Structure of Enzymes All enzymes are tertiary globular
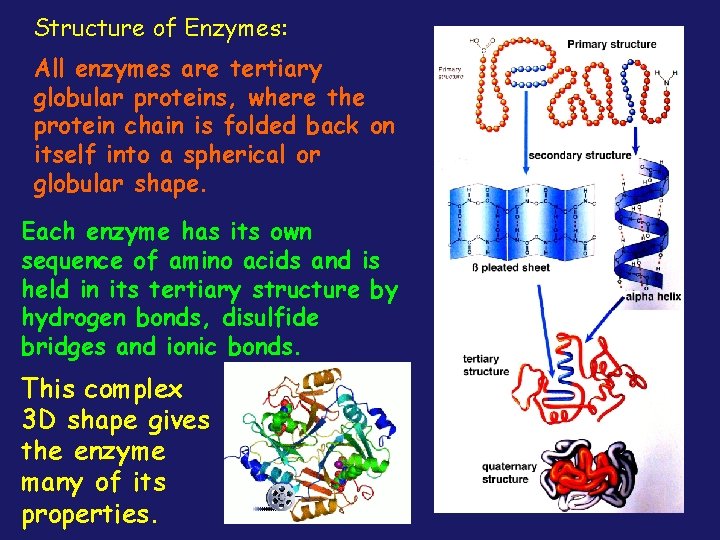
Structure of Enzymes: All enzymes are tertiary globular proteins, where the protein chain is folded back on itself into a spherical or globular shape. Each enzyme has its own sequence of amino acids and is held in its tertiary structure by hydrogen bonds, disulfide bridges and ionic bonds. This complex 3 D shape gives the enzyme many of its properties.

How Enzymes Work: Enzymes are biological catalysts - they speed up the rate of metabolic reactions, without being used up themselves. These reactions can be of two types: 1. Reactions where larger molecules are broken down into smaller molecules. 2. Reactions where small molecules are built up into larger, more complex, molecules. • Enzymes react with another molecule called the SUBSTRATE. • Each enzyme has its own special shape, with an area, the ACTIVE SITE, onto which the substrate molecules bind. • This is known as the lock and key theory.

Models of enzyme action: lock-and-key 3 of 34 © Boardworks Ltd 2008
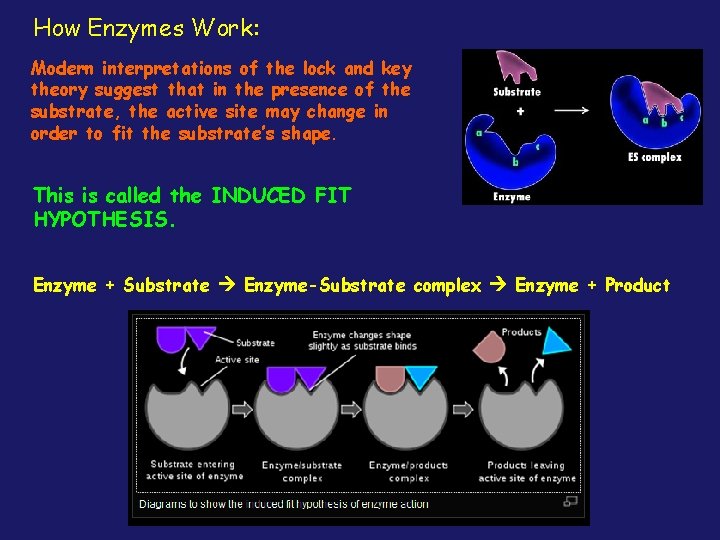
How Enzymes Work: Modern interpretations of the lock and key theory suggest that in the presence of the substrate, the active site may change in order to fit the substrate’s shape. This is called the INDUCED FIT HYPOTHESIS. Enzyme + Substrate Enzyme-Substrate complex Enzyme + Product

