Structural Isomerism Written by A Bourne MChem 2014

Structural Isomerism Written by A Bourne (MChem), 2014

Aims of this Lesson • Increase your understanding of isomers and the term structural isomerism • Increase your spatial skills and awareness using software designed by the CCDC

What are spatial skills? • The ability to mentally view and change 2 -dimensional and 3 -dimensional figures. • Important in chemistry, biology, medicine, mathematics, physics and everyday life. These are both the same molecule, viewed in 2 -D and 3 -D. What is the name of this simple molecule?

The basics You can use the CCDC’s Access Structures to view the 3 -D structures • Structural isomers have the same molecular formula (e. g. C 5 H 12), but a different bonding arrangement of the atoms. • Structural isomers have different physical and chemical properties. • This is a feature which is made use of in the petrochemical industry. CH 3 -CH 2 -CH 3 CH 3 -CH 2 -CH 3

Types of structural isomerism There are three types of structural isomerism: 1. Chain isomerism 2. Functional group isomerism 3. Position isomerism
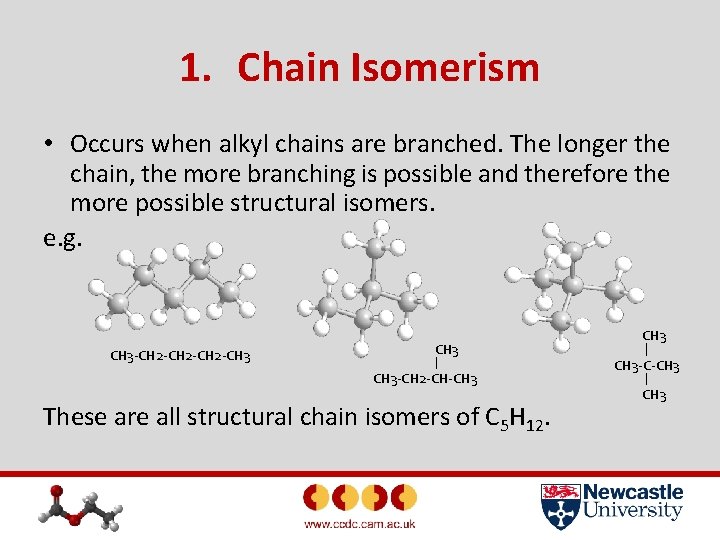
1. Chain Isomerism • Occurs when alkyl chains are branched. The longer the chain, the more branching is possible and therefore the more possible structural isomers. e. g. CH 3 -CH 2 -CH 3 CH 3 -C-CH 3 -CH 2 -CH-CH 3 CH 3 These are all structural chain isomers of C 5 H 12.

Task • Draw all the chain isomers of C 6 H 14?
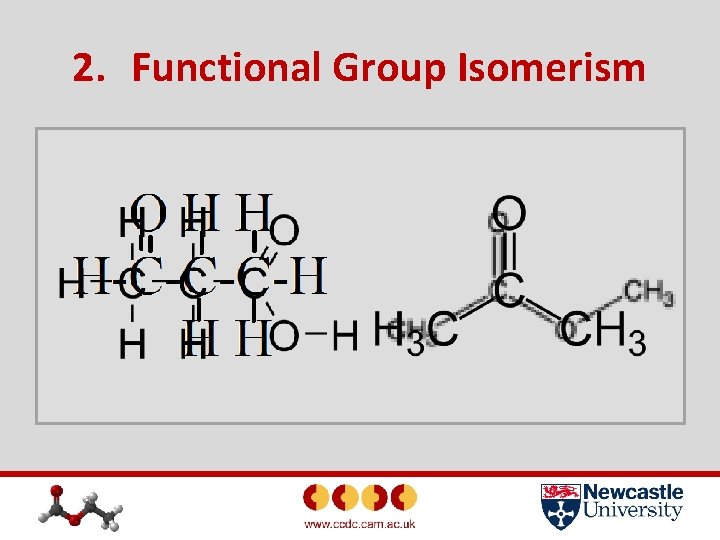
2. Functional Group Isomerism In this case, the compounds again have the same molecular mass, but contain different functional groups due to isomerism. Some examples are: • Propanal and propanone • Propanoic acid and ethyl ethanoate Have a go at drawing the isomers of the two above examples.

3. Position Isomerism • In this case, the alkyl (carbon) chain stays the same, with the functional group(s) moving round the skeleton. • This is NOT the same as functional group isomerism as the functional groups remain constant. e. g. What are their names?
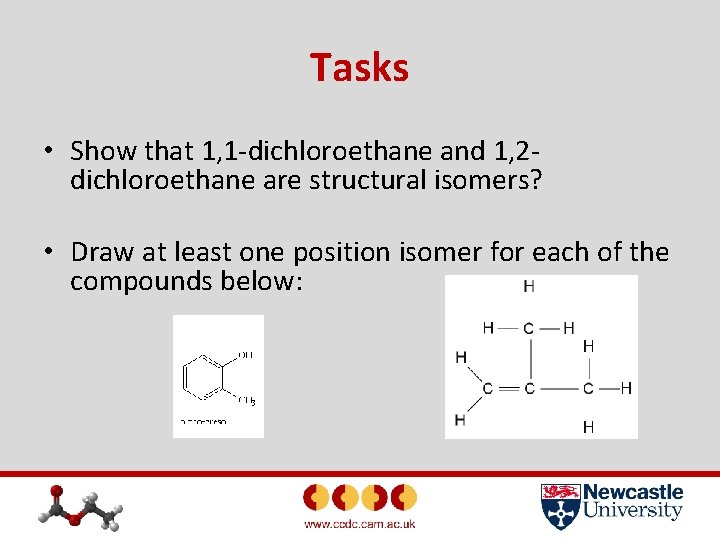
Tasks • Show that 1, 1 -dichloroethane and 1, 2 dichloroethane are structural isomers? • Draw at least one position isomer for each of the compounds below:

Re-cap • There are three types of structural isomerism: – Functional group – Positional – Chain • The molecular formula (and thus the molecular mass, Mr) always stay the same • Structural isomers have different physical and chemical properties, making them useful in many industries
- Slides: 11