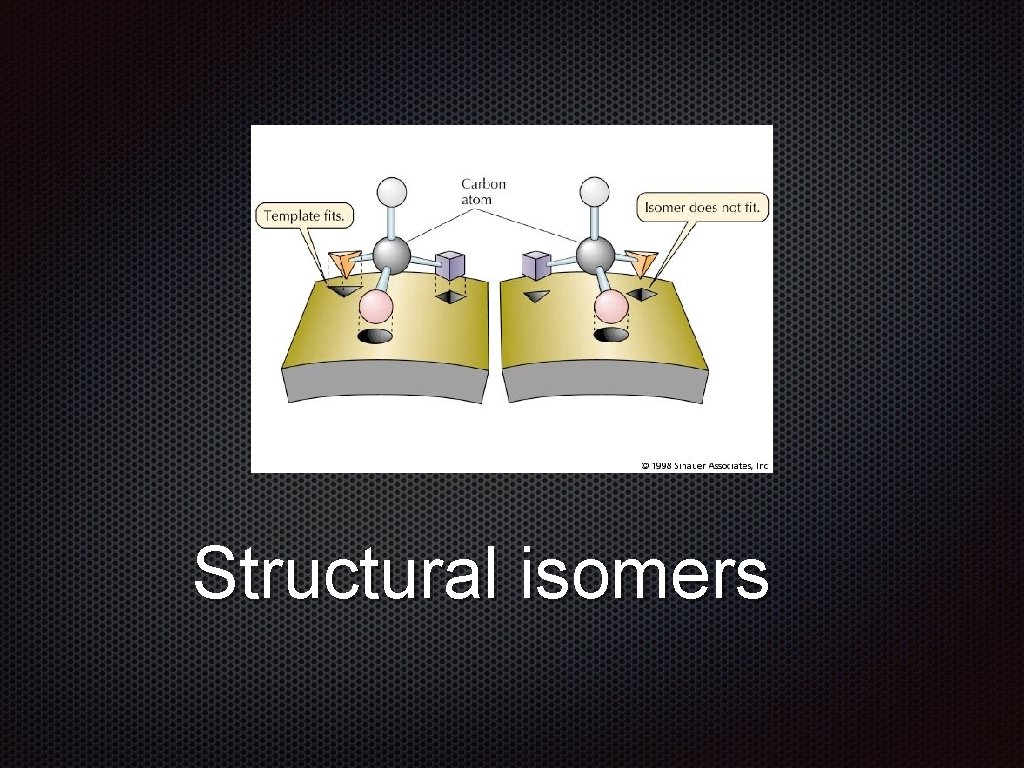
Structural isomers Structural Isomers Molecules with the same



- Slides: 15

Structural isomers

Structural Isomers Molecules with the same molecular formula but with a different arrangement of atoms are called isomers. The differences in the shape of of isomers leads to to adifferences difference in in their physical and chemical properties.

