Structural Geology Spring 2003 Structural Geology Structural geologists




































- Slides: 41

Structural Geology Spring 2003

Structural Geology ► Structural geologists are concerned with why parts of the Earth have been bent into folds and others have been broken by faults. ► Mapping of these structures provides important information to land managers and mineral exploration. ► Understanding of these features help us understand the dynamic Earth.

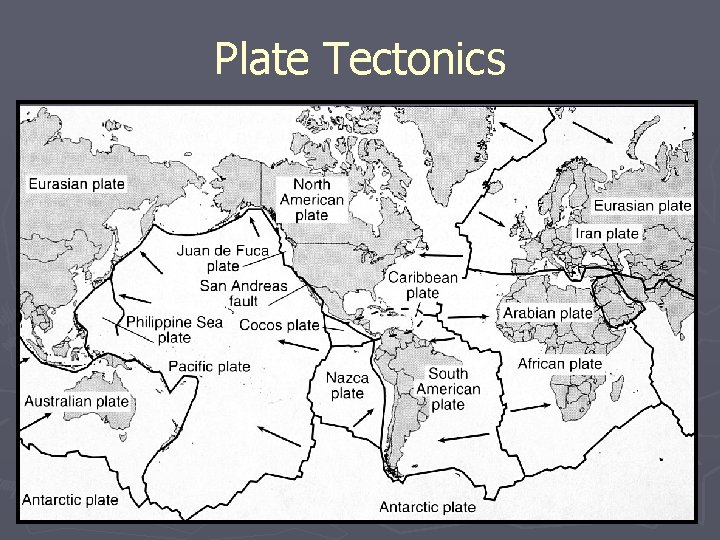
Plate Tectonics

Tectonic Structures ► Most structures are driven by the forces of Plate Tectonics ► The kinds of structures are determined by: § Temperature and pressure § Composition § Layering § Anisotropy or Isotropy of the layers § Amount of fluids present

Tectonic Structures ► Time (or rate of change) is very importance § A rock may behave in a ductile or brittle fashion depending upon how quickly it is deformed

Tectonic Structures ► Ductile deformation produces: § Folds § Ductile Faults § Cleavages § Foliation

Tectonic Structures ► Brittle Deformation § Certain types of folds § Brittle Faults § Joints

Nontectonic Structures ► Nontectonic structures can mimic tectonic § Meteor impacts § Landslides § Structures produce by gravitational forces

3 -Dimensional Objects ► Visualization of 3 -Dimensional Objects



Structural Geology ► Subdisciplines of § Field Relations Structural Geology ► Make accurate geologic maps ► Measure orientations of small structures to inform us of the shape of larger structures ► Study the sequence of development and superposition of different kinds of structures § Rock Mechanics – the application of physics to the study of rock materials. § Tectonic and Regional Structural Geology – Study of mountain ranges, parts of entire continents, trenches and island arcs, oceanic ridges

Applications of Structural Geology ► Engineering Issues § Bridges § Dams § Power Plants § Highway Cuts § Large Buildings § Airports

Applications of Structural Geology ► Environmental Issues § Earthquake hazard § Location of landfill sites § Contamination cleanup § Distribution of groundwater § Mineral exploration

Scale in Structural Geology ► Microscopic – Need magnification § Foliation, Micro folds ► Mesoscopic outcrops – Hand specimens and § Foliation, Folds, Faults ► Macroscopic – Mountainside to map levels § Basins, domes, Metamorphic Core Complexes

Scale in Structural Geology ► Non-penetrative scales structures – not present on all § Faults § Isolated folds ► Penetrative structures – found on any scale that we chose to study § Slaty cleavage § Foliation § Some folds

Scale and Folds Figure 1 -6

Fundamental Concepts ► Doctrine of Uniformitarianism ► Law of Superposition ► Law of Original Horizontality ► Law of Cross-Cutting Relationships ► Law of Faunal Succession ► Multiple Working Hypotheses ► Outrageous Hypothesis

Fundamental Concepts ► Pumpelly’s Rule – Small structures are a key to and mimic the styles and orientations of larger structures of the same generation within a particular area.

