Strong and Weak AcidsBases The strength of an

Strong and Weak Acids/Bases The strength of an acid (or base) is determined by the amount of IONIZATION. 1
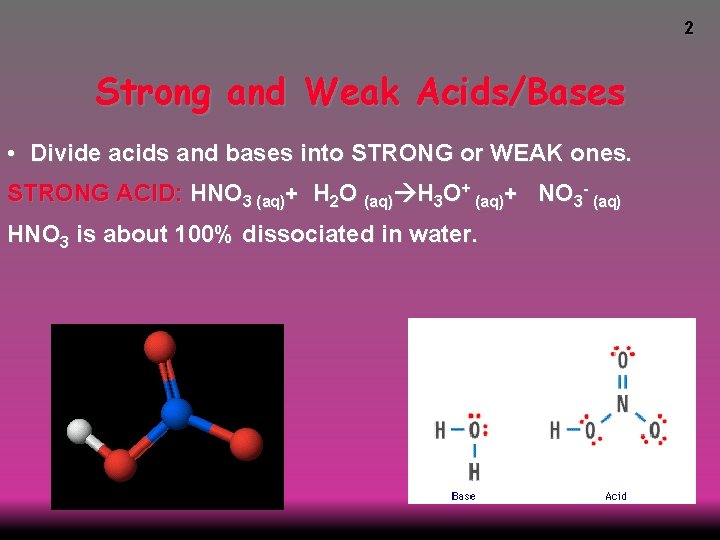
2 Strong and Weak Acids/Bases • Divide acids and bases into STRONG or WEAK ones. STRONG ACID: HNO 3 (aq)+ H 2 O (aq) H 3 O+ (aq)+ NO 3 - (aq) HNO 3 is about 100% dissociated in water.
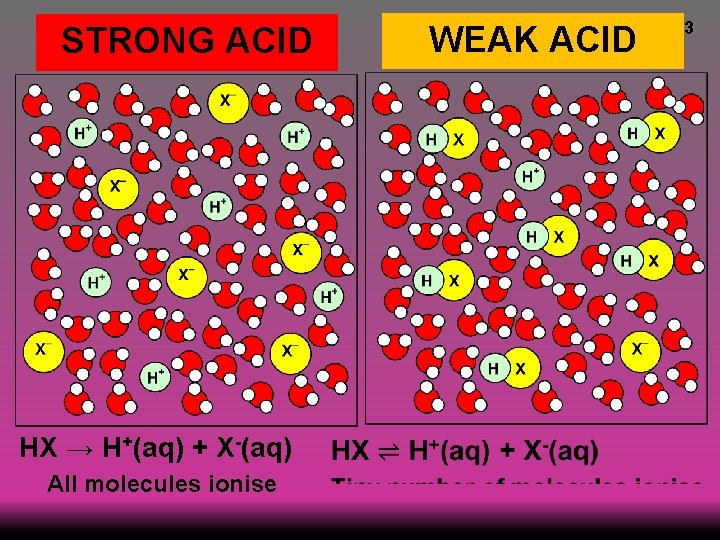
STRONG ACID HX → H+(aq) + X-(aq) All molecules ionise WEAK ACID 3

4 Strong Acids • You will recall that the seven strong acids are HCl, HBr, HI, HNO 3, H 2 SO 4, HCl. O 3, and HCl. O 4. • These are strong electrolytes and exist totally as ions in aqueous solution. • For the monoprotic strong acids, [H 3 O+] = [acid].