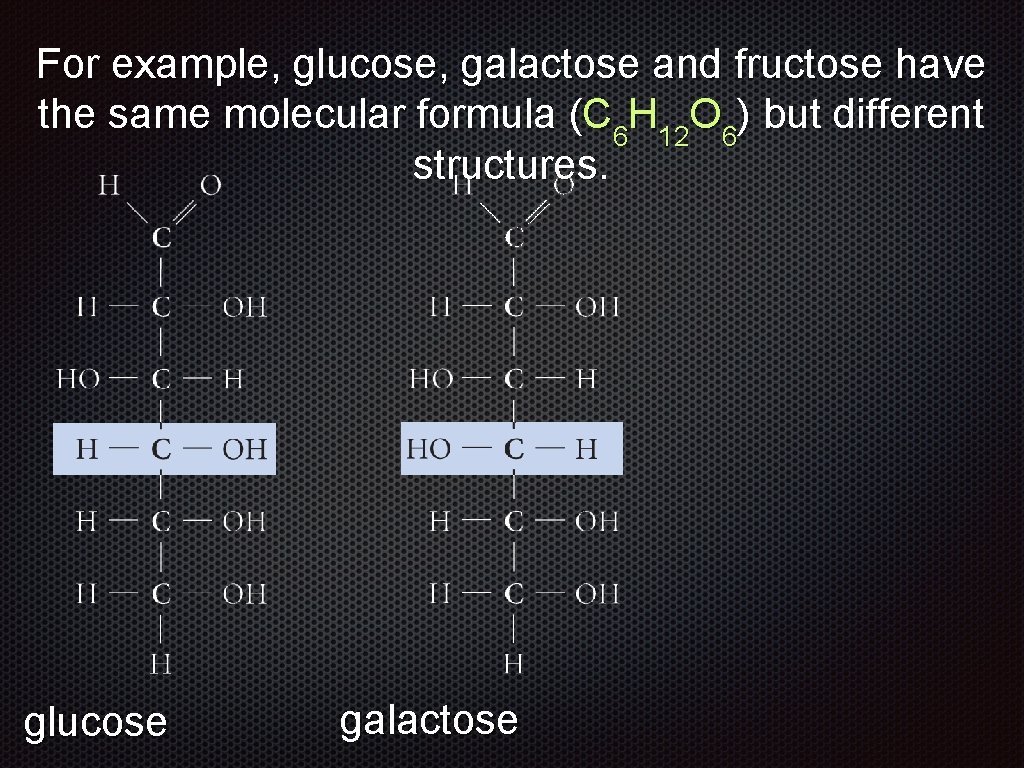
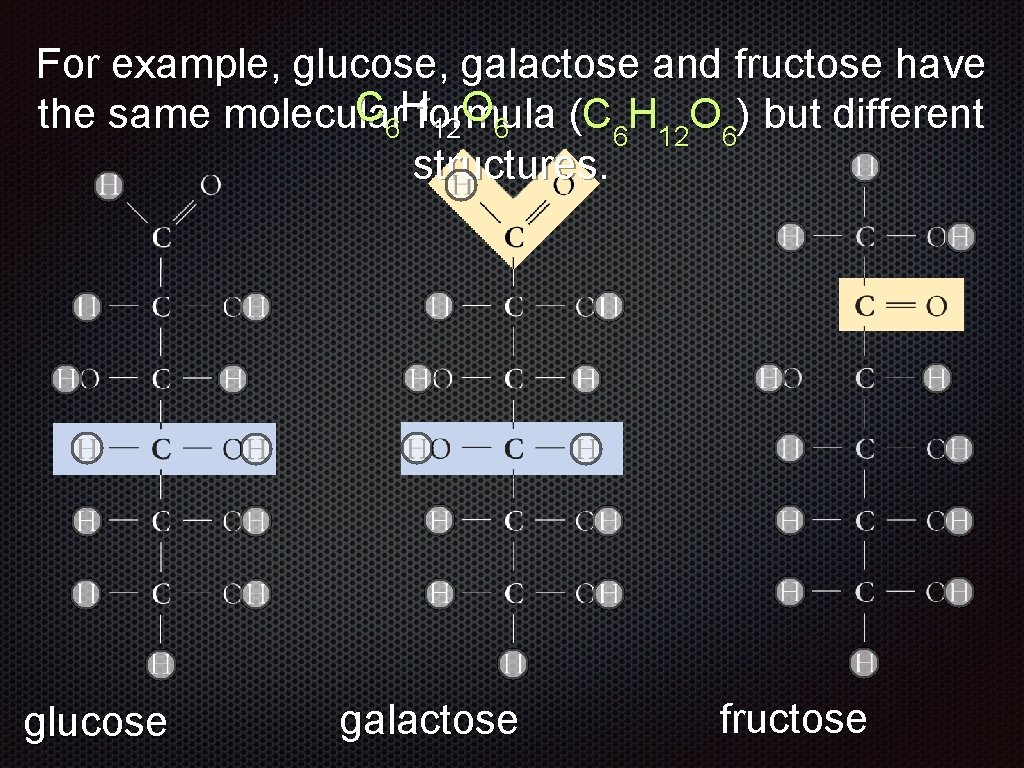
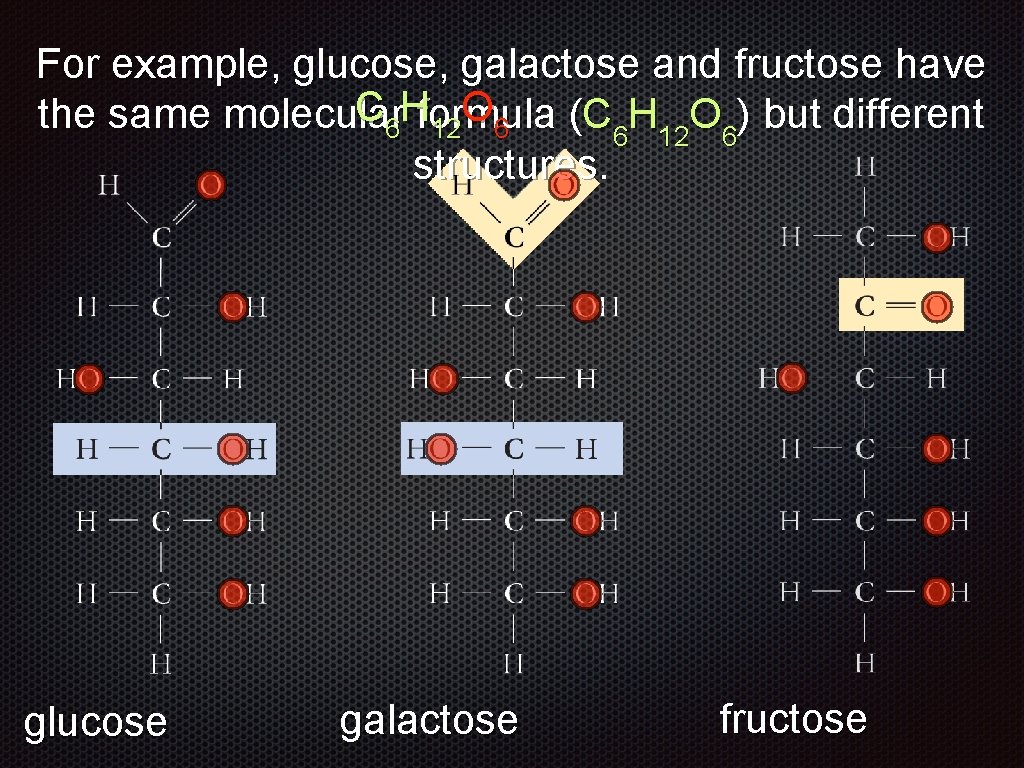
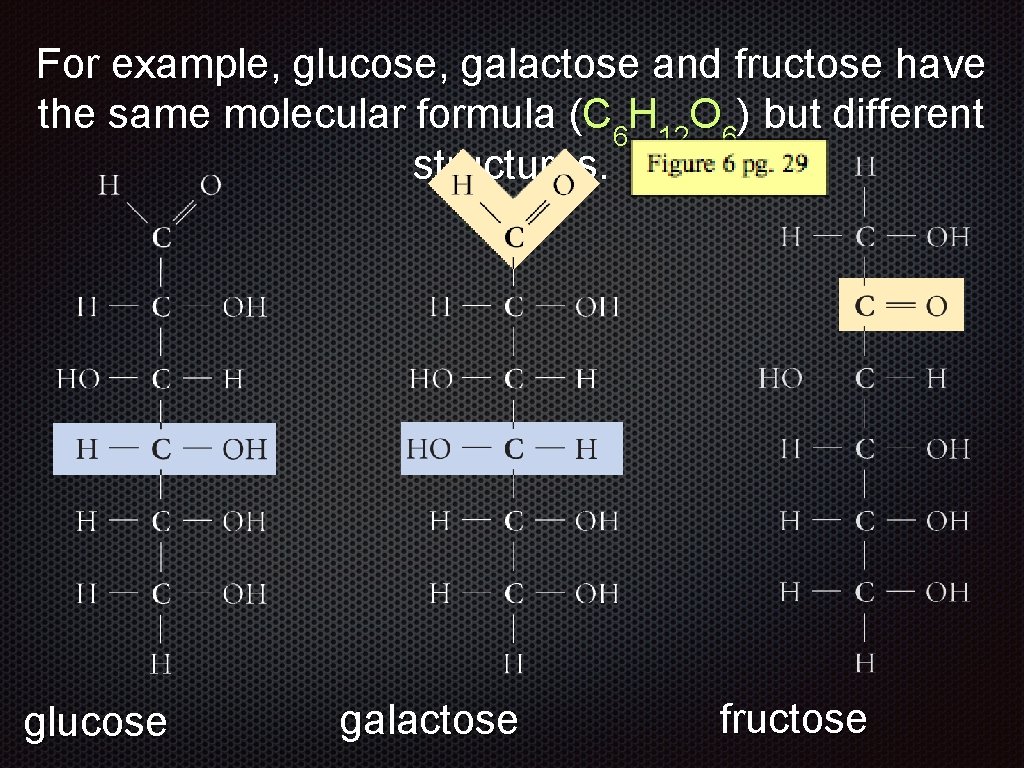
For example, glucose, galactose and fructose have the same molecular formula (C 6 H 12 O 6) but different structures. glucose galactose