Plate Tectonics ► Driving Mechanisms § Convection § Push-Pull Theory ► Plate Boundaries § Divergent § Convergent § Transform

Geochronology ► Absolute Age Dating ► Review of atomic structure ► Most useful isotope decay processes

Using radioactivity in dating ► Reviewing ►Atomic basic atomic structure number § An element’s identifying number § Equal to the number of protons in the atom’s nucleus ►Mass number § Sum of the number of protons and neutrons in an atom’s nucleus

Using radioactivity in dating ► Reviewing basic atomic structure ►Isotope § Variant of the same parent atom § Differs in the number of neutrons § Results in a different mass number than the parent atom

Using radioactivity in dating ► Radioactivity ►Spontaneous changes (decay) in the structure of atomic nuclei ► Types of radioactive decay ►Alpha emission § Emission of 2 protons and 2 neutrons (an alpha particle) § Mass number is reduced by 4 and the atomic number is lowered by 2

Using radioactivity in dating ► Types of radioactive decay ►Beta emission § An electron (beta particle) is ejected from the nucleus § Mass number remains unchanged and the atomic number increases by 1

Using radioactivity in dating ► Types of radioactive decay ►Electron capture § An electron is captured by the nucleus § The electron combines with a proton to form a neutron § Mass number remains unchanged and the atomic number decreases by 1

Common Types of Radioactive Decay

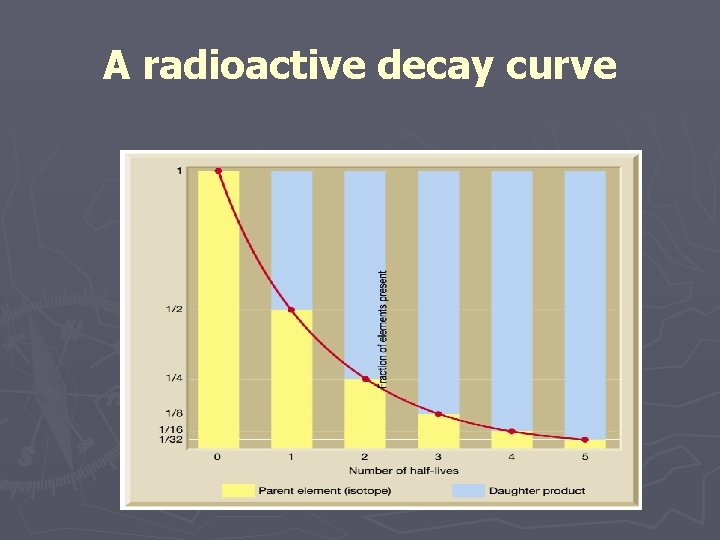
Using radioactivity in dating ► Parent – an unstable radioactive isotope ► Daughter product – the isotopes resulting from the decay of a parent ► Half-life – the time required for one-half of the radioactive nuclei in a sample to decay

A radioactive decay curve

Using radioactivity in dating ► Radiometric ►Principle dating of radioactive dating § The percentage of radioactive atoms that decay during one half-life is always the same (50 percent) § However, the actual number of atoms that decay continually decreases § Comparing the ratio of parent to daughter yields the age of the sample

Using radioactivity in dating ► Radiometric ►Sources dating of error § A closed system is required § To avoid potential problems, only fresh, unweathered rock samples should be used § Blocking Temperature – The temperature below which a crystal lattice traps radioactive daughter products.

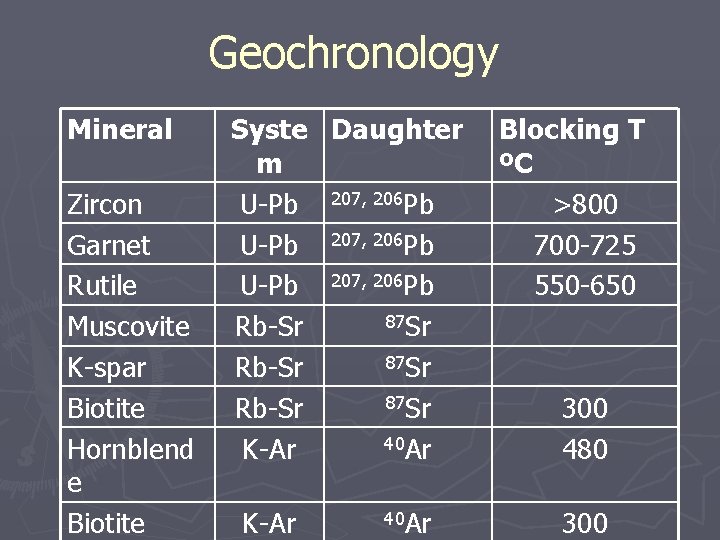
Geochronology Mineral Zircon Garnet Rutile Muscovite K-spar Biotite Hornblend e Biotite Syste Daughter m U-Pb 207, 206 Pb 87 Sr Rb-Sr 40 Ar K-Ar 40 Ar Blocking T ºC >800 700 -725 550 -650 300 480 300