5 Factors Affecting Acid Strength For a series of oxyacids, acidity increases with the number of oxygens.
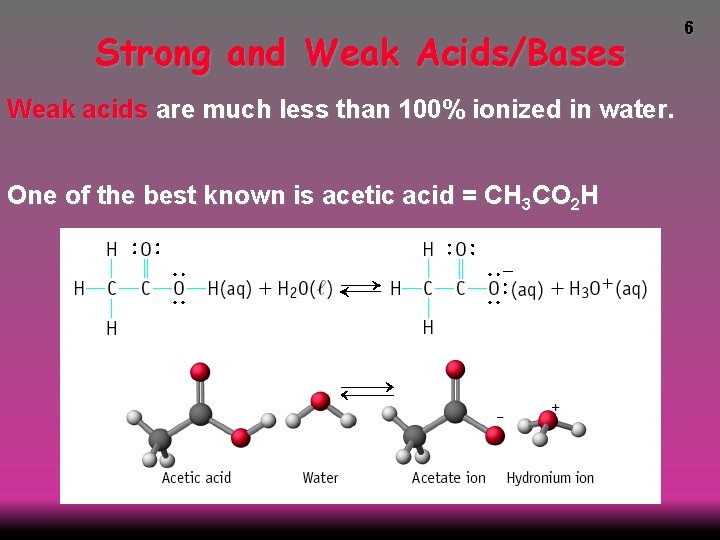
Strong and Weak Acids/Bases Weak acids are much less than 100% ionized in water. One of the best known is acetic acid = CH 3 CO 2 H 6
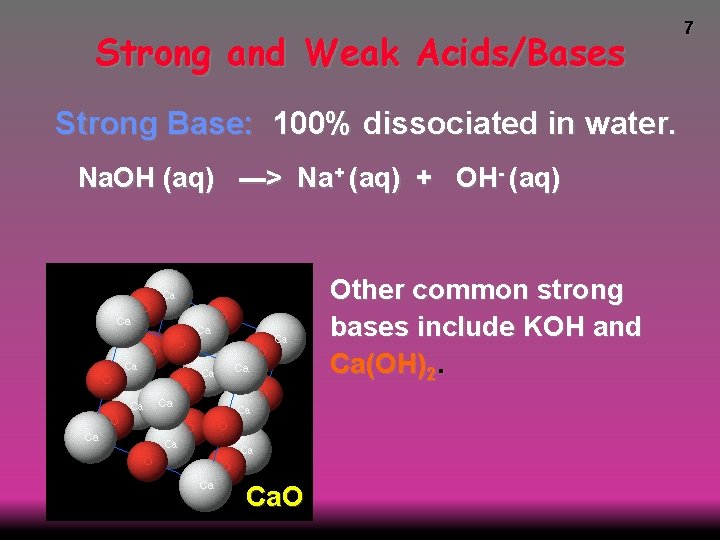
Strong and Weak Acids/Bases Strong Base: 100% dissociated in water. Na. OH (aq) ---> Na+ (aq) + OH- (aq) Other common strong bases include KOH and Ca(OH)2. Ca. O 7
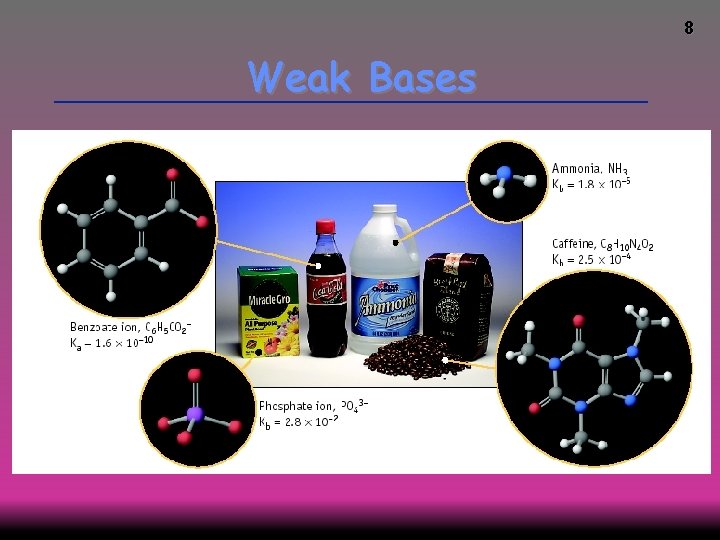
8 Weak Bases
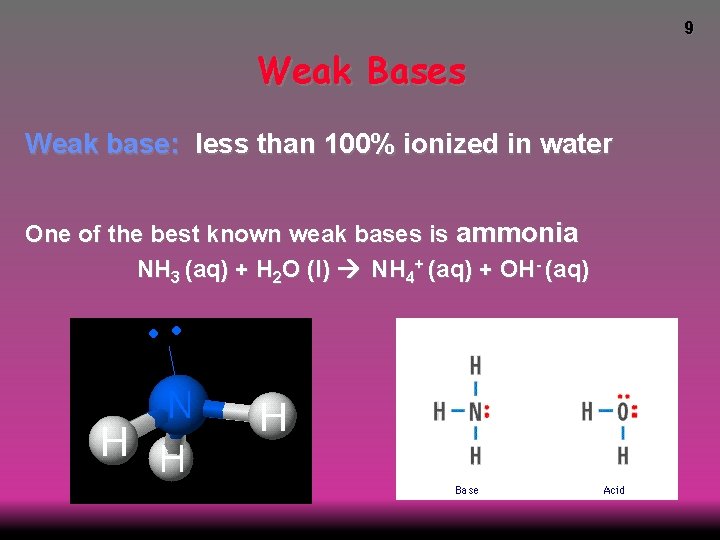
9 Weak Bases Weak base: less than 100% ionized in water One of the best known weak bases is ammonia NH 3 (aq) + H 2 O (l) NH 4+ (aq) + OH- (aq)
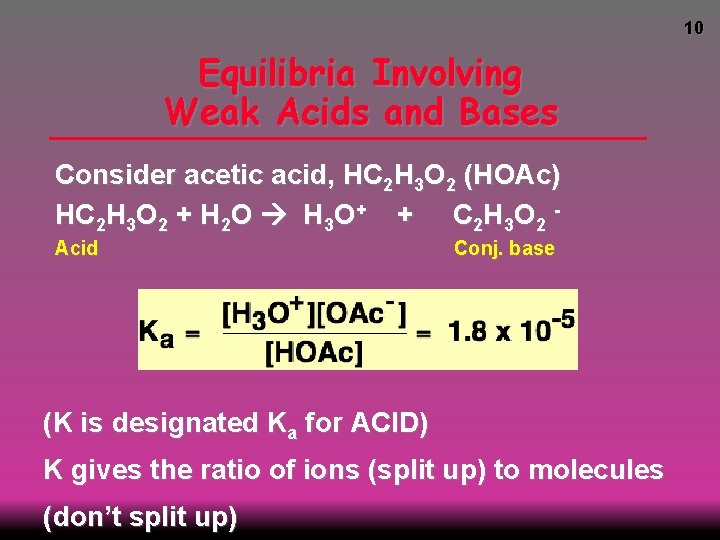
10 Equilibria Involving Weak Acids and Bases Consider acetic acid, HC 2 H 3 O 2 (HOAc) HC 2 H 3 O 2 + H 2 O H 3 O+ + C 2 H 3 O 2 Acid Conj. base (K is designated Ka for ACID) K gives the ratio of ions (split up) to molecules (don’t split up)
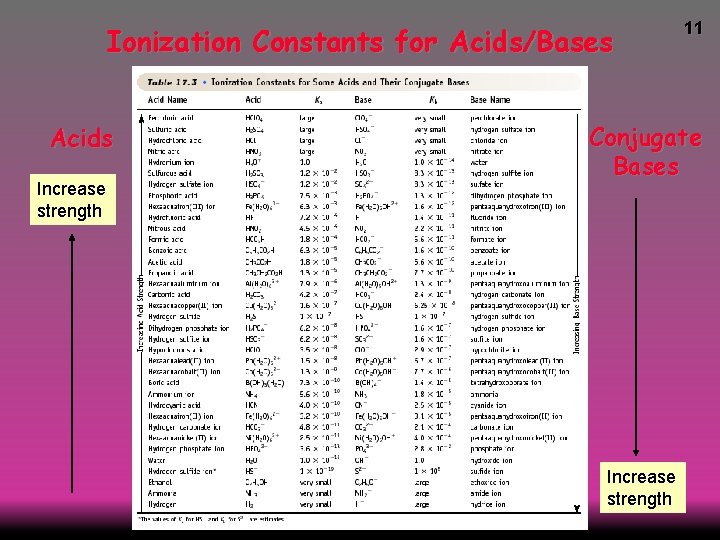
Ionization Constants for Acids/Bases Acids Increase strength 11 Conjugate Bases Increase strength

Equilibrium Constants for Weak Acids Weak acid has Ka < 1 Leads to small [H 3 O+] and a p. H of 2 - 7 12
![Equilibrium Constants for Weak Bases Small [OH-] and a p. H of 12 – Equilibrium Constants for Weak Bases Small [OH-] and a p. H of 12 –](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-13.jpg)
Equilibrium Constants for Weak Bases Small [OH-] and a p. H of 12 – 7. 1 13
![14 Relation of Ka, Kb, [H 3 O+] and p. H 14 Relation of Ka, Kb, [H 3 O+] and p. H](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-14.jpg)
14 Relation of Ka, Kb, [H 3 O+] and p. H

Equilibria Involving A Weak Acid You have 1. 00 M HOAc. Calc. the equilibrium concs. of HOAc, H 3 O+, OAc-, and the p. H. Step 1. Define equilibrium concs. in ICE table. [HOAc] [H 3 O+] [OAc-] initial 1. 00 0 0 change -x 1. 00 -x +x x equilib 15

Equilibria Involving A Weak Acid You have 1. 00 M HOAc. Calc. the equilibrium concs. of HOAc, H 3 O+, OAc-, and the p. H. Step 2. Write Ka expression This is a quadratic. Solve using quadratic formula. or you can make an approximation if x is very small! (Rule of thumb: 10 -5 or smaller is ok) 16

Equilibria Involving A Weak Acid You have 1. 00 M HOAc. Calc. the equilibrium concs. of HOAc, H 3 O+, OAc-, and the p. H. Step 3. Solve Ka expression First assume x is very small because Ka is so small. Now we can more easily solve this approximate expression. 17

