Structure and Synthesis of Alcohols Structure of Alcohols









- Slides: 14

Structure and Synthesis of Alcohols

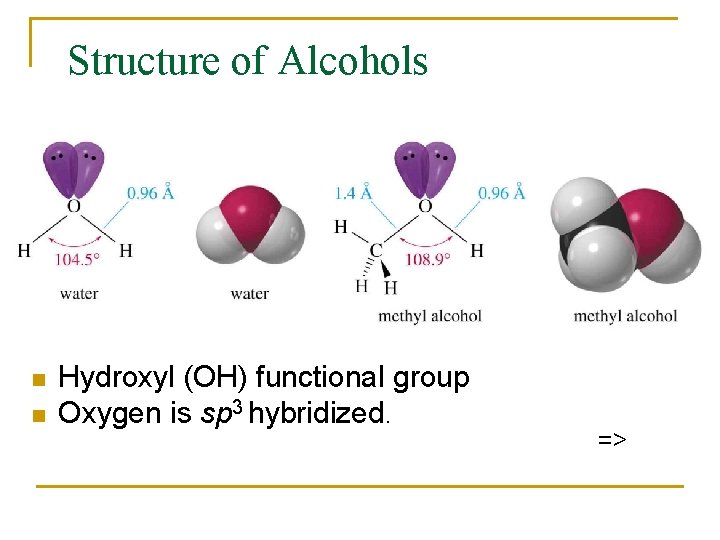
Structure of Alcohols n n Hydroxyl (OH) functional group Oxygen is sp 3 hybridized. =>

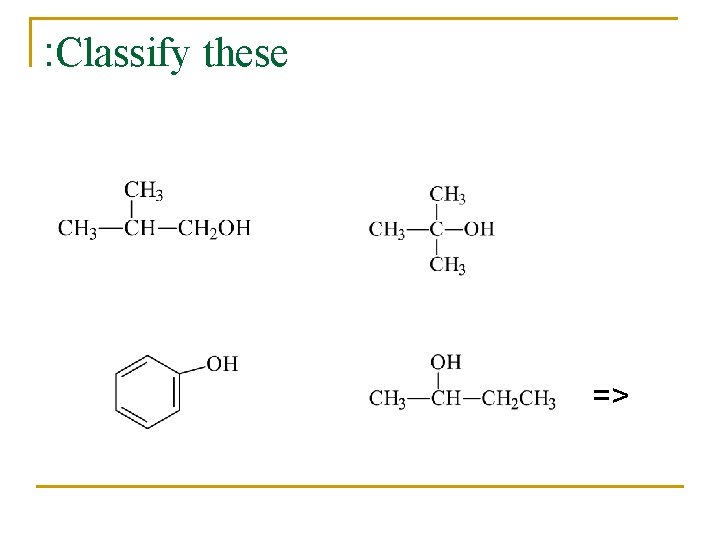
Classification n n Primary: carbon with –OH is bonded to one other carbon. Secondary: carbon with –OH is bonded to two other carbons. Tertiary: carbon with –OH is bonded to three other carbons. Aromatic (phenol): -OH is bonded to a benzene ring. =>

: Classify these =>

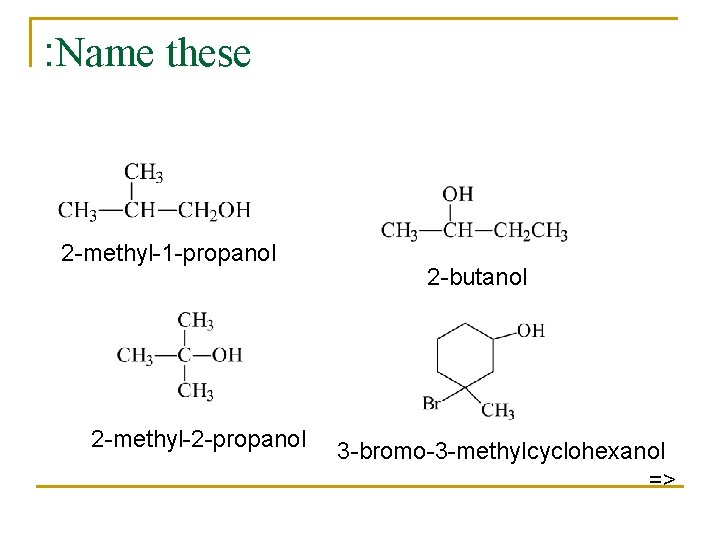
IUPAC Nomenclature n n Find the longest carbon chain containing the carbon with the -OH group. Drop the -e from the alkane name, add -ol. Number the chain, starting from the end closest to the -OH group. Number and name all substituents. =>

