Structure of Atoms Atom Basic unit of matter
Structure of Atoms
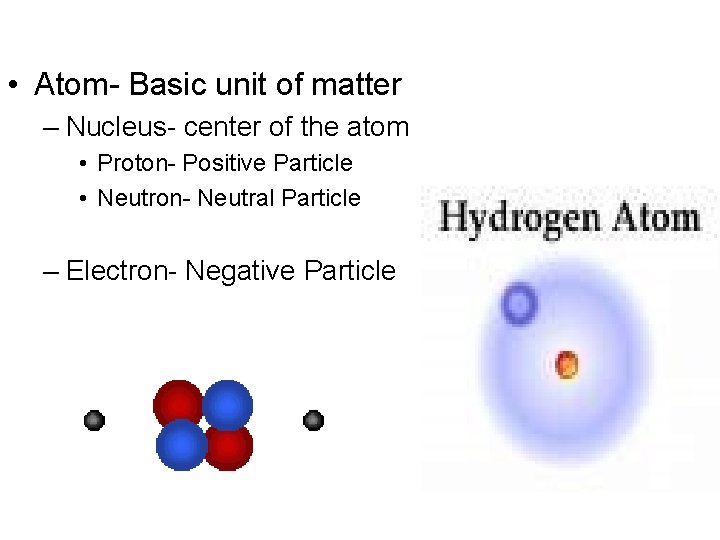
• Atom- Basic unit of matter – Nucleus- center of the atom • Proton- Positive Particle • Neutron- Neutral Particle – Electron- Negative Particle
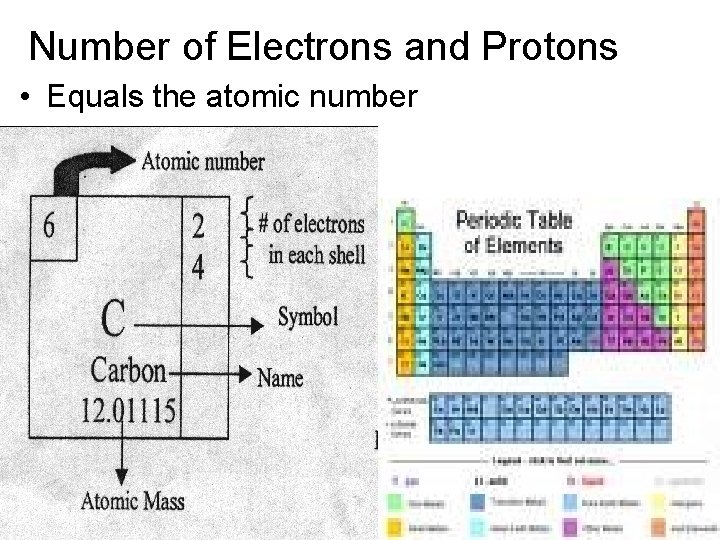
Number of Electrons and Protons • Equals the atomic number

Number of Neutrons • Equals the atomic mass minus the atomic number 12. 0115 – 6 = 6 neutrons
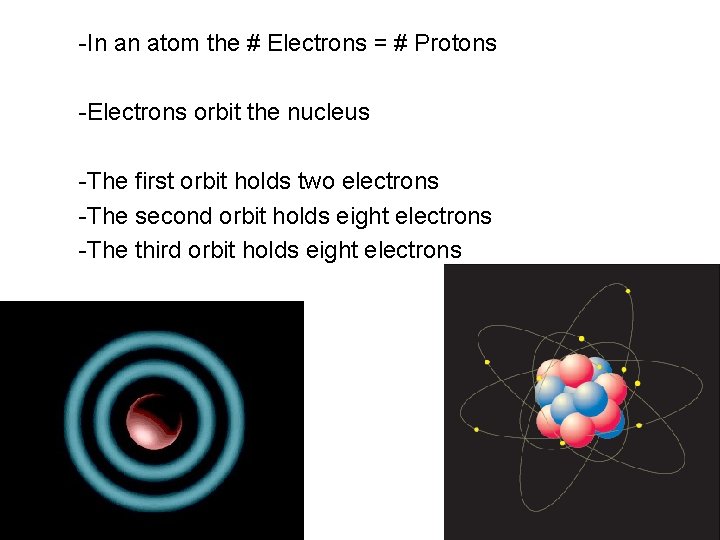
-In an atom the # Electrons = # Protons -Electrons orbit the nucleus -The first orbit holds two electrons -The second orbit holds eight electrons -The third orbit holds eight electrons