Models of enzyme action: induced fit 5 of 34 © Boardworks Ltd 2008
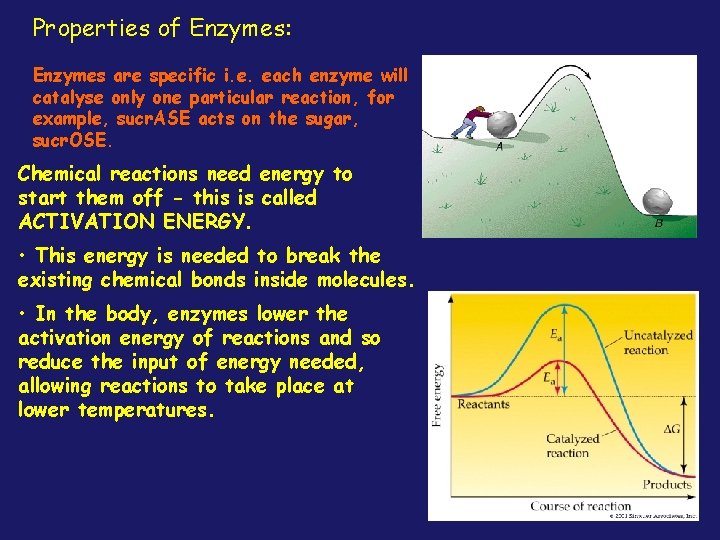
Properties of Enzymes: Enzymes are specific i. e. each enzyme will catalyse only one particular reaction, for example, sucr. ASE acts on the sugar, sucr. OSE. Chemical reactions need energy to Enzymes efficient and have start themare offvery - this is called a high TURNOVER NUMBER. ACTIVATION ENERGY. Thisenergy means isthat theyto can convert • • This needed break the many molecules substrate per existing chemical of bonds inside molecules. unit time, for example, catalase, • which In thebreaks body, down enzymes the lower wastethe activation energy ofperoxide reactions product hydrogen in and the so reduce of energy needed, body, the has input a turnover number of allowing tosecond! take place at severalreactions million per lower temperatures.
Why do enzymes increase the rate? 7 of 34 © Boardworks Ltd 2008

What factors affect enzymes? The rate of an enzyme-controlled reaction is affected by several factors: l temperature l p. H l enzyme concentration l substrate concentration. Each enzyme works best within a range of conditions, and this range is different for each enzyme. Enzymes are also affected by the presence of inhibitors. 8 of 34 © Boardworks Ltd 2008
Effect of temperature on enzymes 9 of 34 © Boardworks Ltd 2008
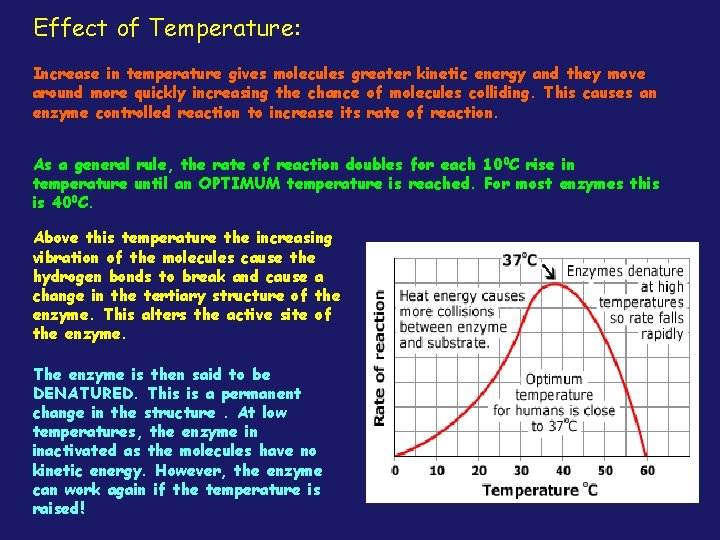
Effect of Temperature: Increase in temperature gives molecules greater kinetic energy and they move around more quickly increasing the chance of molecules colliding. This causes an enzyme controlled reaction to increase its rate of reaction. As a general rule, the rate of reaction doubles for each 100 C rise in temperature until an OPTIMUM temperature is reached. For most enzymes this is 400 C. Above this temperature the increasing vibration of the molecules cause the hydrogen bonds to break and cause a change in the tertiary structure of the enzyme. This alters the active site of the enzyme. The enzyme is then said to be DENATURED. This is a permanent change in the structure. At low temperatures, the enzyme in inactivated as the molecules have no kinetic energy. However, the enzyme can work again if the temperature is raised!

Effect of p. H on enzymes 11 of 34 © Boardworks Ltd 2008

Effect of p. H: The rate of an enzyme catalysed reaction will vary with changes in p. H. Enzymes have a narrow optimum range and small changes in p. H can affect the rate of reaction without affecting the structure of the enzyme. Small changes in p. H outside the optimum can cause small reversible changes in enzyme structure and results in inactivation. Extremes of p. H can denature the enzyme. The charges on the amino acid side-chains of the enzyme’s active site are affected by free hydrogen ions or hydroxyl ions. In the formation of an enzyme substrate complex the charge on the active site must match those of the substrate. E. g. if the active site has too many H+ ions, the active site and the substrate may both BUFFERS maintain a constant p. H. When a have the same charge and the enzyme willbuffer is used in an experiment the p. H repel the substrate. changes little when a small quantity of acid or alkali is added. At extremes of p. H, the hydrogen bonding is affected and the 3 D shape of the enzyme is altered and so is the shape of the active site.