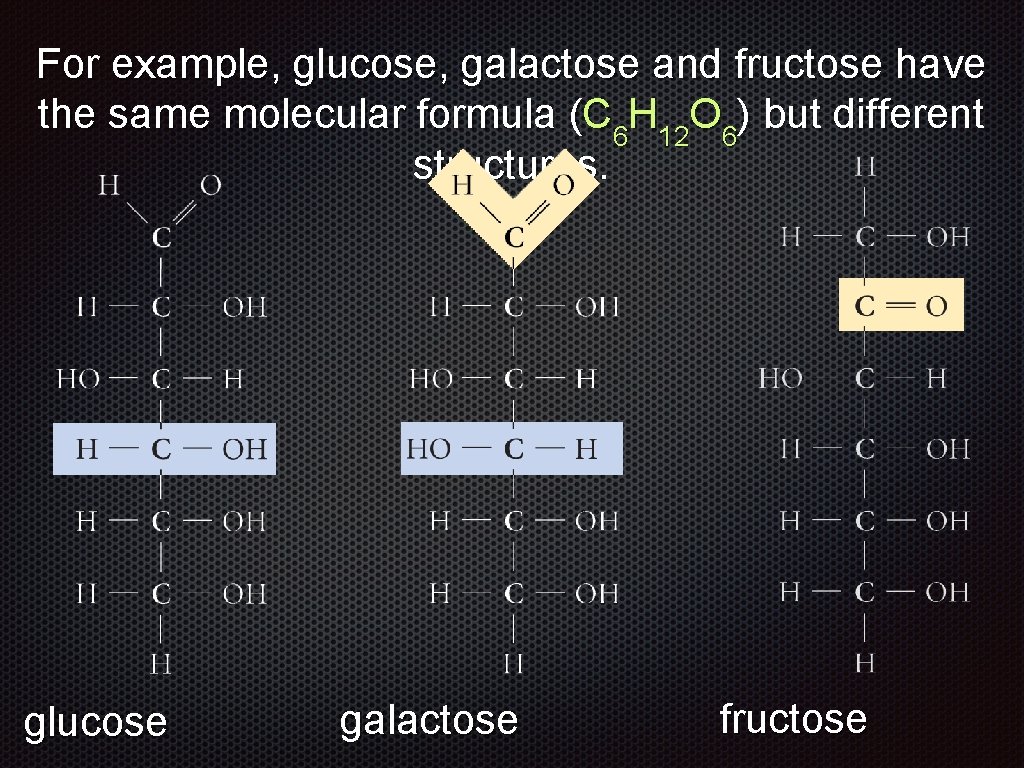
For example, glucose, galactose and fructose have the same molecular formula (C 6 H 12 O 6) but different structures. glucose galactose fructose

