Structural Insights into Kinase Inhibition Ramesh Sistla and

Structural Insights into Kinase Inhibition Ramesh Sistla and Subramanya H. S. Aurigene Discovery Technologies Ltd. #39 -40, KIADB Industrial Area, Electronic City Phase II Bangalore 560 100
Kinases - Introduction • Kinases are enzymes that catalyze phosphorylation • ATP + protein = ADP + phosphoprotein • Key signaling enzyme • Human genome encodes > 500 kinases - Kinome • They have been implicated in different diseases including cancer, metabolic disorders and central nervous system indications. • Depending on the amino acid a kinase phosphorylates, they are known as Serine/Threonine or Tyorsine kinases. AURIGENE……Acccelerating Discovery www. cellsignal. com 2
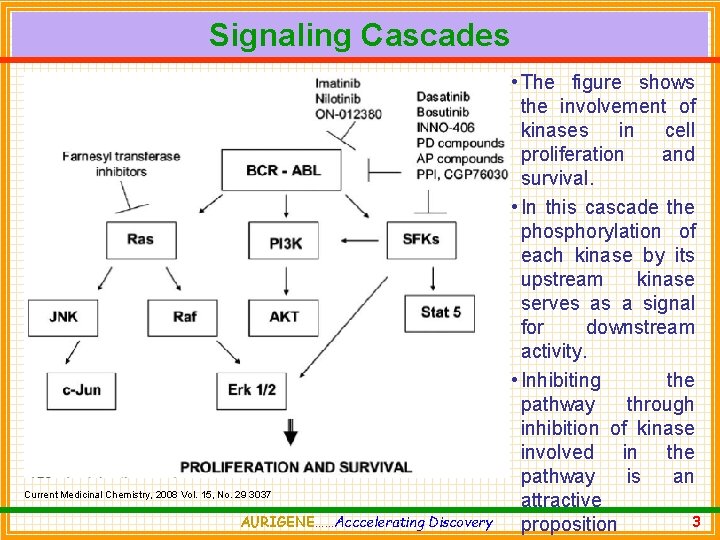
Signaling Cascades Current Medicinal Chemistry, 2008 Vol. 15, No. 29 3037 AURIGENE……Acccelerating Discovery • The figure shows the involvement of kinases in cell proliferation and survival. • In this cascade the phosphorylation of each kinase by its upstream kinase serves as a signal for downstream activity. • Inhibiting the pathway through inhibition of kinase involved in the pathway is an attractive 3 proposition
