Stress Ulcer Prophylaxis As Clear as Mud Keith


























- Slides: 46

Stress Ulcer Prophylaxis “As Clear as Mud” Keith M. Olsen, Pharm. D, FCCP, FCCM Dean and Professor College of Pharmacy University of Arkansas for Medical Sciences Little Rock, AR kolsen@uams. edu

Disclosures • I am the Vice-Chancellor for the American College of Critical Care Medicine (ACCM) and therefore have knowledge on guidelines published and not published by the Society of Critical Care Medicine • I am one of the developers of Zegerid© (omeprazole/sodium bicarbonate) • I have published research on stress-ulcer prophylaxis and acid-suppressive therapy in the intensive care unit

Objectives At completion of this seminar on stress ulcer prophylaxis (SUP), the pharmacist will able to: üIdentify patients likely to benefit from acid suppression. üIdentify patients who may benefit from SUP. üDescribe the relationship between acid suppressive drugs and adverse events. üList the consequences of inappropriate SUP.

ICU Indications For Acid Suppression • Upper gastrointestinal (non-variceal) bleeding • Stress related mucosal damage

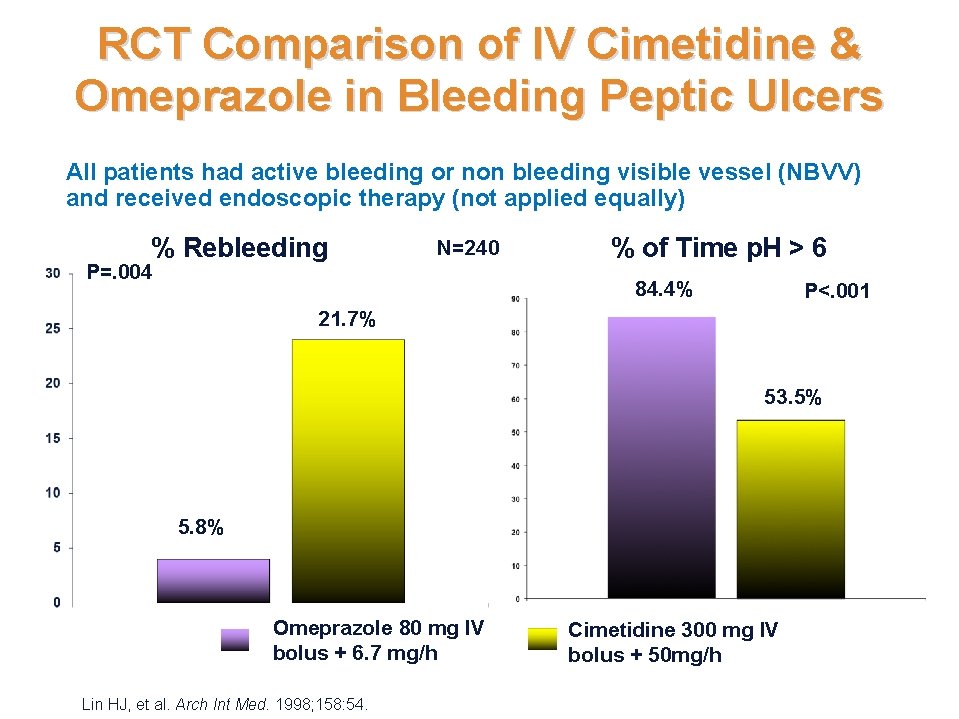
RCT Comparison of IV Cimetidine & Omeprazole in Bleeding Peptic Ulcers All patients had active bleeding or non bleeding visible vessel (NBVV) and received endoscopic therapy (not applied equally) % Rebleeding N=240 P=. 004 % of Time p. H > 6 84. 4% P<. 001 21. 7% 53. 5% 5. 8% Omeprazole 80 mg IV bolus + 6. 7 mg/h Lin HJ, et al. Arch Int Med. 1998; 158: 54. Cimetidine 300 mg IV bolus + 50 mg/h