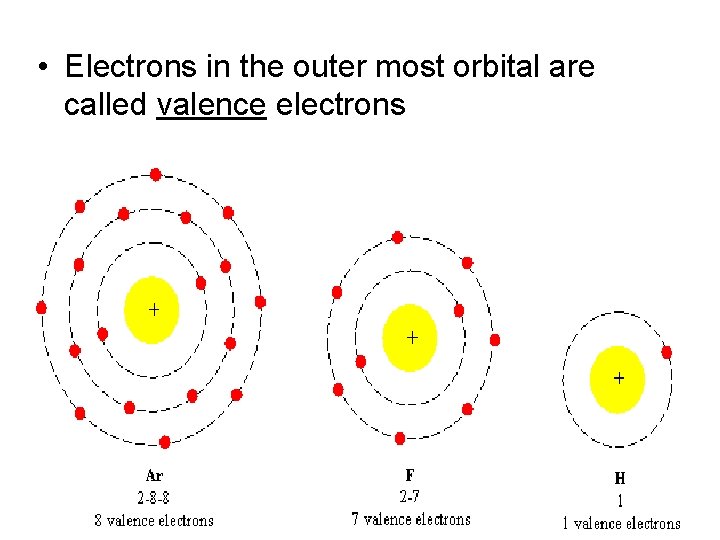
• Electrons in the outer most orbital are called valence electrons
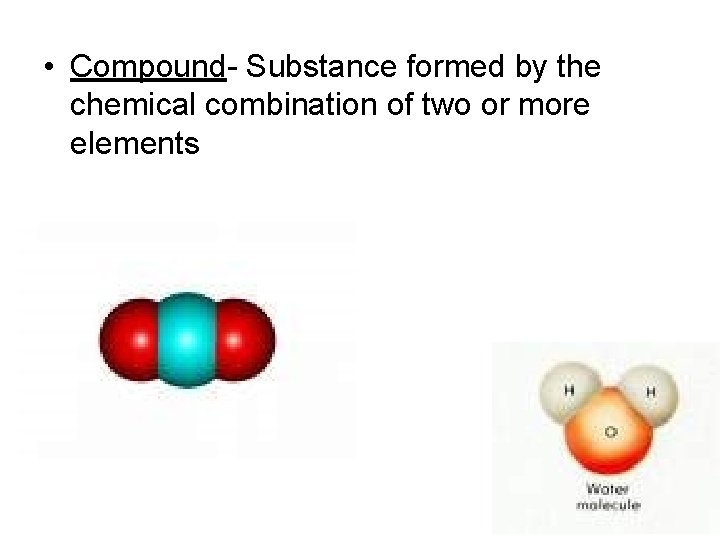
• Compound- Substance formed by the chemical combination of two or more elements
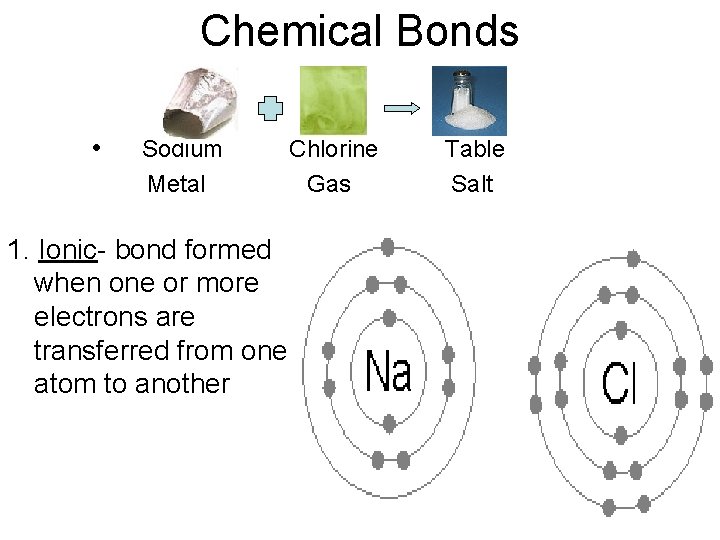
Chemical Bonds • Sodium Metal 1. Ionic- bond formed when one or more electrons are transferred from one atom to another Chlorine Gas Table Salt
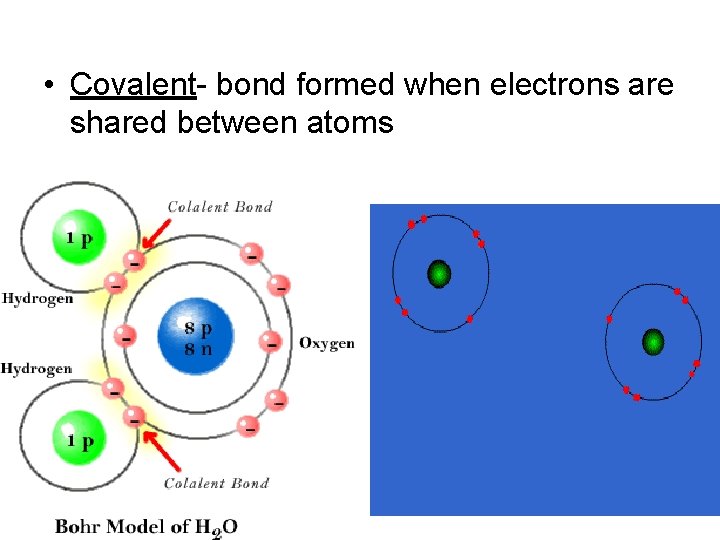
• Covalent- bond formed when electrons are shared between atoms