For example, glucose, galactose and fructose have C 6 Hformula O the same molecular (C 6 H 12 O 6) but different 12 6 structures. glucose galactose fructose

For example, glucose, galactose and fructose have C 6 Hformula O the same molecular (C 6 H 12 O 6) but different 12 6 structures. glucose galactose fructose

For example, glucose, galactose and fructose have C 6 Hformula O the same molecular (C 6 H 12 O 6) but different 12 6 structures. glucose galactose fructose

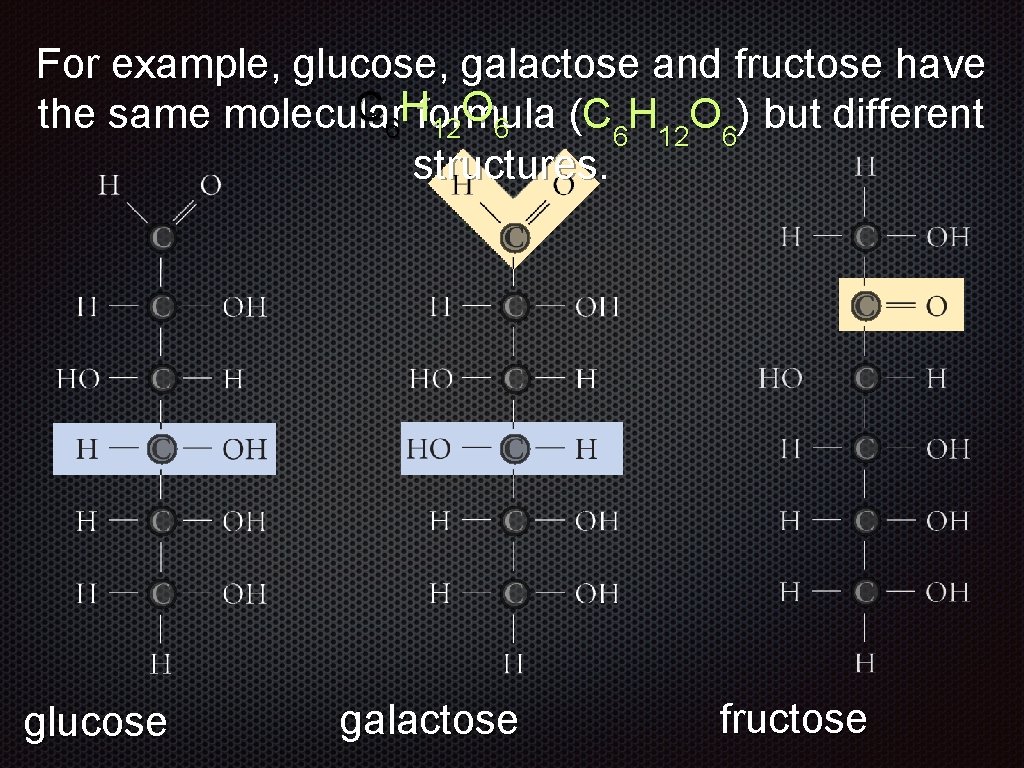
For example, glucose, galactose and fructose have the same molecular formula (C 6 H 12 O 6) but different structures. glucose galactose fructose