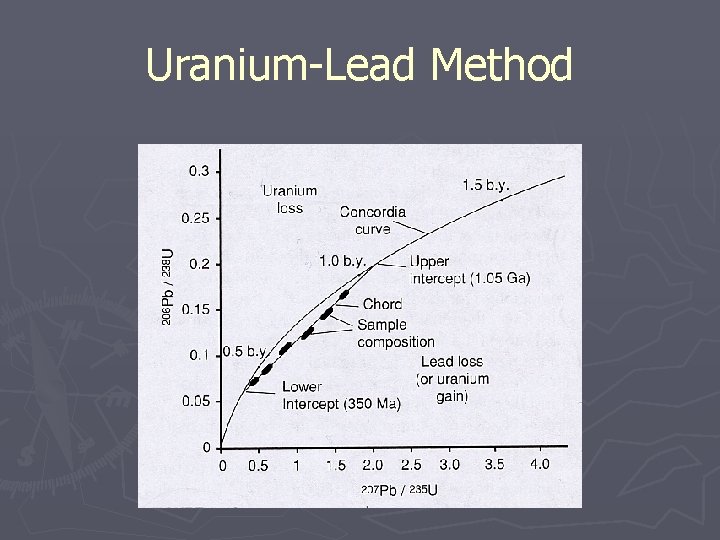
Geochronology ► Uranium-Lead Method (U-Pb) § Most reliable technique for rocks § Ages exceed 10 million years § Use of Zircons for dating 238 U 235 U 232 Th 206 Pb (half-life = 4. 5 x 109 yrs) 207 Pb (half-life = 0. 7 x 109 yrs) 208 Pb (half-life = 1. 4 x 109 yrs)

Uranium-Lead Method

Uranium-Lead Method

Geochronology ► Robidium-Strontium (Rb-Sr) § Most applicable in rocks over 100 million years old § Whole-rock ages are more reliable in Rb-Sr § No gaseous daughter elements § Principle source of error is later metamorphism and hydrothermal alteration. 87 Rb 87 Sr + ß– (half-life = 48. 8 x 109 yrs)

Geochronology ► Potassium-Argon § § § (K-Ar) Used for rocks around 1 million years old Ar is a gas and can be easily released from most rocks Biotite, muscovite, hornblende retain argon better than other minerals § Low blocking temperatures (300ºC - 480 ºC) 40 Ca 40 K + ß– (half-life = 1. 2 x 109 yrs) 40 Ar

Geochronology ►Argon-Argon (40 Ar-39 Ar) § Samples must be irradiated to convert 39 K to 39 Ar § Can determine the cooling history of the rocks § Useful for determining the time of uplift, metamorphism, or emplacement of structures

Geochronology ► Samarium - Neodynium (Sm-Nd) § Used mainly for dating ocean floor basalts because sea water is abundant in Sr but depleted in Nd § Therefore, can be used to determine contamination by sea water and hydrothermal alteration 147 Sm 143 Nd (half-life = 106 x 109 yrs)

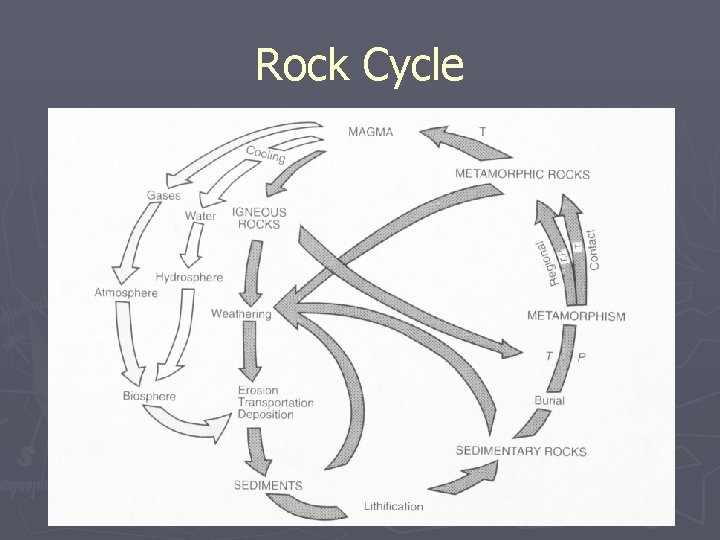
Rock Cycle