: Name these 2 -methyl-1 -propanol 2 -methyl-2 -propanol 2 -butanol 3 -bromo-3 -methylcyclohexanol =>

Unsaturated Alcohols n n Hydroxyl group takes precedence. Assign that carbon the lowest number. Use alkene or alkyne name. 4 -penten-2 -ol (old) pent-4 -ene-2 -ol (1997 revision of IUPAC rules) =>

Naming Priority n n n Acids Esters Aldehydes Ketones Alcohols Amines n n n Alkenes Alkynes Alkanes Ethers Halides =>

Hydroxy Substituent n n When -OH is part of a higher priority class of compound, it is named as hydroxy. Example: also known as GHB 4 -hydroxybutanoic acid =>

Common Names n n n Alcohol can be named as alkyl alcohol. Useful only for small alkyl groups. Examples: isobutyl alcohol sec-butyl alcohol =>

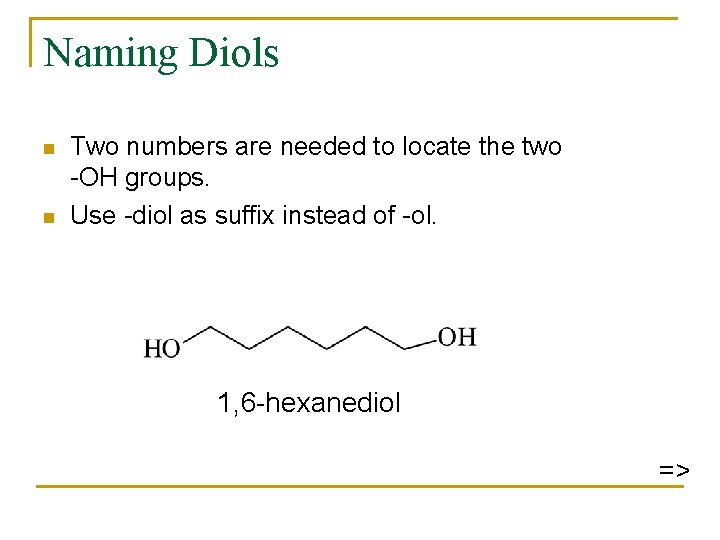
Naming Diols n n Two numbers are needed to locate the two -OH groups. Use -diol as suffix instead of -ol. 1, 6 -hexanediol =>

Glycols n n 1, 2 diols (vicinal diols) are called glycols. Common names for glycols use the name of the alkene from which they were made. 1, 2 -ethanediol 1, 2 -propanediol ethylene glycol propylene glycol =>

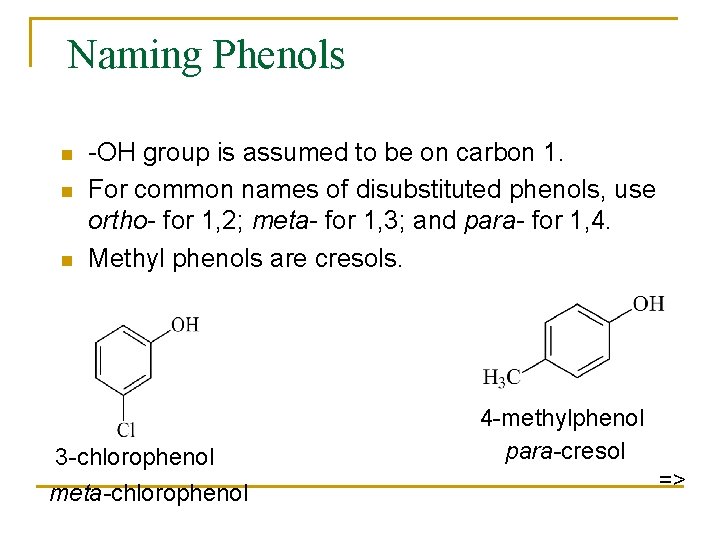
Naming Phenols n n n -OH group is assumed to be on carbon 1. For common names of disubstituted phenols, use ortho- for 1, 2; meta- for 1, 3; and para- for 1, 4. Methyl phenols are cresols. 3 -chlorophenol meta-chlorophenol 4 -methylphenol para-cresol =>

Physical Properties n n Unusually high boiling points due to hydrogen bonding between molecules. Small alcohols are miscible in water, but solubility decreases as the size of the alkyl group increases. =>