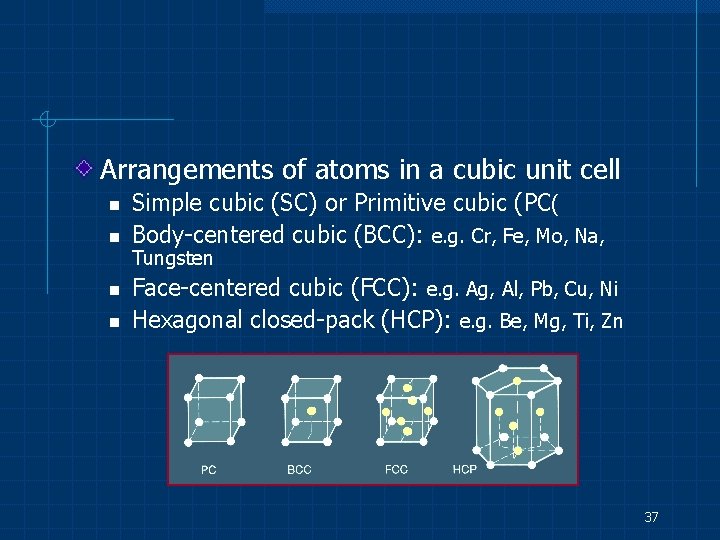
Arrangements of atoms in a cubic unit cell n n Simple cubic (SC) or Primitive cubic (PC( Body-centered cubic (BCC): e. g. Cr, Fe, Mo, Na, Tungsten Face-centered cubic (FCC): e. g. Ag, Al, Pb, Cu, Ni Hexagonal closed-pack (HCP): e. g. Be, Mg, Ti, Zn 37

In the solid state, a crystalline polymer is stiff and rigid with a very high “viscosity”. When heated to the melting point (Tm), the viscosity drops significantly and abruptly to a much lower level. (~ ice, when it melts). 38

Atoms in a space lattice are constantly in vibration about their centers. The average kinetic energy of vibration over the entire crystal is related to the temperature. n n All the atoms do not posses the same amount of energy. If the energy of an atom exceeds the bonding energy, it can move to another position in the lattice. “Diffusion” 39

Noncrystalline Structure e. g. glasses, amorphous polymers Do not have regular, repeating 3 -D arrays of atoms Do not represent low internal energies Do not have a definite Tm but gradually soften as the temperature is raised. The temperature at which there is an abrupt increase in thermal expansion coefficient, indicating increased molecular mobility, is called the glass transition temperature (Tg( 40
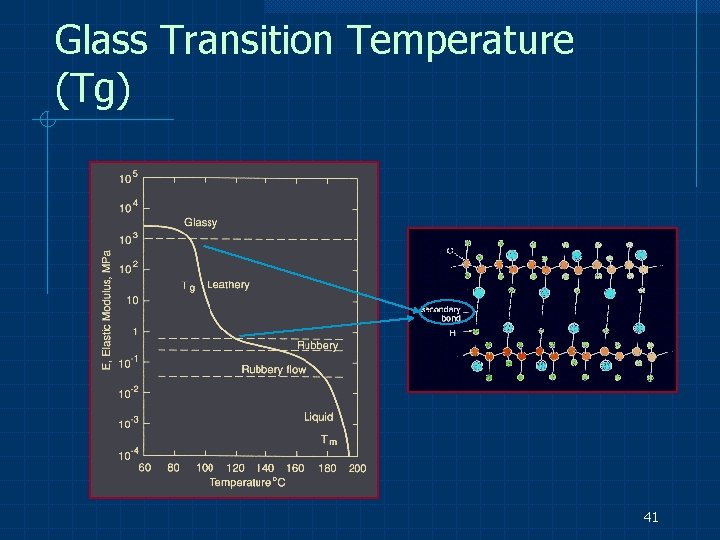
Glass Transition Temperature (Tg) 41

How does the structure effect material properties?

Strong primary bonds are resistant to stretching High Elastic modulus (E( w High E stiff/rigid material Examples: n n n Covalent bonds in diamond: E ~ 1000 GPa Metallic bonds in metals: E ~ 3 x 100 GPa Covalent and secondary bonds in polymers (glassy state): E ~ 3 GPa w 1 Pa (Pascal) = 1 N/m 2 43

Crystalline vs. Amorphous Structure Polymer properties are significantly affected by the degree of crystallinity. Crystallinity tends to n n Increase mechanical properties e. g. tensile strength and hardness Diminish ductility, toughness, and elongation Decrease chemical permeability and solubility Generally optically opaque or translucent (while amorphous polymers optical clarity) 44

The Nature of Metals solids composed of atoms held together by a matrix of electrons Strong Good ductility and malleability due to the ability of the atomic centers to slide against each other into new positions within the same crystal lattice (nature of metallic bond) n Ductility = ability to be drawn into a wire n Malleability = ability to be pounded into a thin sheet Good electrical and thermal conductivity 45

The Nature of Ceramics a combination of one or more metals with a nonmetallic element very rigid covalent or ionic bonds between adjacent atoms Strong Brittle Both ionic and covalent bonds involve very strong bonds between neighboring atoms. Thus crystalline ceramics with this type of bond tend to be very brittle. Loading tends to result in crystal cleavage or separation. Tends to be electrical insulators because the electrons are tied up in bonding and are not free to move throughout the crystal. 46

The Nature of Polymers Composed of long-chain repeating molecules The atoms in the repeating molecule are strongly bonded (usually covalent), and the bonds between molecules may be weaker secondary bonds or similar covalent bonds. Tend to form long chains and the degree of crystallinity depends on chain alignment Usually weaker than most ceramics and metals because the molecular chains are bonded to each other only with secondary bond. 47

The Nature of Composites A combination of two or more materials that has properties that the component materials do not have by themselves. Examples: Wood, Fiberglass (fiberreinforced plastic), Sport equipment, . . etc 48

General Characteristics of Materials 49

Summary Atomic level n n Atomic bondings: Primary, Secondary Bonding distance/ energy Structural level n n Crystalline vs. Noncrystalline structures Tg How does the structure effect the material properties? 50

Relax! 51

End of Structure of Materials 52
- Slides: 52