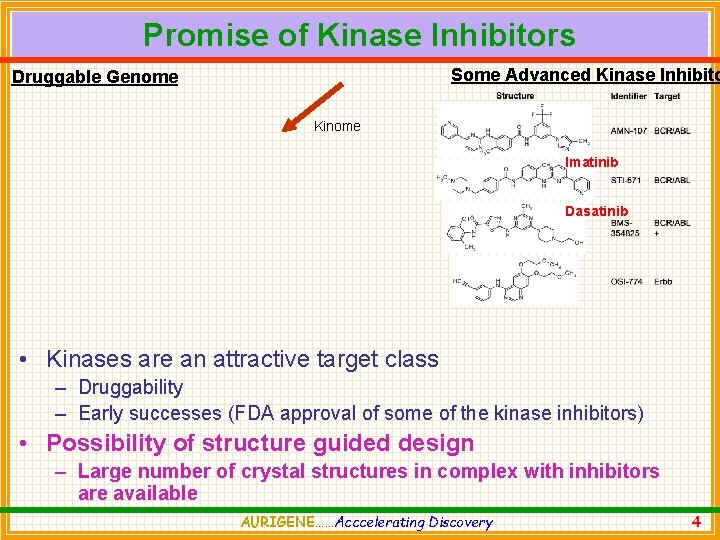
Promise of Kinase Inhibitors Some Advanced Kinase Inhibito Druggable Genome Kinome Imatinib Dasatinib • Kinases are an attractive target class – Druggability – Early successes (FDA approval of some of the kinase inhibitors) • Possibility of structure guided design – Large number of crystal structures in complex with inhibitors are available AURIGENE……Acccelerating Discovery 4
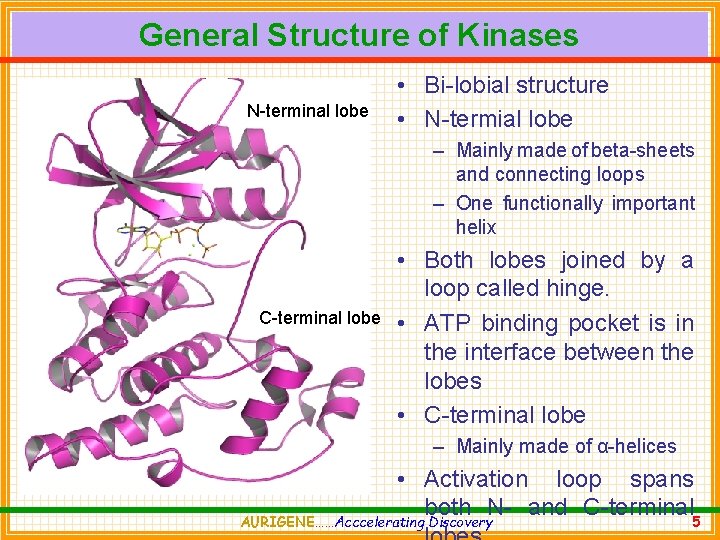
General Structure of Kinases N-terminal lobe • Bi-lobial structure • N-termial lobe – Mainly made of beta-sheets and connecting loops – One functionally important helix C-terminal lobe • Both lobes joined by a loop called hinge. • ATP binding pocket is in the interface between the lobes • C-terminal lobe – Mainly made of α-helices • Activation loop spans both N- and C-terminal 5 AURIGENE……Acccelerating Discovery
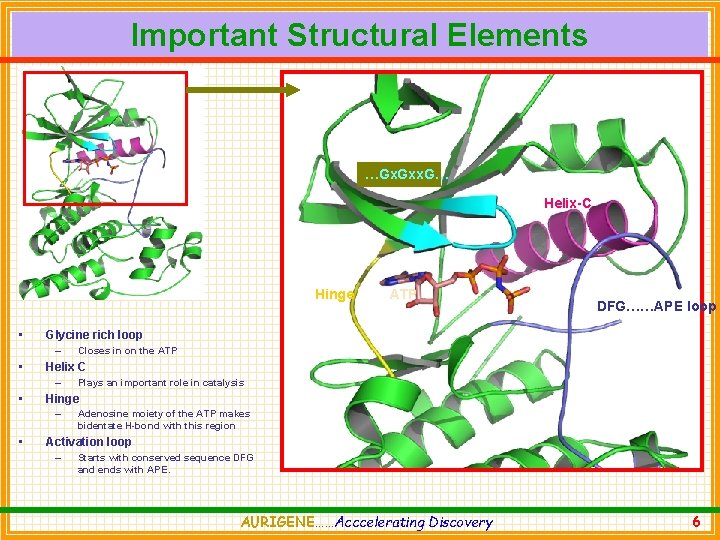