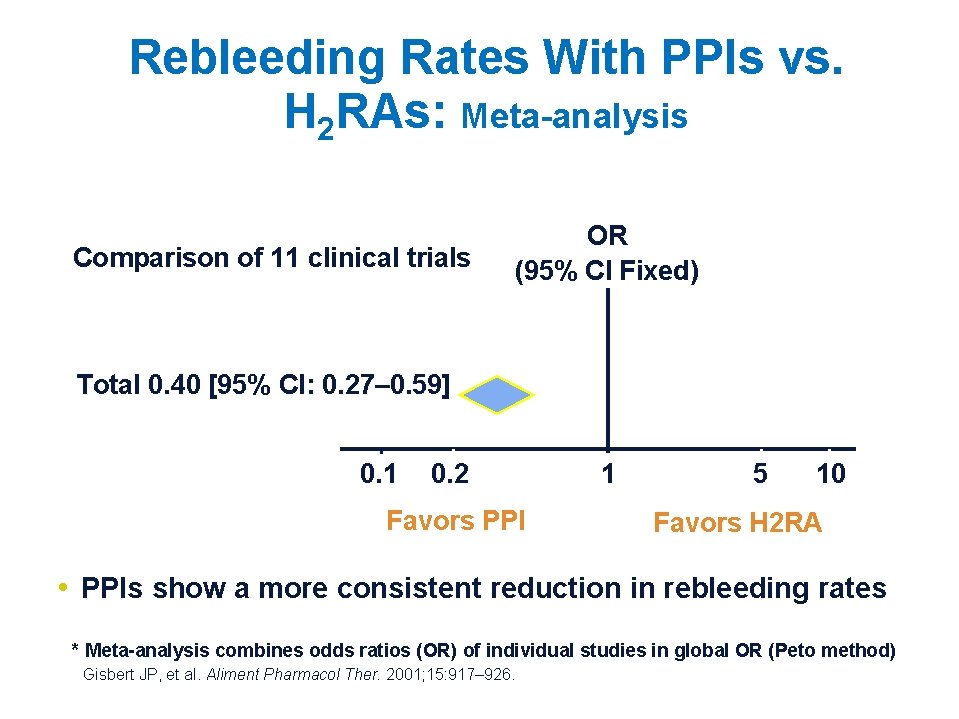
Rebleeding Rates With PPIs vs. H 2 RAs: Meta-analysis Comparison of 11 clinical trials OR (95% CI Fixed) Total 0. 40 [95% CI: 0. 27– 0. 59] 0. 1 0. 2 Favors PPI 1 5 10 Favors H 2 RA • PPIs show a more consistent reduction in rebleeding rates * Meta-analysis combines odds ratios (OR) of individual studies in global OR (Peto method) Gisbert JP, et al. Aliment Pharmacol Ther. 2001; 15: 917– 926.

JAMA Int Med 2014


Guidelines for Stress Ulcer Prophylaxis in Adult Critically Ill Patients Henry Cohen, Pharm. D, MSc, Robert Mac. Laren, Pharm. D, MPH, Robert G. Martindale, MD, Ph. D, Lena M. Napolitano, MD, Douglas F. Naylor Jr. , MD, Jill A. Rebuck, Pharm. D, H. David Reines, MD, Richard H. Savel, MD, Anne S. Pohlman, RN, MSN, Joseph F. Dasta, MSc Crit Care Med 2015; in review H 2 RAs PPIs or H 2 RAs