Approximating If K is really small, the equilibrium concentrations will be nearly the same as the initial concentrations. Example: 0. 20 – x is just about 0. 20 if x is really small. If the K is 10 -5 or smaller (10 -6, 10 -7, etc. ), you should approximate. Otherwise, you have to use the quadratic. 18

Equilibria Involving A Weak Acid You have 1. 00 M HOAc. Calc. the equilibrium concs. of HOAc, H 3 O+, OAc-, and the p. H. Step 3. Solve Ka approximate expression x = [H 3 O+] = [OAc-] = 4. 2 x 10 -3 M p. H = - log [H 3 O+] = -log (4. 2 x 10 -3) = 2. 37 19

Equilibria Involving A Weak Acid Calculate the p. H of a 0. 0010 M solution of formic acid, HCO 2 H + H 2 O HCO 2 - + H 3 O+ Ka = 1. 8 x 10 -4 Approximate solution [H 3 O+] = 4. 2 x 10 -4 M, p. H = 3. 37 Exact Solution [H 3 O+] = [HCO 2 -] = 3. 4 x 10 -4 M [HCO 2 H] = 0. 0010 - 3. 4 x 10 -4 = 0. 0007 M p. H = 3. 47 20

21 Calculating Ka from the p. H The p. H of a 0. 10 M solution of formic acid, HCOOH, at 25°C is 2. 38. Calculate Ka formic acid at this temperature. To calculate Ka, we need all equilibrium concentrations. We can find [H 3 O+], which is the same as [HCOO−], from the p. H.
![22 Calculating Ka from p. H In table form: [HCOOH], M Initially 0. 10 22 Calculating Ka from p. H In table form: [HCOOH], M Initially 0. 10](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-22.jpg)
22 Calculating Ka from p. H In table form: [HCOOH], M Initially 0. 10 Change – 4. 2 10 -3 At Equilibrium 0. 10 – 4. 2 10 -3 = 0. 0958 = 0. 10 [H 3 O+], M [HCOO−], M 0 0 +4. 2 10 -3 4. 2 10 - 3
![23 Calculating Ka from p. H [4. 2 10 -3] Ka = [0. 10] 23 Calculating Ka from p. H [4. 2 10 -3] Ka = [0. 10]](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-23.jpg)
23 Calculating Ka from p. H [4. 2 10 -3] Ka = [0. 10] = 1. 8 10 -4
![24 Calculating Percent Ionization In the example: [A-]eq = [H 3 O+]eq = 4. 24 Calculating Percent Ionization In the example: [A-]eq = [H 3 O+]eq = 4.](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-24.jpg)
24 Calculating Percent Ionization In the example: [A-]eq = [H 3 O+]eq = 4. 2 10 -3 M [A-]eq + [HCOOH]eq = [HCOOH]initial = 0. 10 M

25 Calculating Percent Ionization % Ionization = 4. 2 10 -3 0. 10 100 = 4. 2%

26 Calculating p. H from Ka Calculate the p. H of a 0. 30 M solution of acetic acid, C 2 H 3 O 2 H, at 25°C. Ka for acetic acid at 25°C is 1. 8 10 -5. Is acetic acid more or less ionized than formic acid (Ka=1. 8 x 10 -4)?

27 Calculating p. H from Ka The equilibrium constant expression is:
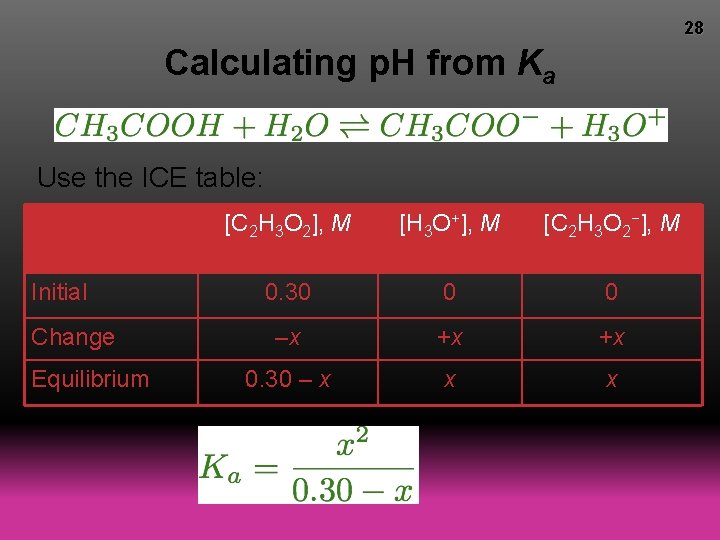
28 Calculating p. H from Ka Use the ICE table: Initial Change Equilibrium [C 2 H 3 O 2], M [H 3 O+], M [C 2 H 3 O 2−], M 0. 30 0 0 –x +x +x 0. 30 – x x x
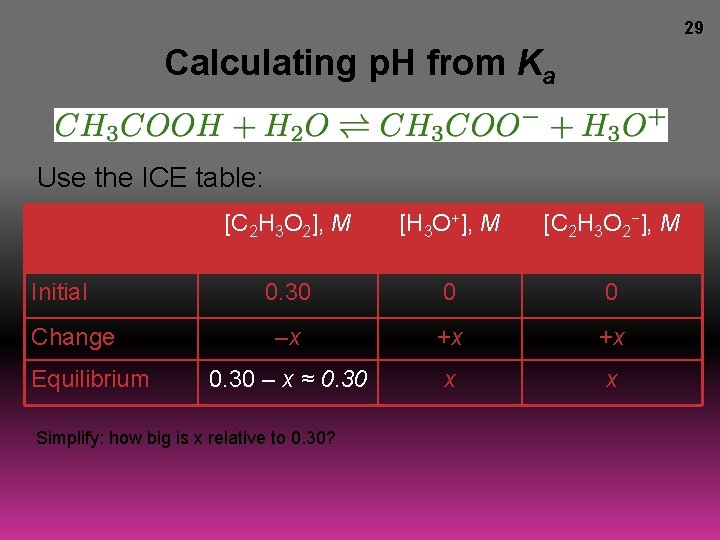
29 Calculating p. H from Ka Use the ICE table: Initial Change Equilibrium [C 2 H 3 O 2], M [H 3 O+], M [C 2 H 3 O 2−], M 0. 30 0 0 –x +x +x 0. 30 – x ≈ 0. 30 x x Simplify: how big is x relative to 0. 30?

