Structure and Functions of National Health Laboratory Quality

Structure and Functions of National Health Laboratory Quality Assurance and Training Centre (NHLQATC) Muchunguzi V

Outline • Background • Vision and Mission • Structure, staff and Organogram • Surveillance and response to epidemics • Functions • Collaborating institutions 2

Background – 1/2 • NHLQATC handed over to MOH in 2008 by CDC (PEPFAR Fund) • Currently operating under diagnostic section • Accredited by SADCAS ISO 15189 (MED 001) 2014 • Selected to be ASLM regional collaborating center in 2012 • Recommended by WHO for prequalification of diagnostics 2016 • Mandated by the Ministry to oversee Public Health responsibilities 3

Vision and Mission • Vision – To become a centre of excellence for reliable and sustainable quality Public Health laboratory services that are effective, accessible and affordable to all • Mission – To deliver quality Public Health laboratory services for the achievement of better health for all 4

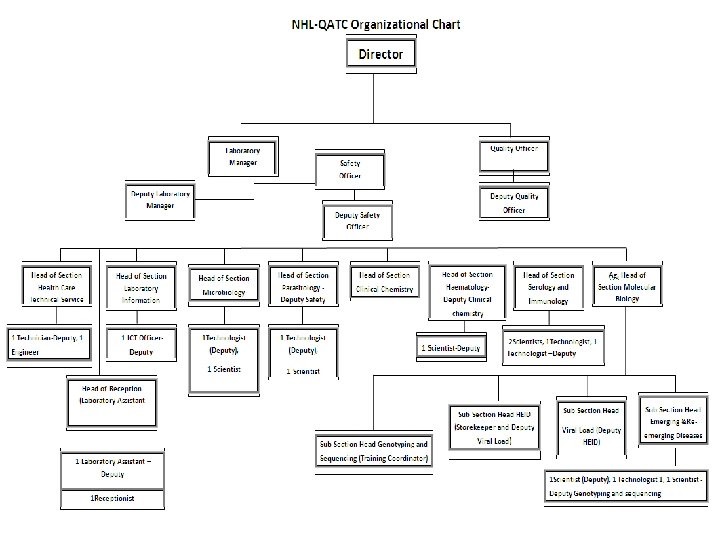
Structure and Staff • Management Structure – Director – Manager – Deputy Manager – Quality officer – Deputy Quality Officer – Safety officer – Deputy Safety Officer – Section Heads(8 sections and 3 subsections – Staff required 48 – Currently available 33 5
6

Functional Laboratory sections • • Health care Technical services Laboratory information(data management) Microbiology Parasitology Clinical Chemistry Hematology Serology and Immunology Molecular Biology – Emerging and Re emerging diseases – HEID and Viral Load – Sequencing and Genotyping 7

Surveillance and Notification system • Works closely with Regional Laboratories on confirmation of outbreaks • Reports surveillance and outbreak update to Emergency Operation Centre (EOC) • Communicate with epidemiology section on surveillance and outbreak issues • Coordinates specimen referral and quality assurance 8

Functions of NHLQATC – 1/4 • Serves as a central reference laboratory for HIV related and other clinical testing • Provides technical assistance and leadership in assuring quality systems (Prepare QC and distribute to end users) • Coordinates External Quality Assessment schemes • Collaborate with other reference laboratories in clinical issues 9

Functions of NHLQATC – 2/4 • Training laboratory personnel in collaboration with other training institutions • Evaluating various Methods and Technologies • Performs Post market surveillance and lot testing • Performs WHO prequalification of diagnostic • Testing clinical related research samples • Promotes operational research in medical health laboratory services 10

Functions of NHLQATC – 3/4 • Coordinates networking of Health laboratory IDSR activities • Respond promptly to epidemics, outbreaks and emergency situations • Collaborate with other reference laboratories in public health issues • Promote and coordinate effective referral system of specimens within and outside the country 11

Functions of NHLQATC – 4/4 • Test food and water samples for microbial contaminates • Identify non communicable diseases by accurately performing related tests • Perform Molecular testing for identification of epidemic causative agents • Coordinate National and international Laboratory Networking 12

NHLQATC Product scope – 1/2 • Proficiency tests (PT) for (over 6000 sites): – CD 4, HIV rapid tests using Dried tube specimens, Parasitology (malaria), microbiology HIV early Infant Diagnosis and clinical chemistry. • Reference samples material for more than 45 bacteria species, e. g. E. Coli, Klebsella, and Pseudomonas, salmonella, Shigella, enterobactor, staphylococcus, Neisseria, streptococcus and cholera 13

NHLQATC Product scope – 2/2 • Training manuals: • Guidelines: National Standard for Health Laboratory Services, National Quality Assurance Framework, Health Laboratory Waste management, National health Laboratory Standard Supplies List (NHLSSL), Antimicrobial resistance guideline (AMR). . • Liquid Nitrogen for preservation of biological material 14

NHLQATC Service scope – 1/2 • Laboratory testing: – Specialized testing e. g. Viral Load test, HIV DNA PCR – Reference testing: Dengue fever, Chikungunya, RVF, Influenza virus, ARV drug resistance testing, HIV and Antimicrobial resistance testing • Training programs • Mentorship program to clinical laboratories • Surveillance for emerging and re-emerging diseases of public health importance – Conducts Sentinel Laboratory Based Influenza surveillance – Coordination of specimen referral within and outside the country 15

NHLQATC Service scope-2/2 • Evaluation of new laboratory diagnostic technologies imported in the country • Post Market Surveillance including Batch release and Lot testing of diagnostics before sending to end users • WHO Prequalification for diagnostics 16

NHLQATC Market Scope • Clinical Laboratories – private sectors, public and international • Public Health Laboratories • Lab personnel • Regulatory authorities, institutions and programs (MSD, PHLB, TFDA, NACP) • Development partners 17

Geographic scope • Tanzania Mainland • Zanzibar • East Africa (EAPHLNP, EAC) • Provision of Proficient Testing Panels to SADCC Countries • CDC Atlanta • Negotiating for provision of PT for malaria to Caribbean Countries 18

Projects through NHLQATC • Lab support project – Supporting staff and quality activities • EAPHLNP – Supporting capacity building, equipment maintenance, supplies and construction and equipping of the NPHL • GHSA – Supporting staff, and respond to emerging and re emerging threats • Bushmeet testing – Pathogens in Bushmeet • HIV Population Impact Assessment 19

• • • International Collaborating institutions WHO CDC WALTER REEDS ASLM ASM ICAP GIZ APHL AFENET CLSI Kf. W 20
- Slides: 20