• Van der Waals Forces- slight attraction between nearby atoms
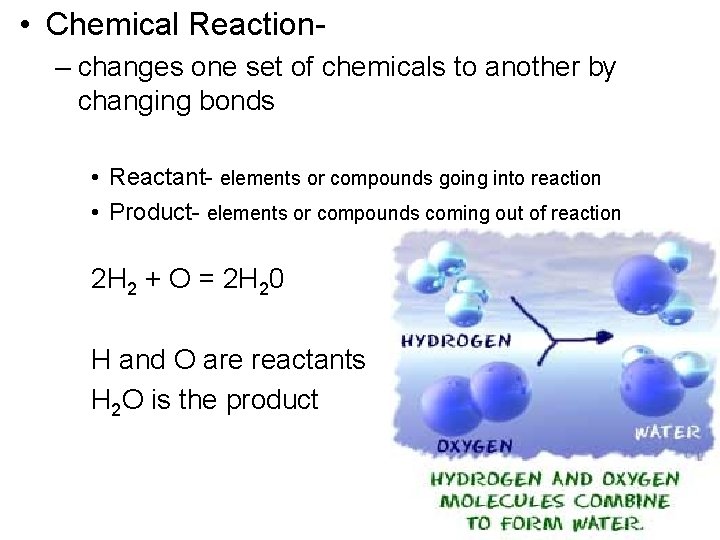
• Chemical Reaction– changes one set of chemicals to another by changing bonds • Reactant- elements or compounds going into reaction • Product- elements or compounds coming out of reaction 2 H 2 + O = 2 H 20 H and O are reactants H 2 O is the product

Reactions can be Reversible CO 2 + H 2 O H 2 CO 3 CO 2 + H 2 O • How many atoms of oxygen are in the products of the second reaction? • How many molecules of carbon are in the products of the first reaction?
- Slides: 12