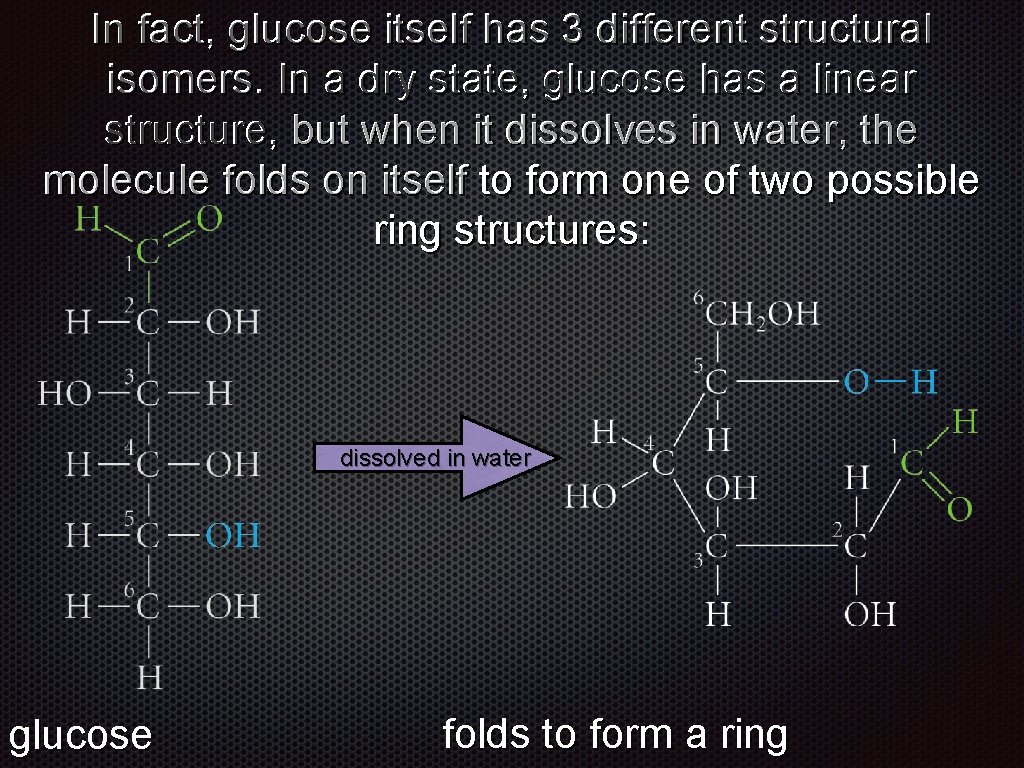
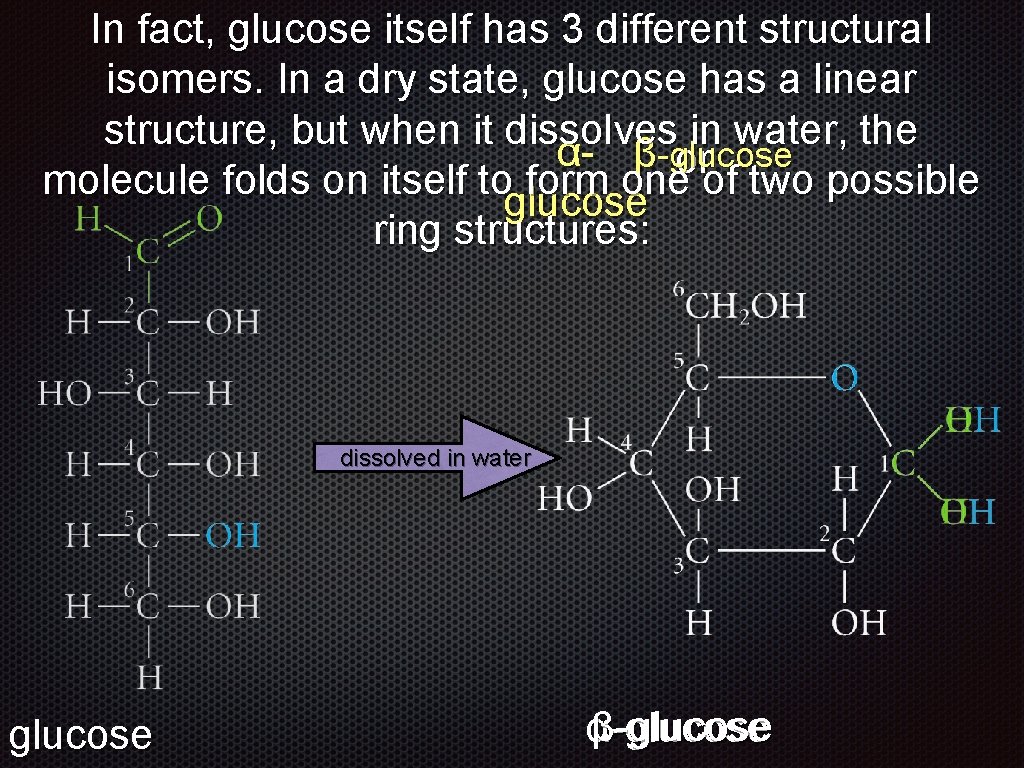
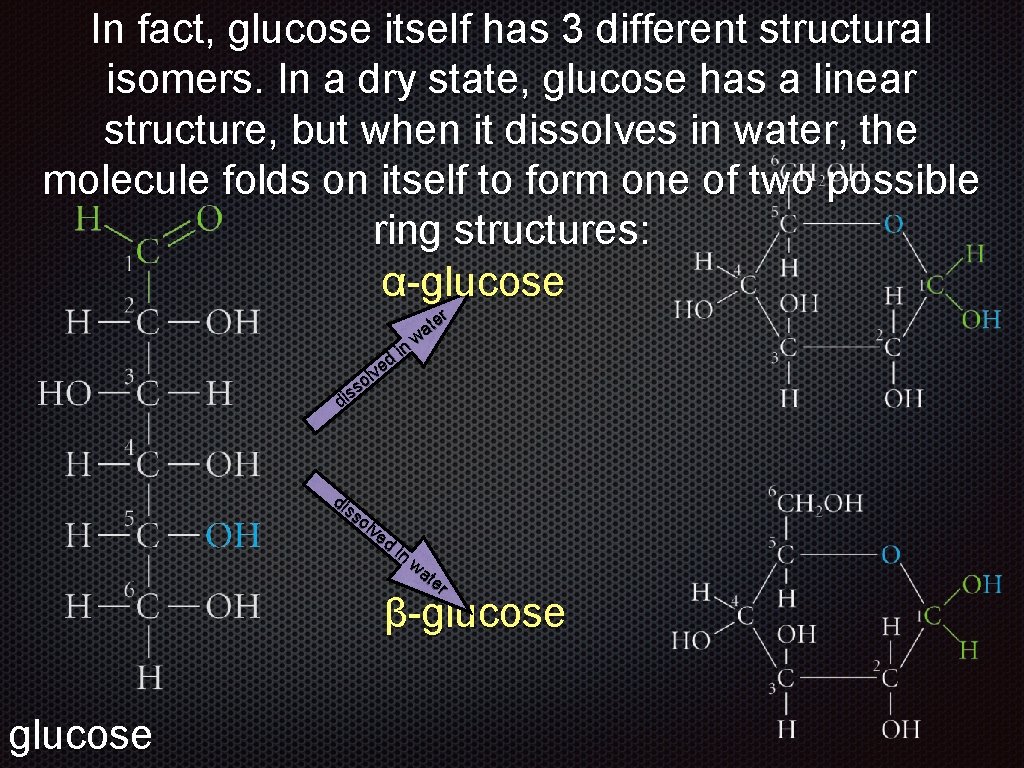
In fact, glucose itself has 3 different structural isomers. In a dry state, glucose has a linear structure, but when it dissolves in water, the molecule folds on itself to form one of two possible ring structures: dissolved in water glucose folds to form a ring