Effect of substrate concentration on rate 13 of 34 © Boardworks Ltd 2008
Effect of enzyme concentration on rate 14 of 34 © Boardworks Ltd 2008

Concentration: SUBSTRATE CONCENTRATION – the rate of an enzyme catalysed reaction will vary with changes in substrate concentrations. If the amount of enzyme is constant the rate of reaction will increase as the substrate increases. But there must come a point when all the enzyme’s active sites are working to full capacity i. e. all the active sites are filled. ENZYME CONCENTRATION – the rate of an enzyme catalysed reaction will vary the changes in enzyme concentration. Increasing the enzyme concentration will increase the rate of reaction.

What are enzyme inhibitors? Substances can interfere with enzyme activity are called inhibitors. They can be classed in two ways, depending on their mode of action: l Inhibitors can be either competitive (active site directed) or noncompetitive (non-active site directed), depending on whether they compete with the substrate for binding at the active site or not. l Inhibitors can be either reversible or irreversible, depending on whether their inhibitory effect on the enzyme is permanent or not. 16 of 34 © Boardworks Ltd 2008

Enzyme inhibitors: mode of action 17 of 34 © Boardworks Ltd 2008

Effect of inhibitors on enzymes 18 of 34 © Boardworks Ltd 2008
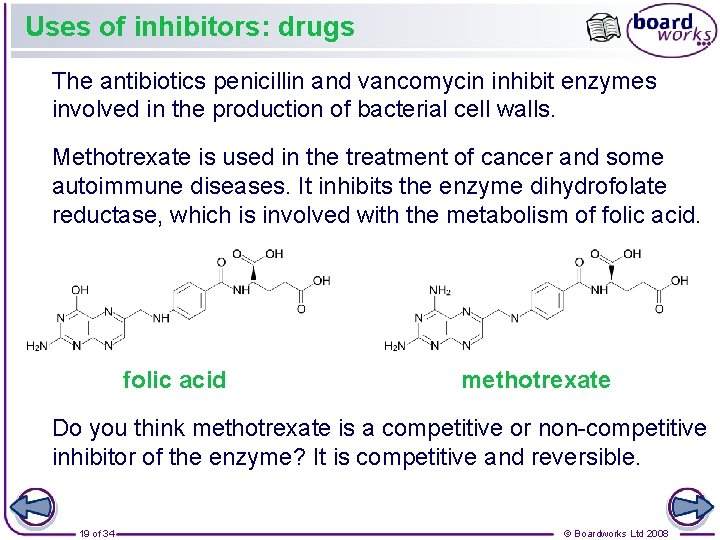
Uses of inhibitors: drugs The antibiotics penicillin and vancomycin inhibit enzymes involved in the production of bacterial cell walls. Methotrexate is used in the treatment of cancer and some autoimmune diseases. It inhibits the enzyme dihydrofolate reductase, which is involved with the metabolism of folic acid methotrexate Do you think methotrexate is a competitive or non-competitive inhibitor of the enzyme? It is competitive and reversible. 19 of 34 © Boardworks Ltd 2008

Test yourself Try to answer the graded questions. 20 minutes!

E – Condition for best/highest rate of enzyme activity/most favourable condition (1) D – Increased temperature increases rate of reaction (1) Until optimum/around 40°C (1) Above this, rate of reaction decreases (1) D – Competitive – competes with substrate for active site (1) Non-competitive – does not compete with substrate for active site (1)

C – Competitive – has a complementary shape to active site/similar shape to substrate so competes with substrate for the active site (1) Non-competitive – binds with a part of the enzyme other than the active site, causing a change in the shape of the enzyme so the substrate can no longer bind (1) C – (If the p. H is too high, there are too many OH- ions; if the p. H is too low, there are too many H+ ions. ) These ions interfere with the hydrogen/ionic bonds in the enzyme (1) So the tertiary structure will change (1) OR the active site will have a different charge so repel the substrate (1)

B – Competitive has a similar shape to the substrate (noncompetitive doesn’t) (1) C – binds with active site/NC – binds to an area that isn’t the active site (1) C – competes with the substrate for the active site/NC – doesn’t (1) B – As substrate concentration increases, more of the active sites of the enzymes will be occupied at any one time (1) Eventually, all of the active sites will be occupied so the rate of reaction can’t further increase (1)

A – Inhibitors may only bind with weak/H-bonds which are easily broken (1) Once an enzyme is denatured, the bonds (or named bond) supporting its tertiary structure will no longer be present (1) A – The inhibitor has a similar shape to the substrate/complementary shape to the active site (1) Binds with the active site, preventing the normal activity of the enzyme (1) A – With a C inhibitor, active sites are available to be used, so the more substrate present, the greater the proportion of active sites it will fill (1) With a NC inhibitor, the active site changes shape, so no substrate molecules can bind with the affected enzymes (1)
- Slides: 24