Important Structural Elements …Gx. Gxx. G… Helix-C Hinge • Plays an important role in catalysis Hinge – • Closes in on the ATP Helix C – • DFG……APE loop Glycine rich loop – • ATP Adenosine moiety of the ATP makes bidentate H-bond with this region Activation loop – Starts with conserved sequence DFG and ends with APE. AURIGENE……Acccelerating Discovery 6
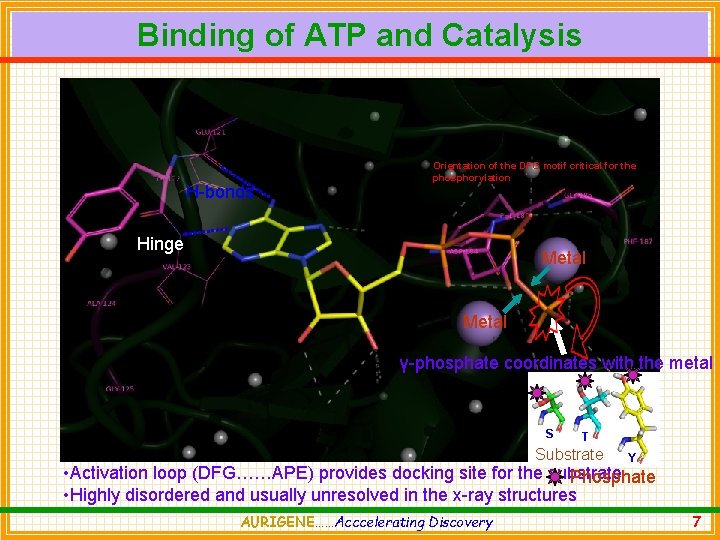
Binding of ATP and Catalysis H-bonds Orientation of the DFG motif critical for the phosphorylation Hinge Metal γ-phosphate coordinates with the metal S T Substrate Y • Activation loop (DFG……APE) provides docking site for the substrate Phosphate • Highly disordered and usually unresolved in the x-ray structures AURIGENE……Acccelerating Discovery 7
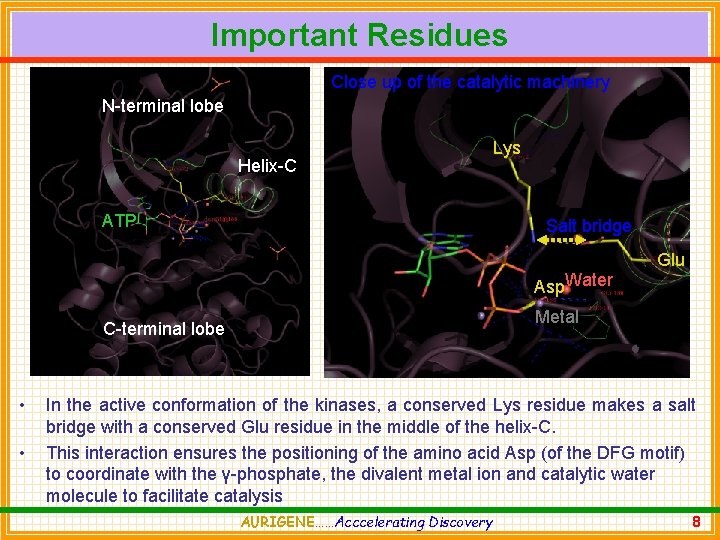
Important Residues Close up of the catalytic machinery N-terminal lobe Helix-C ATP Lys Salt bridge Asp. Water Metal C-terminal lobe • • Glu In the active conformation of the kinases, a conserved Lys residue makes a salt bridge with a conserved Glu residue in the middle of the helix-C. This interaction ensures the positioning of the amino acid Asp (of the DFG motif) to coordinate with the γ-phosphate, the divalent metal ion and catalytic water molecule to facilitate catalysis AURIGENE……Acccelerating Discovery 8