In fact, glucose itself has 3 different structural isomers. In a dry state, glucose has a linear structure, but when it dissolves in water, the α- β-glucose or molecule folds on itself to form one of two possible glucose ring structures: dissolved in water glucose αβ-glucose

In fact, glucose itself has 3 different structural isomers. In a dry state, glucose has a linear structure, but when it dissolves in water, the molecule folds on itself to form one of two possible ring structures: α-glucose er t wa in d lve o ss i d di ss ol ve d in wa te r β-glucose

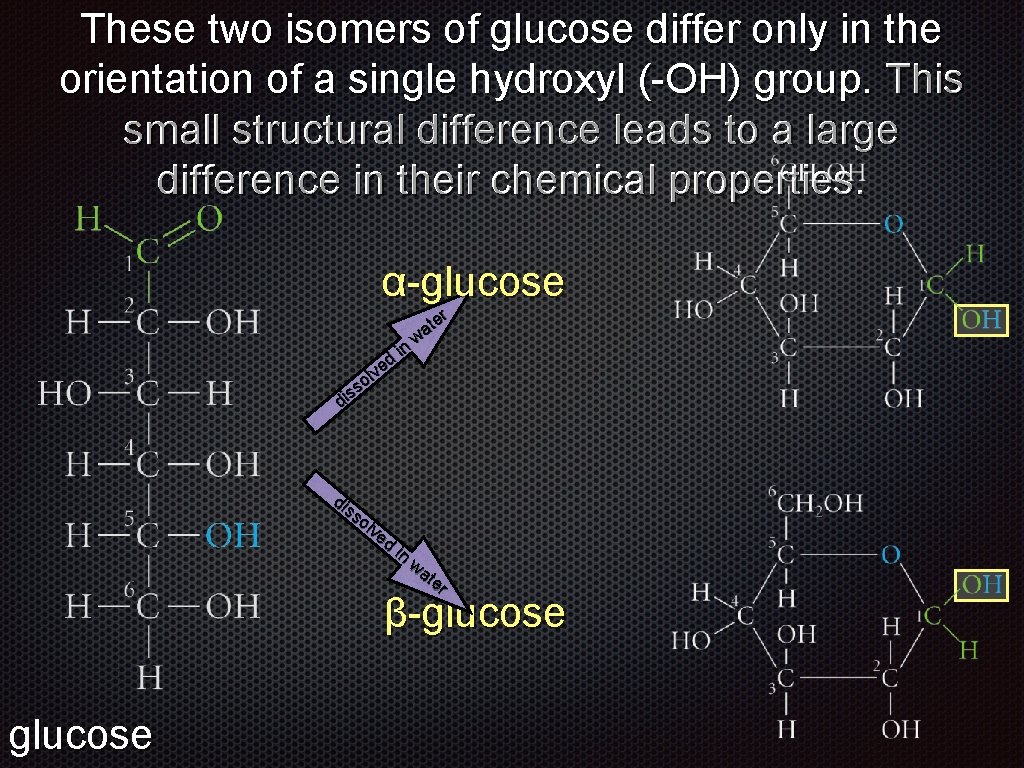
These two isomers of glucose differ only in the orientation of a single hydroxyl (-OH) group. This small structural difference leads to a large difference in their chemical properties. α-glucose d e v ol s s di in er t wa di ss ol ve d in wa te r β-glucose