30 Calculating p. H from Ka Now, (1. 8 10 -5) (0. 30) = x 2 5. 4 10 -6 = x 2 2. 3 10 -3 = x Check: is approximation ok?
![31 Calculating p. H from Ka p. H = –log [H 3 O+] p. 31 Calculating p. H from Ka p. H = –log [H 3 O+] p.](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-31.jpg)
31 Calculating p. H from Ka p. H = –log [H 3 O+] p. H = – log (2. 3 10− 3) p. H = 2. 64

HONORS ONLY! Equilibria Involving A Weak Base You have 0. 010 M NH 3. Calc. the p. H. NH 3 + H 2 O NH 4+ + OH- Kb = 1. 8 x 10 -5 Step 1. Define equilibrium concs. in ICE table [NH 3] [NH 4+] [OH-] initial 0. 010 0 0 change -x +x +x equilib 0. 010 - x x x 32

Equilibria Involving A Weak Base You have 0. 010 M NH 3. Calc. the p. H. NH 3 + H 2 O NH 4+ + OH- Kb = 1. 8 x 10 -5 Step 1. Define equilibrium concs. in ICE table [NH 3] [NH 4+] [OH-] initial 0. 010 0 0 change -x +x +x equilib 0. 010 - x x x 33

Equilibria Involving A Weak Base 34 You have 0. 010 M NH 3. Calc. the p. H. NH 3 + H 2 O NH 4+ + OH- Kb = 1. 8 x 10 -5 Step 2. Solve the equilibrium expression Assume x is small, so x = [OH-] = [NH 4+] = 4. 2 x 10 -4 M and [NH 3] = 0. 010 - 4. 2 x 10 -4 ≈ 0. 010 M The approximation is valid !

Equilibria Involving A Weak Base You have 0. 010 M NH 3. Calc. the p. H. NH 3 + H 2 O NH 4+ + OH- Kb = 1. 8 x 10 -5 Step 3. Calculate p. H [OH-] = 4. 2 x 10 -4 M so p. OH = - log [OH-] = 3. 37 Because p. H + p. OH = 14, p. H = 10. 63 35

36 p. H of Basic Solutions What is the p. H of a 0. 15 M solution of NH 3? [NH 4+] [OH−] Kb = = 1. 8 10 -5 [NH 3]
![37 p. H of Basic Solutions Tabulate the data. [NH 3], M Initial Equilibrium 37 p. H of Basic Solutions Tabulate the data. [NH 3], M Initial Equilibrium](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-37.jpg)
37 p. H of Basic Solutions Tabulate the data. [NH 3], M Initial Equilibrium [NH 4+], M [OH−], M 0. 15 0 0 0. 15 - x 0. 15 x x Simplify: how big is x relative to 0. 15?

p. H of Basic Solutions 2 (x) 1. 8 10 -5 = (0. 15) (1. 8 10 -5) (0. 15) = x 2 2. 7 10 -6 = x 2 1. 6 10 -3 = x 2 Check: is approximation ok? Ka / i > 500 38
![39 p. H of Basic Solutions Therefore, [OH–] = 1. 6 10 -3 M 39 p. H of Basic Solutions Therefore, [OH–] = 1. 6 10 -3 M](http://slidetodoc.com/presentation_image_h2/f125273266745f24dd4972f55ef950f9/image-39.jpg)
39 p. H of Basic Solutions Therefore, [OH–] = 1. 6 10 -3 M p. OH = –log (1. 6 10 -3) p. OH = 2. 80 p. H = 14. 00 – 2. 80 p. H = 11. 20

40 PRACTICE EXERCISES 1. Niacin, a B vitamin, has the following structure: A 0. 020 M solution of niacin has a p. H of 3. 26. (a) What percentage of the acid is ionized in this solution? Answer: (a) 2. 7%, (b) What is the acid-dissociation constant, Ka, for niacin? Answer: 1. 5 10– 5 2. What is the p. H of (a) a 0. 028 M solution of Na. OH ANSWER: p. H (Na. OH)=11. 04; (b) a 0. 0011 M solution of Ca(OH)2? What percentage of the bases are ionized? Answer: p. H= 11. 34 3. Calculate the percentage of HF molecules ionized in a 0. 10 M HF solution Answer: HF: 7. 8%,

Types of Acid/Base Reactions: Summary 41

42 What? Why? An anion that is the conjugate base of a strong acid does not affect p. H. = very weak base An anion that is the conjugate base of a weak acid increases p. H. = strong base A cation that is the conjugate acid of a weak base decreases p. H. = strong acid Cations of the strong Arrhenius bases (Na+, Ca 2+) do not affect p. H. = very weak acid (not really acidic at all) Other metal ions cause a decrease = moderate in p. H. (cations) Weak acid + weak bases Depends on Ka and Kb

43 Factors Affecting Acid Strength In oxyacids, in which an OH is bonded to another atom, Y, the more electronegative Y is, the more acidic the acid.

p. H paper 44

45 p. H meter • Tests the voltage of the electrolyte • Converts the voltage to p. H • Very cheap, accurate • Must be calibrated with a buffer solution
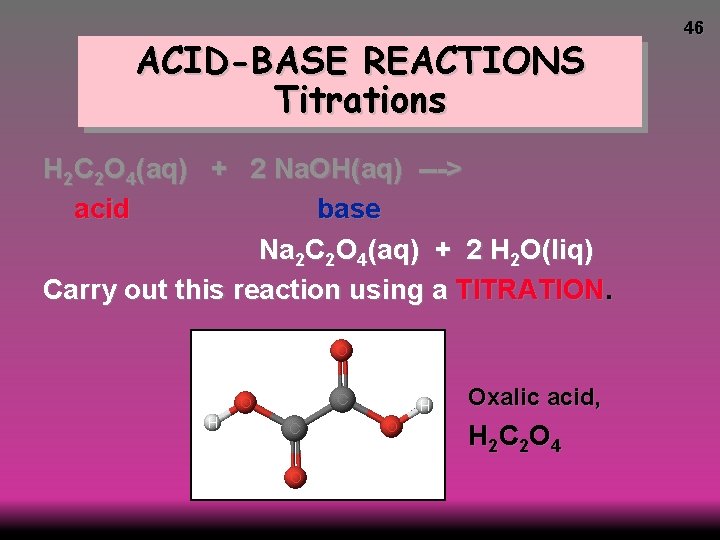
ACID-BASE REACTIONS Titrations H 2 C 2 O 4(aq) + 2 Na. OH(aq) ---> acid base Na 2 C 2 O 4(aq) + 2 H 2 O(liq) Carry out this reaction using a TITRATION. Oxalic acid, H 2 C 2 O 4 46

Setup for titrating an acid with a base 47

48 Titration 1. Add solution from the buret. 2. Reagent (base) reacts with compound (acid) in solution in the flask. 3. Indicator shows when exact stoichiometric reaction has occurred. (Acid = Base) This is called NEUTRALIZATION.

LAB PROBLEM #1: Standardize a solution of Na. OH — i. e. , accurately determine its concentration. 35. 62 m. L of Na. OH is neutralized with 25. 2 m. L of 0. 0998 M HCl by titration to an equivalence point. What is the concentration of the Na. OH? 49

50 PROBLEM: You have 50. 0 m. L of 3. 0 M Na. OH and you want 0. 50 M Na. OH. What do you do? Add water to the 3. 0 M solution to lower its concentration to 0. 50 M Dilute the solution!
51 PROBLEM: You have 50. 0 m. L of 3. 0 M Na. OH and you want 0. 50 M Na. OH. What do you do? But how much water do we add?