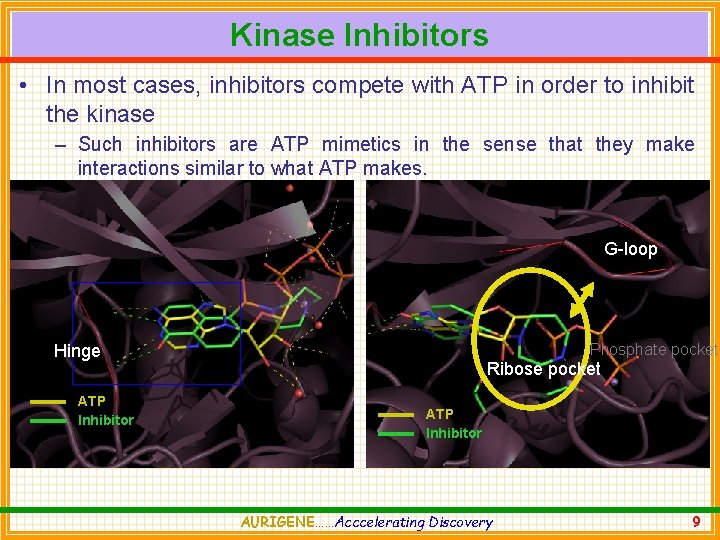
Kinase Inhibitors • In most cases, inhibitors compete with ATP in order to inhibit the kinase – Such inhibitors are ATP mimetics in the sense that they make interactions similar to what ATP makes. G-loop Phosphate pocket Hinge ATP Inhibitor Ribose pocket ATP Inhibitor AURIGENE……Acccelerating Discovery 9
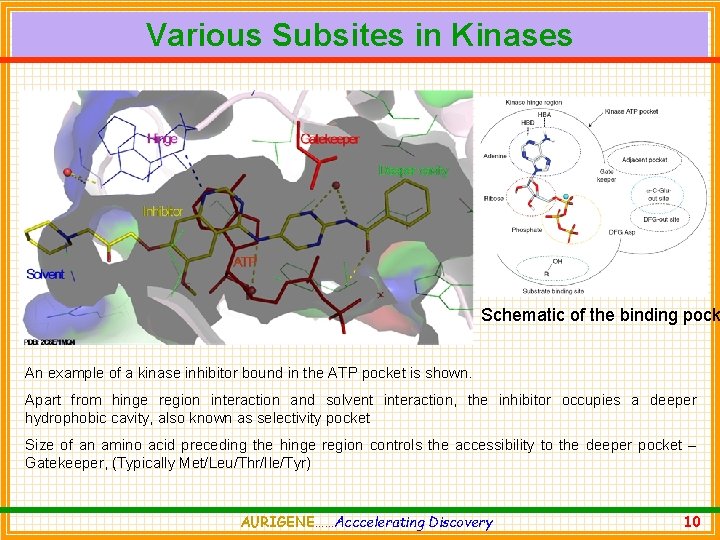
Various Subsites in Kinases Schematic of the binding pock An example of a kinase inhibitor bound in the ATP pocket is shown. Apart from hinge region interaction and solvent interaction, the inhibitor occupies a deeper hydrophobic cavity, also known as selectivity pocket Size of an amino acid preceding the hinge region controls the accessibility to the deeper pocket – Gatekeeper, (Typically Met/Leu/Thr/Ile/Tyr) AURIGENE……Acccelerating Discovery 10
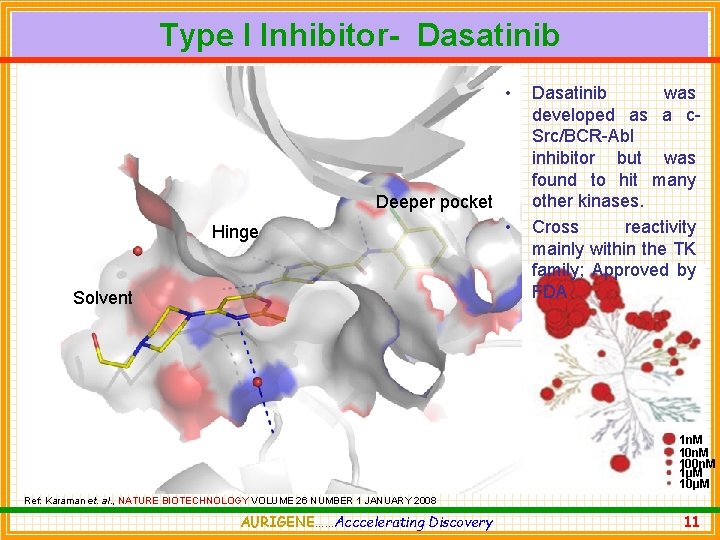
