Strong vs Weak AcidsBases Strong acids or bases
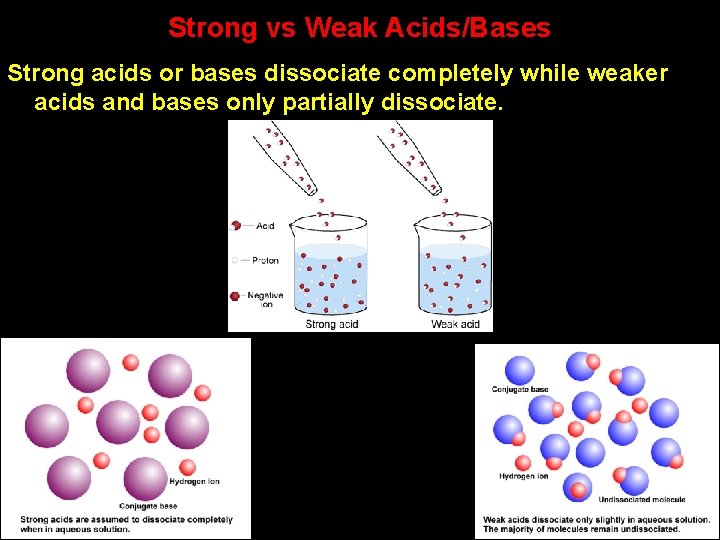
Strong vs Weak Acids/Bases Strong acids or bases dissociate completely while weaker acids and bases only partially dissociate.

Strong vs Weak Acid Strong Acid: HCl (aq) H+ (aq) + Cl- (aq) Exists only in the form of ions HCl + H 2 O H 3 O+ + Cl- The actual ion species (Lots of H 3 O+ ) Weak Acid: CH 3 COOH (acetic acid-vinegar is 4 -18% acetic acid by mass) CH 3 COOH + H 2 O H 3 O+ + CH 3 COO(Little of H 3 O+) Most of the aa is in the combined form to the left of

Experimental Evidence for Strong vs Weak Acids Solutions with many charged ions will conduct electricity better which can be measured with a qualitative (showing a general change) or quantitative (giving exact numbers) device. The conductivity device shown shows qualitative differences. strong acid weak acid
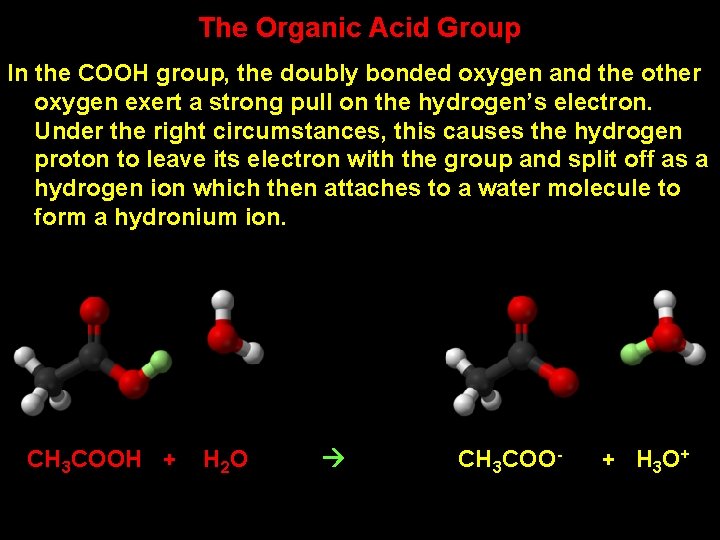
The Organic Acid Group In the COOH group, the doubly bonded oxygen and the other oxygen exert a strong pull on the hydrogen’s electron. Under the right circumstances, this causes the hydrogen proton to leave its electron with the group and split off as a hydrogen ion which then attaches to a water molecule to form a hydronium ion. CH 3 COOH + H 2 O CH 3 COO- + H 3 O +