52 PROBLEM: You have 50. 0 m. L of 3. 0 M Na. OH and you want 0. 50 M Na. OH. What do you do? How much water is added? The important point is that ---> moles of Na. OH in ORIGINAL solution = moles of Na. OH in FINAL solution

PROBLEM: You have 50. 0 m. L of 3. 0 M Na. OH and you want 0. 50 M Na. OH. What do you do? Amount of Na. OH in original solution = M • V = (3. 0 mol/L)(0. 050 L) = 0. 5 M Na. OH X V Amount of Na. OH in final solution must also = 0. 15 mol Na. OH Volume of final solution = (0. 15 mol Na. OH) / (0. 50 M) = 0. 30 L or 300 m. L 53
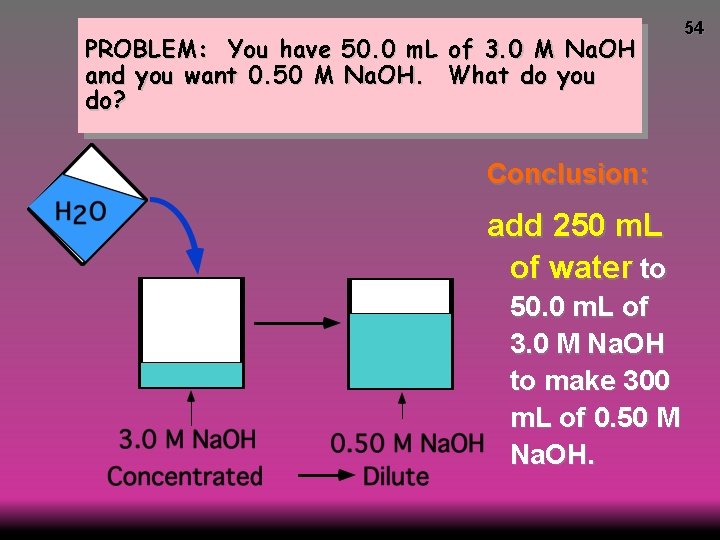
PROBLEM: You have 50. 0 m. L of 3. 0 M Na. OH and you want 0. 50 M Na. OH. What do you do? Conclusion: add 250 m. L of water to 50. 0 m. L of 3. 0 M Na. OH to make 300 m. L of 0. 50 M Na. OH. 54
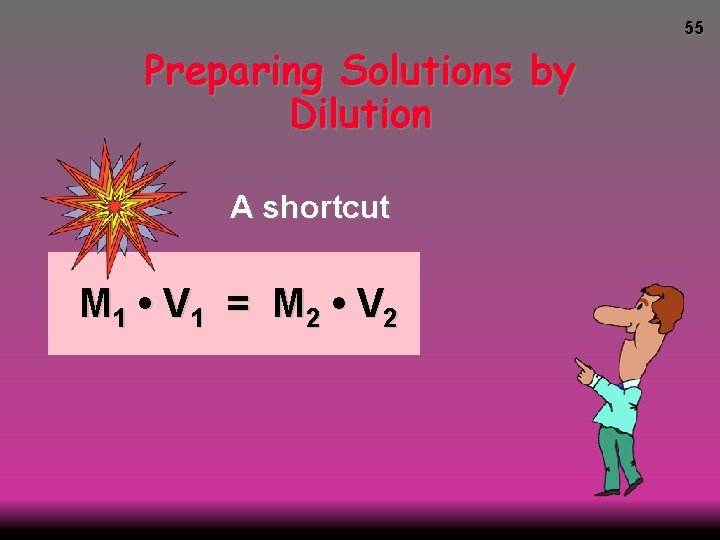
55 Preparing Solutions by Dilution A shortcut M 1 • V 1 = M 2 • V 2
You try this dilution problem • You have a stock bottle of hydrochloric acid, which is 12. 1 M. You need 400. m. L of 0. 10 M HCl. How much of the acid and how much water will you need? 56

INDICATORS low p. H: equilibrium pushed left = colour 1 high p. H: equilibrium pushed right = colour 2 57
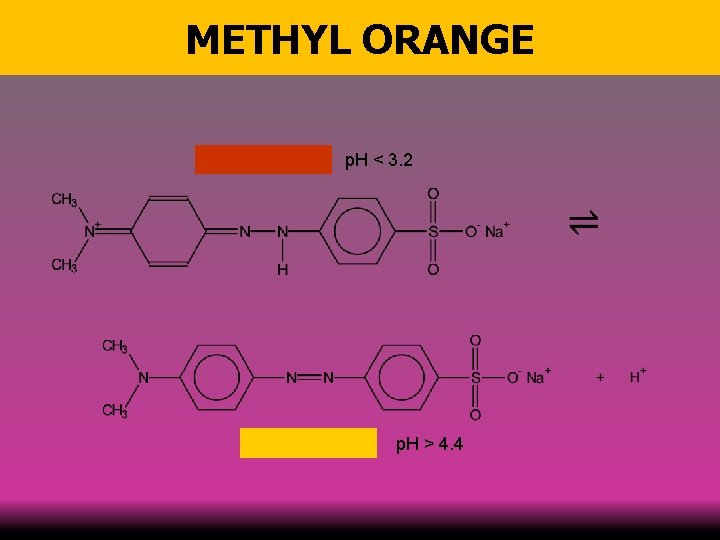
METHYL ORANGE p. H < 3. 2 p. H > 4. 4 58

METHYL ORANGE acid end-point 59 alkali
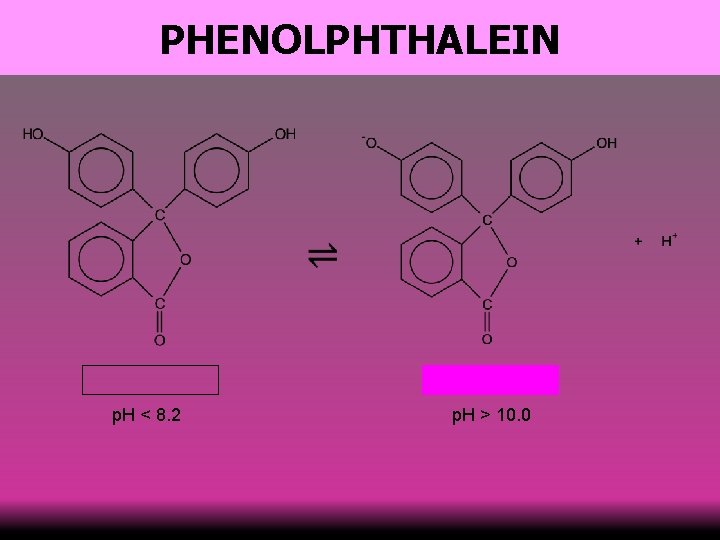
PHENOLPHTHALEIN p. H < 8. 2 p. H > 10. 0 60