Type I Inhibitor- Dasatinib • Deeper pocket Hinge Solvent • Dasatinib was developed as a c. Src/BCR-Abl inhibitor but was found to hit many other kinases. Cross reactivity mainly within the TK family; Approved by FDA 1 n. M 100 n. M 1μM 10μM Ref: Karaman et. al. , NATURE BIOTECHNOLOGY VOLUME 26 NUMBER 1 JANUARY 2008 AURIGENE……Acccelerating Discovery 11
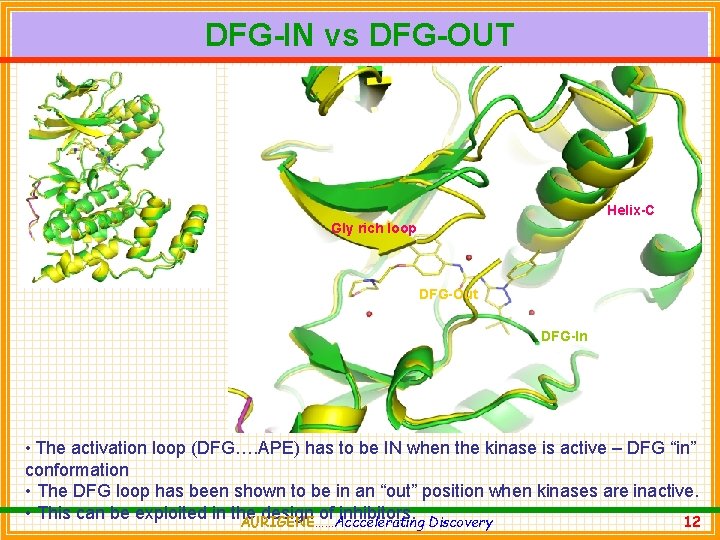
DFG-IN vs DFG-OUT Helix-C Gly rich loop DFG-Out DFG-In • The activation loop (DFG…. APE) has to be IN when the kinase is active – DFG “in” conformation • The DFG loop has been shown to be in an “out” position when kinases are inactive. • This can be exploited in the design of inhibitors. AURIGENE……Acccelerating Discovery 12
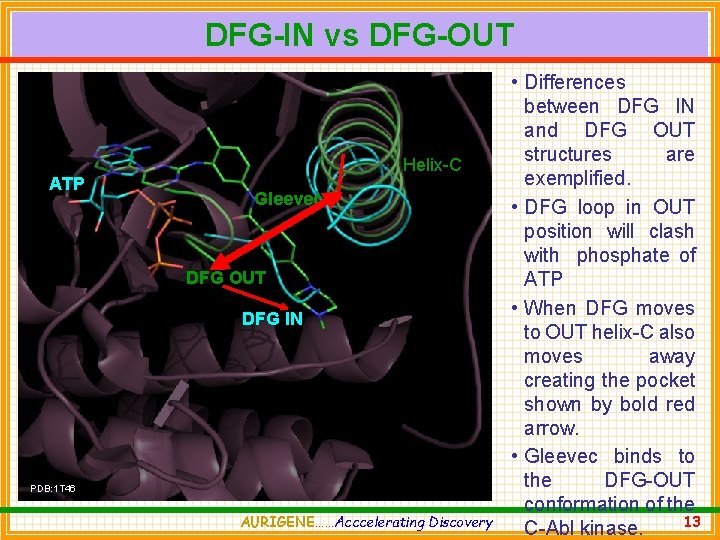
DFG-IN vs DFG-OUT ATP Helix-C Gleevec DFG OUT DFG IN PDB: 1 T 46 AURIGENE……Acccelerating Discovery • Differences between DFG IN and DFG OUT structures are exemplified. • DFG loop in OUT position will clash with phosphate of ATP • When DFG moves to OUT helix-C also moves away creating the pocket shown by bold red arrow. • Gleevec binds to the DFG-OUT conformation of the 13 C-Abl kinase.