Dilute/Concentrated - Not the same as Weak/Strong Dilute and concentrated refers to the molarity of a solution (particles of acid/base per volume of solution). Weak vs Strong refers to the % ionization of an acid/base. A 10. 0 M HF (aq) solution is CONCENTRATED but WEAK. A. 001 M HCl (aq) solution is DILUTE but STRONG A 10. 0 M HF(aq) solution actually has less H 3 O+ than a. 1 M HCl(aq) solution even though it is 100 X more concentrated – because it is much less ionized.
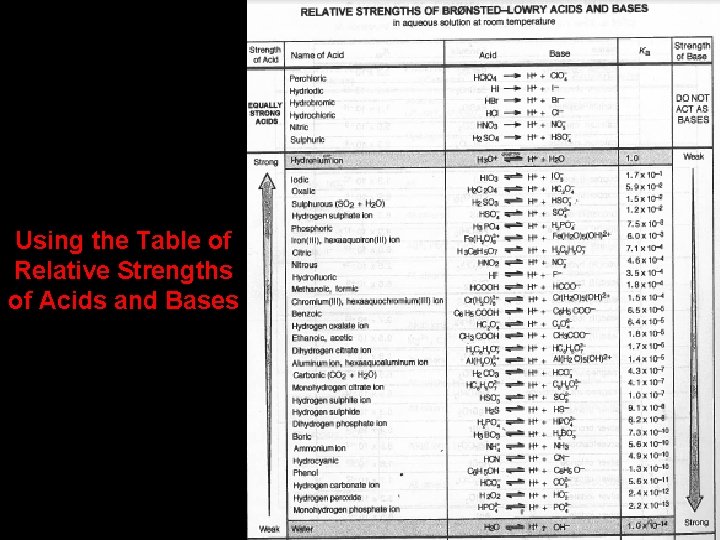
Using the Table of Relative Strengths of Acids and Bases
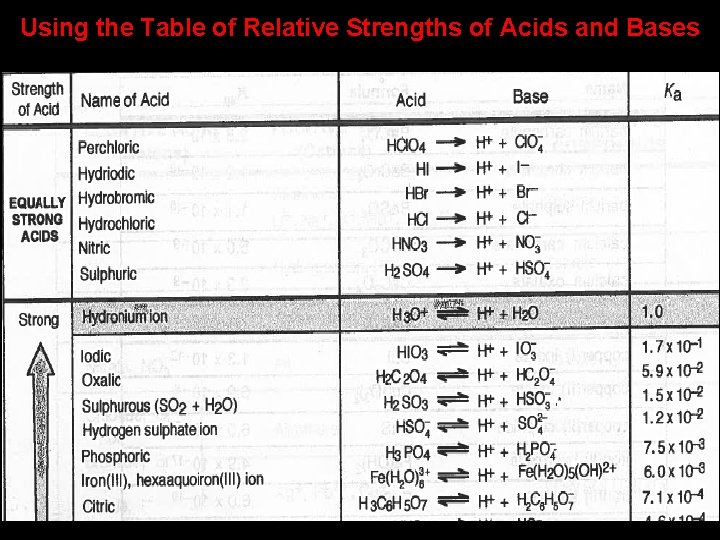
Using the Table of Relative Strengths of Acids and Bases
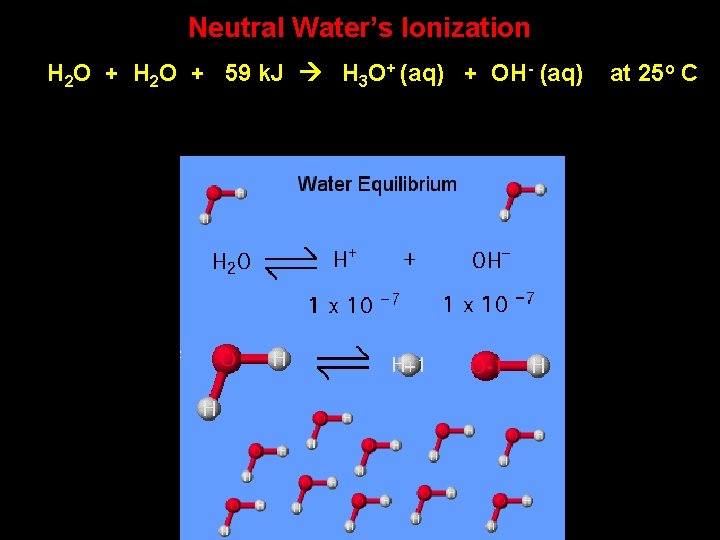
Neutral Water’s Ionization H 2 O + 59 k. J H 3 O+ (aq) + OH- (aq) at 25 o C

Water’s Ionization Constant H 2 O + 59 k. J H 3 O+ (aq) + OH- (aq) Kw = [H 3 O][OH-] = at 25 o C 1 x 10 -14 [H 3 O] = 1 x 10 -7 [OH-] = 1 x 10 -7 Note that with neutral water at 25 o C, the concentrations of H 3 O+ and OH- are equal.
![Water’s [H 3 O+] Over 25 o C H 2 O + 59 k. Water’s [H 3 O+] Over 25 o C H 2 O + 59 k.](http://slidetodoc.com/presentation_image_h2/45c1bdb4f371af7d4af94bc614ff833f/image-10.jpg)
Water’s [H 3 O+] Over 25 o C H 2 O + 59 k. J H 3 O+ (aq) + OH- (aq) at 25 o C At temperatures above 25 C, the equilibrium shifts right. This produces more [H 3 O+] and [OH-]. More [H 3 O+] typically means greater acidity but not here. Since the [H 3 O+] = [OH-] , the water is still neutral (although its p. H drops due to the extra hydronium [H 3 O+] ions).

A A

A A

A A

A A

A A

A A

A A

A A
- Slides: 18