PHENOLPHTHALEIN acid alkali end-point acid 61 alkali

INDICATORS 62 Indicator colour at low p. H range of colour change colour at high p. H methyl orange red 3. 2 – 4. 4 orange 8. 2 – 10. 0 purple phenolphthalein colourless
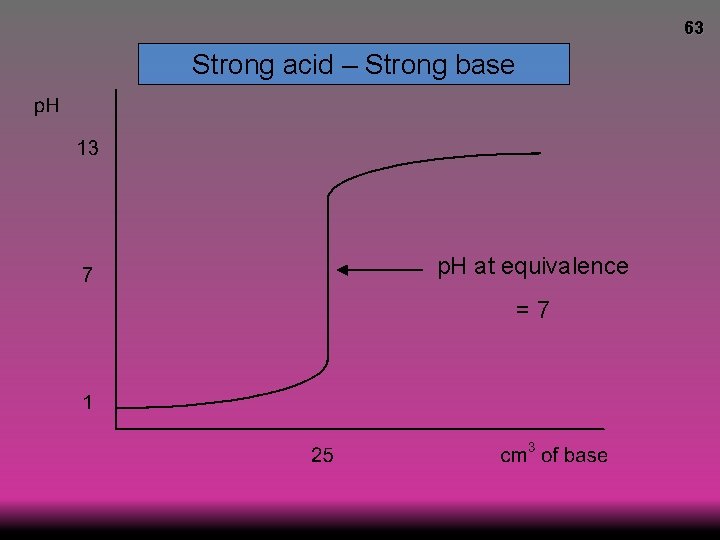
63 Strong acid – Strong base p. H at equivalence =7

64 Strong acid – Strong base
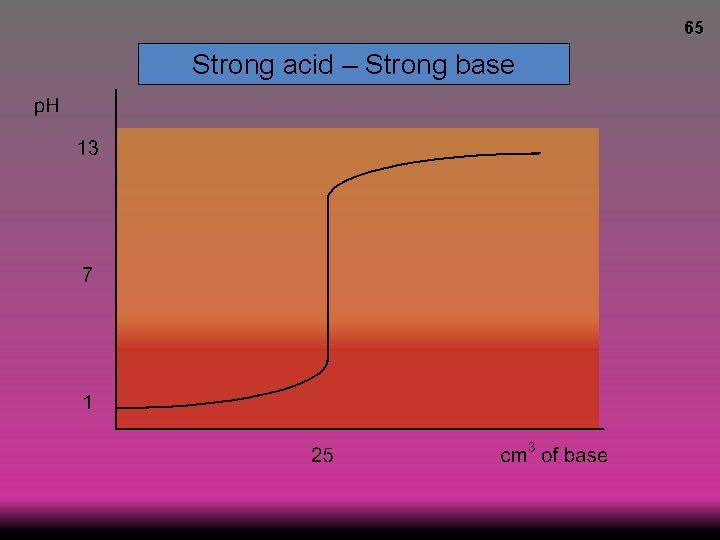
65 Strong acid – Strong base
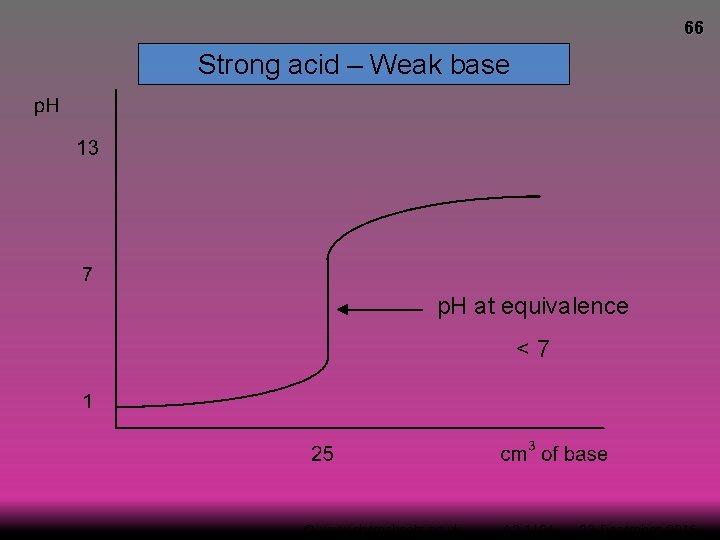
66 Strong acid – Weak base p. H at equivalence <7 © www. chemsheets. co. uk A 2 1104 23 -December-2016
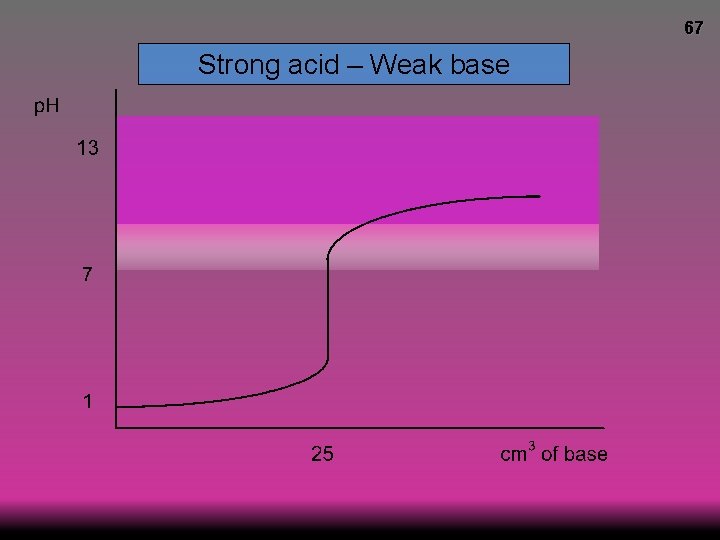
67 Strong acid – Weak base
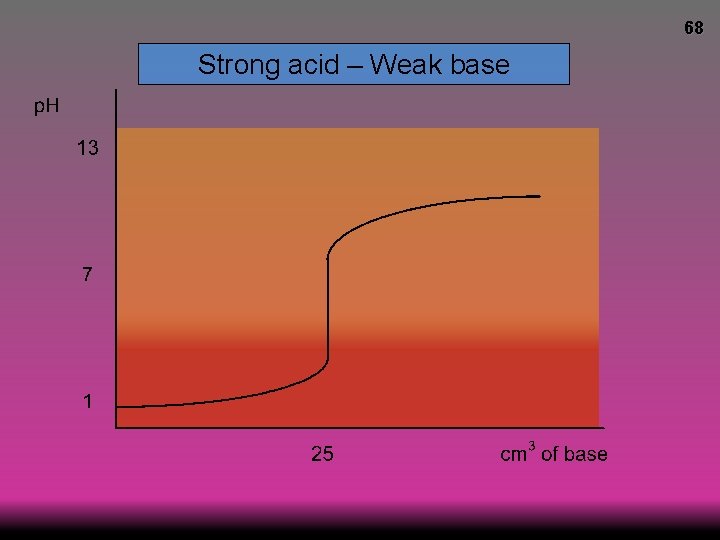
68 Strong acid – Weak base
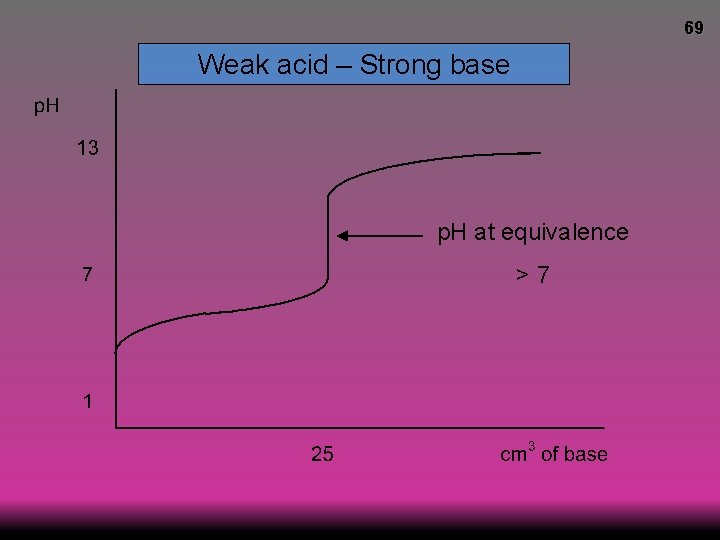
69 Weak acid – Strong base p. H at equivalence >7
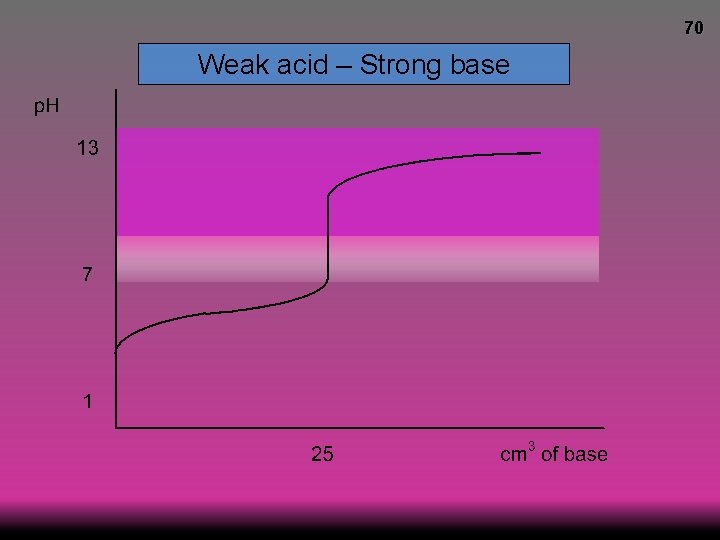
70 Weak acid – Strong base
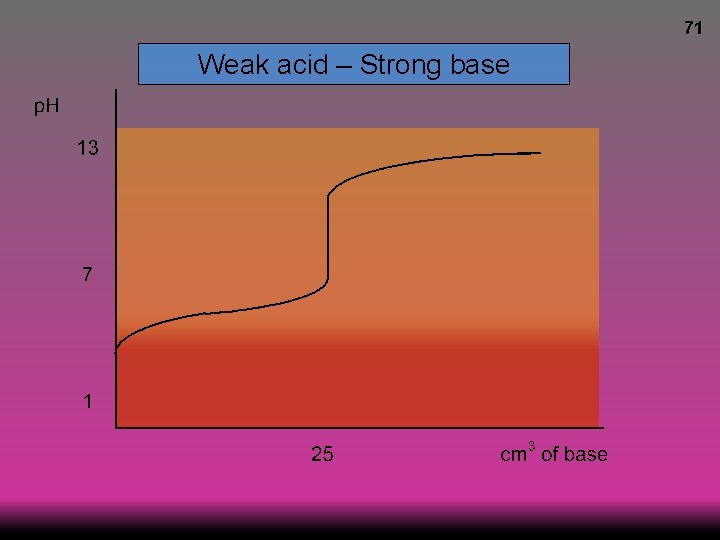
71 Weak acid – Strong base