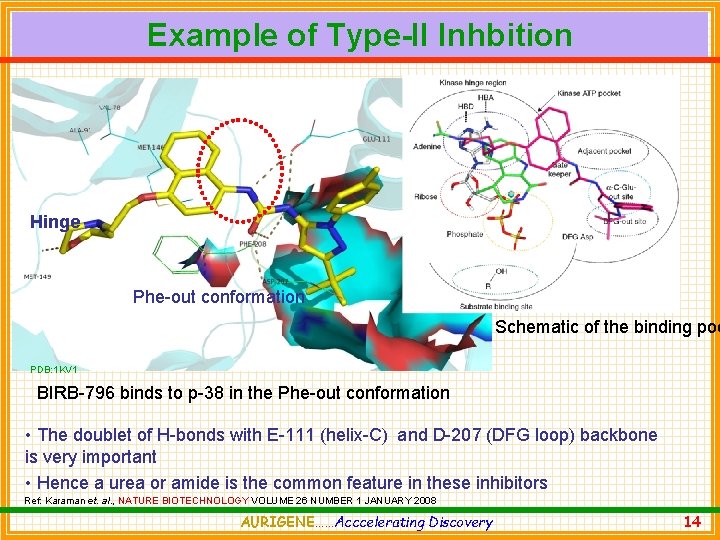
Example of Type-II Inhbition Hinge Phe-out conformation Schematic of the binding poc PDB: 1 KV 1 BIRB-796 binds to p-38 in the Phe-out conformation • The doublet of H-bonds with E-111 (helix-C) and D-207 (DFG loop) backbone is very important • Hence a urea or amide is the common feature in these inhibitors Ref: Karaman et. al. , NATURE BIOTECHNOLOGY VOLUME 26 NUMBER 1 JANUARY 2008 AURIGENE……Acccelerating Discovery 14
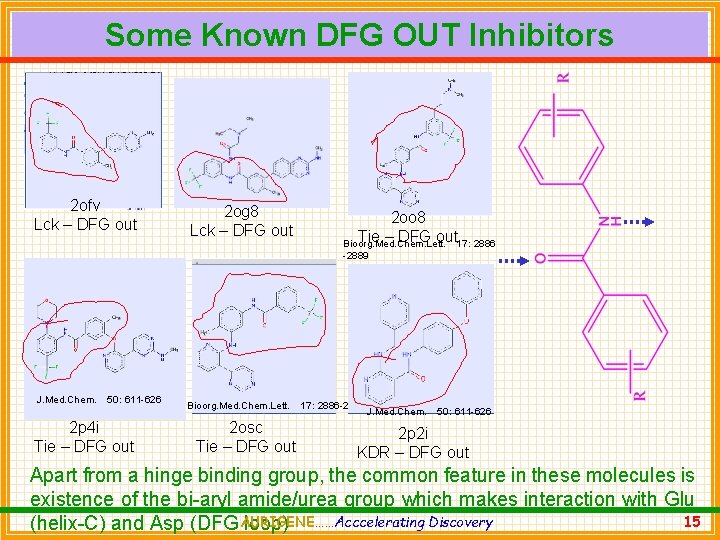
Some Known DFG OUT Inhibitors 2 ofv Lck – DFG out 2 og 8 Lck – DFG out 2 oo 8 Tie – DFG out 17: 2886 Bioorg. Med. Chem. Lett. -2889 J. Med. Chem. 50: 611 -626 2 p 4 i Tie – DFG out Bioorg. Med. Chem. Lett. 2 osc Tie – DFG out 17: 2886 -2889 J. Med. Chem. 50: 611 -626 2 p 2 i KDR – DFG out Apart from a hinge binding group, the common feature in these molecules is existence of the bi-aryl amide/urea group which makes interaction with Glu Discovery 15 (helix-C) and Asp (DFG AURIGENE……Acccelerating loop)