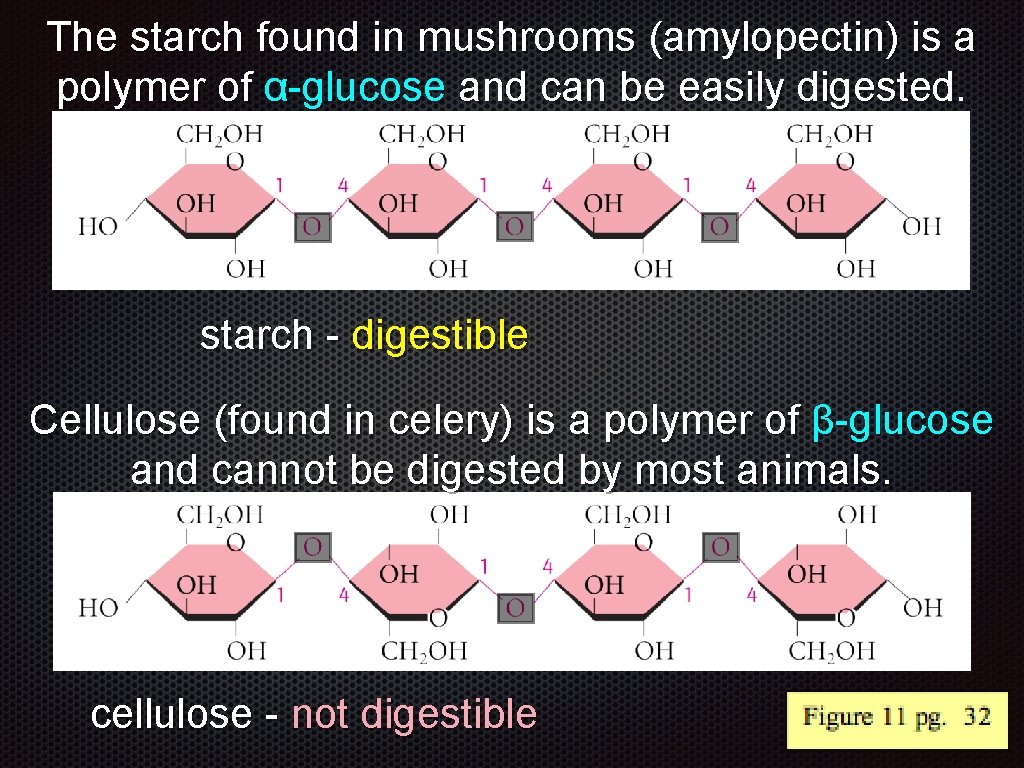
The starch found in mushrooms (amylopectin) is a polymer of α-glucose and can be easily digested. starch - digestible Cellulose (found in celery) is a polymer of β-glucose and cannot be digested by most animals. cellulose - not digestible

Isomers illustrate that the structure of a molecule determines the function of that molecule. Structure = Function

Read pgs. 24 -27 Do #1 pg. 28