72 Weak acid – Weak base p. H at equivalence depends on relative strength of acid and base

73 Weak acid – Weak base

74 Weak acid – Weak base © www. chemsheets. co. uk A 2 1104 23 -December-2016

75 SUMMARY

76 SUMMARY

77 SUMMARY

78

79 Key Points about Buffered Solutions • Buffered Solution – resists a change in p. H. • They are weak acids or bases containing a common ion. • After addition of strong acid or base, deal with stoichiometry first, then the equilibrium. Copyright © Cengage Learning. All rights reserved 79

80 Buffers • Solutions of a weak conjugate acid–base pair that resist drastic changes in p. H are called buffers. • These solutions contain relatively high concentrations (10– 3 M or more) of both the acid and base. Their concentrations are approximately equal.

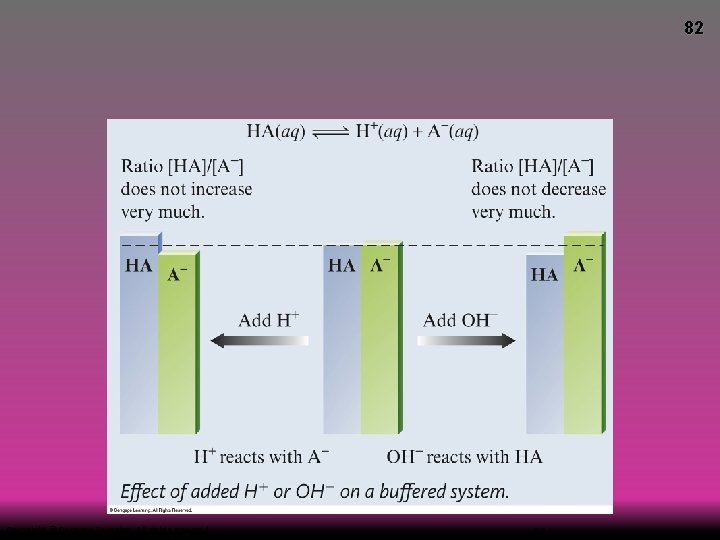
81 Ways to Make a Buffer 1) Mix a weak acid and a salt of its conjugate base or a weak base and a salt of its conjugate acid. 2) Add strong acid and partially neutralize a weak base or add strong base and partially neutralize a weak acid.
82 Copyright © Cengage Learning. All rights reserved 82

83 Calculating the p. H of a Buffer • For a weak acid: Ka = [H+][A–]/[HA] • Take –log of both sides: –log Ka = –log[H+] + –log([A–]/[HA]) • Rearrange: –log[H+] = –log Ka +log([A–]/[HA]) • Which is: p. H = p. Ka + log([A–]/[HA]) • This equation is known as the Henderson–Hasselbalch equation. This applies only to buffers.

84 Henderson–Hasselbalch Equation • For a particular buffering system (conjugate acid–base pair), all solutions that have the same ratio [A–] / [HA] will have the same p. H. Copyright © Cengage Learning. All rights reserved 84

85 Calculating the p. H of a Buffer What is the p. H of a buffer that is 0. 12 M in lactic acid [CH 3 CH(OH)COOH, or HC 3 H 5 O 3] and 0. 10 M in sodium lactate [CH 3 CH(OH)COONa or Na. C 3 H 5 O 3]? For lactic acid, Ka = 1. 4 × 10– 4. Solution Analyze We are asked to calculate the p. H of a buffer containing lactic acid (HC 3 H 5 O 3) and its conjugate base, the lactate ion (C 3 H 5 O 3–). Plan We will first determine the p. H using the method learned previously. Because HC 3 H 5 O 3 is a weak electrolyte and Na. C 3 H 5 O 3 is a strong electrolyte, the major species in solution are HC 3 H 5 O 3, Na+, and C 3 H 5 O 3–. The Na+ ion is a spectator ion. The HC 3 H 5 O 3/C 3 H 5 O 3– conjugate acid–base pair determines [H+] and, thus, p. H; [H+] can be determined using the acid-dissociation equilibrium of lactic acid.