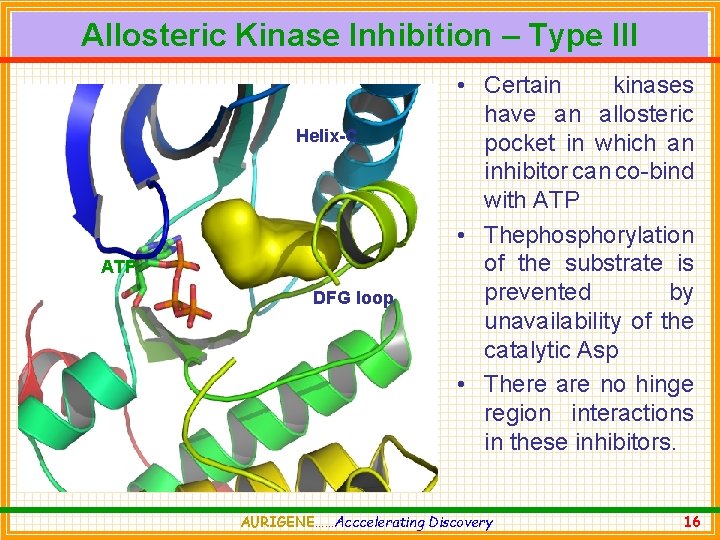
Allosteric Kinase Inhibition – Type III Helix-C ATP DFG loop • Certain kinases have an allosteric pocket in which an inhibitor can co-bind with ATP • Thephosphorylation of the substrate is prevented by unavailability of the catalytic Asp • There are no hinge region interactions in these inhibitors. AURIGENE……Acccelerating Discovery 16
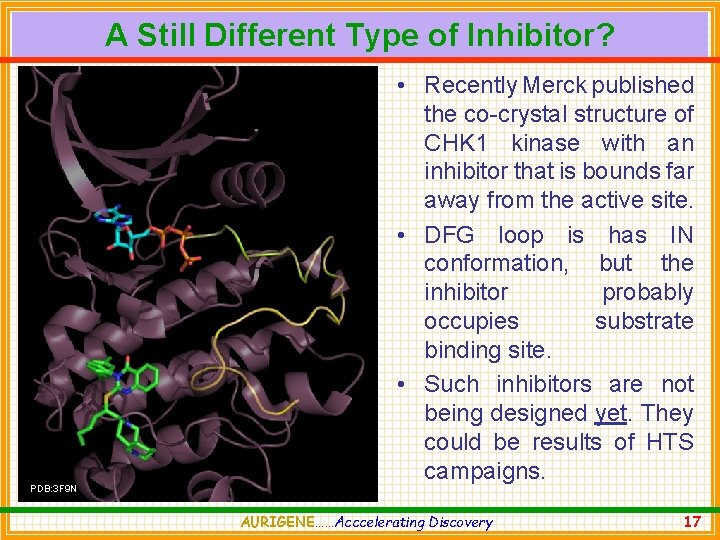
A Still Different Type of Inhibitor? PDB: 3 F 9 N • Recently Merck published the co-crystal structure of CHK 1 kinase with an inhibitor that is bounds far away from the active site. • DFG loop is has IN conformation, but the inhibitor probably occupies substrate binding site. • Such inhibitors are not being designed yet. They could be results of HTS campaigns. AURIGENE……Acccelerating Discovery 17
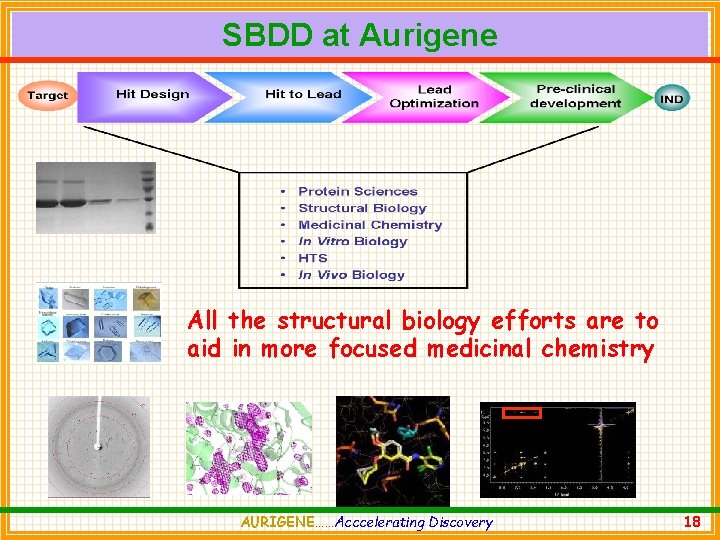
SBDD at Aurigene All the structural biology efforts are to aid in more focused medicinal chemistry AURIGENE……Acccelerating Discovery 18
- Slides: 18