86 Calculating the p. H of a Buffer - continued Solve The initial and equilibrium concentrations of the species involved in this equilibrium are The equilibrium concentrations are governed by the equilibrium expression: Because Ka is small and a common is present, we expect x to be small relative to either 0. 12 or 0. 10 M. Thus, our equation can be simplified to give

87 Calculating the p. H of a Buffer - continued Solving for x gives a value that justifies our approximation: Then, we can solve for p. H: Alternatively, we can use the Henderson–Hasselbalch equation with the initial concentrations of acid and base to calculate p. H directly:

88 Calculating p. H When the Henderson–Hasselbalch Equation May Not Be Accurate Calculate the p. H of a buffer that initially contains 1. 00 × 10– 3 M CH 3 COOH and 1. 00 × 10– 4 M CH 3 COONa in the following two ways: (i) using the Henderson– Hasselbalch equation and (ii) making no assumptions about quantities (which means you will need to use the quadratic equation). The Ka of CH 3 COOH is 1. 80 × 10– 5. Solution Analyze We are asked to calculate the p. H of a buffer two different ways. We know the initial concentrations of the weak acid and its conjugate base, and the Ka of the weak acid. Plan We will first use the Henderson–Hasselbalch equation, which relates p. Ka and ratio of acid–base concentrations to the p. H. This will be straightforward. Then, we will redo the calculation making no assumptions about any quantities, which means we will need to write out the initial/change/equilibrium concentrations, as we have done before. In addition, we will need to solve for quantities using the quadratic equation (since we cannot make assumptions about unknowns being small).

89 Calculating p. H When the Henderson–Hasselbalch Equation May Not Be Accurate Solve (i) equation is We know the Ka of the acid (1. 8 × 10– 5), so we know p. Ka (p. Ka = –log Ka = 4. 742. We know the initial concentrations of the base, sodium acetate, and the acid, acetic acid, which we will assume are the same as the equilibrium concentrations. The Henderson–Hasselbalch p. H = 4. 74 – 1. 00 = 3. 74

90 Calculating p. H When the Henderson–Hasselbalch Equation May Not Be Accurate (ii)Now we will redo the calculation, without making any assumptions at all. We will solve for x, which represents the H+ concentration at equilibrium, in order to calculate p. H.

91 Calculating p. H When the Henderson–Hasselbalch Equation May Not Be Accurate Comment In the previous calculation, the calculated p. H is the same whether we solve exactly using the quadratic equation or make the simplifying assumption that the equilibrium concentrations of acid and base are equal to their initial concentrations. The simplifying assumption works because the concentrations of the acid–base conjugate pair are both a thousand times larger than Ka. In this example, the acid–base conjugate pair concentrations are only 10– 100 as large as Ka. Therefore, we cannot assume that x is small compared to the initial concentrations (that is, that the initial concentrations are essentially equal to the equilibrium concentrations). The best answer to this example is p. H = 4. 06, obtained without assuming x is small. Thus we see that the assumptions behind the Henderson–Hasselbalch equation becomes less accurate as the acid/base becomes stronger and/or its concentration becomes smaller.

92 Buffered Solution Characteristics • Buffers contain relatively large concentrations of a weak acid and corresponding conjugate base. • Added H+ reacts to completion with the weak base. • Added OH- reacts to completion with the weak acid. Copyright © Cengage Learning. All rights reserved 92

93 Buffered Solution Characteristics • The p. H in the buffered solution is determined by the ratio of the concentrations of the weak acid and weak base. As long as this ratio remains virtually constant, the p. H will remain virtually constant. This will be the case as long as the concentrations of the buffering materials (HA and A– or B and BH+) are large compared with amounts of H+ or OH– added. Copyright © Cengage Learning. All rights reserved 93

94 Addition of a Strong Acid or a Strong Base to a Buffer 1) Adding of the strong acid or base is a neutralization reaction; calculate like stoichiometry problem to find [HA] and [A–] when all of the added acid or base reacts. 2) Use the Henderson–Hasselbalch (HH) equation to find p. H or use ICE chart if [acid-base pair] is too low. © 2015 Pearson Education, Inc.

95 • A buffer is made by adding 0. 300 mol HC 2 H 3 O 2 and 0. 300 mol Na. C 2 H 3 O 2 to enough water to make 1. 00 L of solution. Calculate the p. H after 0. 020 mol of Na. OH is added. (Ka = 1. 8 x 10 -5) 1) CH 3 COOH(aq) + OH- → H 2 O(l) + CH 3 COO–(aq) [CH 3 COOH] [OH–] [CH 3 COO–] Before reaction (mol) 0. 300 0. 020 0. 300 Change (mol) – 0. 020 (LR) +0. 020 After reaction (mol) 0. 280 0 0. 320

96 Example (completed) 2) Use the Henderson–Hasselbalch equation: p. H = p. Ka + log([A–]/[HA) Since this is a buffer, the volume for each concentration is the same, so the ratio of molarity can be calculated using a ratio of moles. p. H = p. Ka + log (n. HA/n. A–) p. H = 4. 74 + log(0. 320/0. 280) = 4. 80

97 AP Learning Objectives, Margin Notes and References • Learning Objectives • • LO 6. 1 The student is able to, given a set of experimental observations regarding physical, chemical, biological, or environmental processes that are reversible, construct an explanation that connects the observations to the reversibility of the underlying chemical reactions or processes. LO 6. 20 The student can identify a solution as being a buffer solution and explain the buffer mechanism in terms of the reactions that would occur on addition of acid or base. • Additional AP References • LO 6. 20 (see APEC 16, “Acids, Bases and Buffers”)

98 • The amount of protons or hydroxide ions the buffer can absorb without a significant change in p. H. • Determined by the magnitudes of [HA] and [A–]. • A buffer with large capacity contains large concentrations of the buffering components. • Using the Henderson–Hasselbalch equation, p. H will be the same for a conjugate acid– base pair of 1 M each or 0. 1 M each; however, the buffer which is 1 M can Copyright © Cengage Learning. All rights reserved 98

99 p. H Range • The range of p. H values over which a buffer system works effectively • Optimal p. H: where p. H = p. Ka([HA] = [A–]) • If one concentration is more than 10 times the other, the buffering action is poor; this means that the p. H range of a buffer is usually ± 1 p. H unit from p. Ka.

100 Choosing a Buffer • p. Ka of the weak acid to be used in the buffer should be as close as possible to the desired p. H. Copyright © Cengage Learning. All rights reserved 100
- Slides: 100