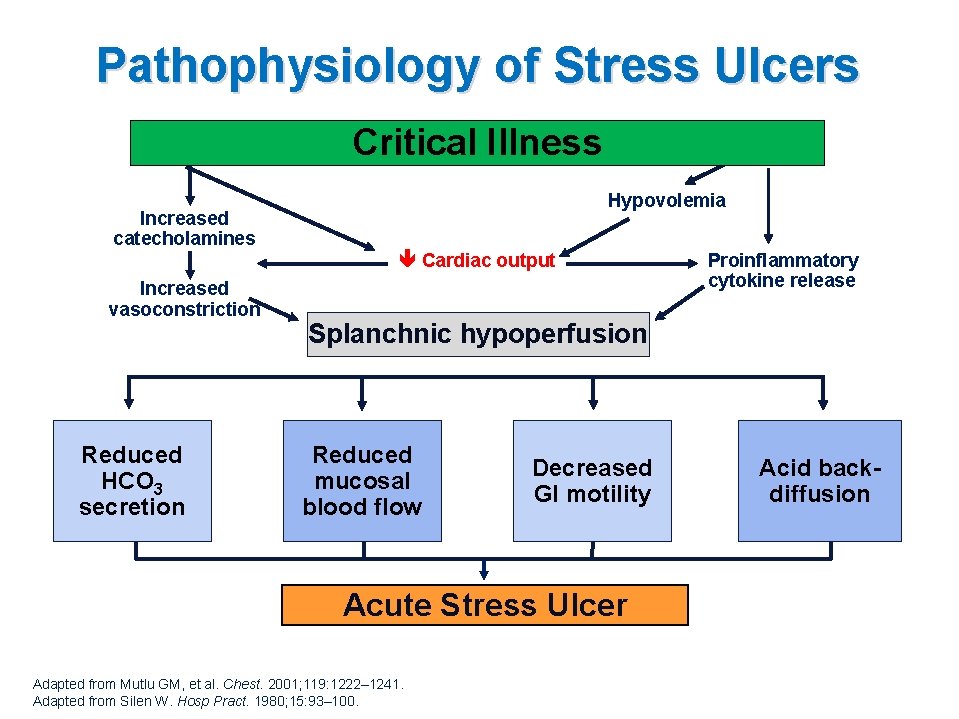
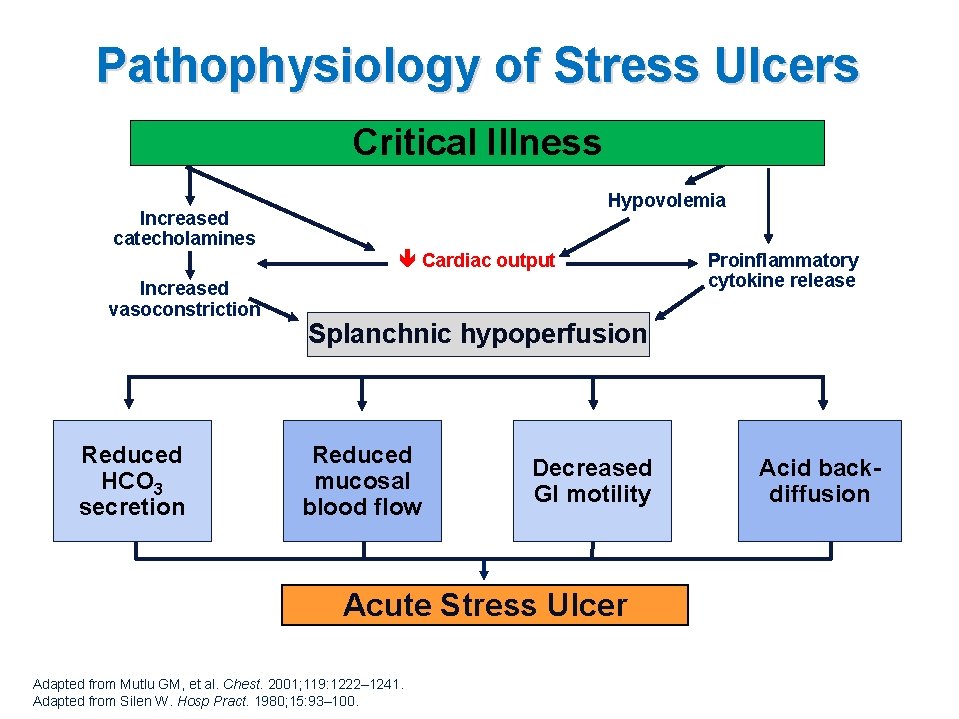
Pathophysiology of Stress Ulcers Critical Illness Hypovolemia Increased catecholamines Cardiac output Increased vasoconstriction Reduced HCO 3 secretion Proinflammatory cytokine release Splanchnic hypoperfusion Reduced mucosal blood flow Decreased GI motility Acute Stress Ulcer Adapted from Mutlu GM, et al. Chest. 2001; 119: 1222– 1241. Adapted from Silen W. Hosp Pract. 1980; 15: 93– 100. Acid backdiffusion

Stress Ulcer Prophylaxis Ideal Pharmacologic Agent • Effective • Safe • Minimal interactions • Diverse administration options • Economically justified

“We are what we repeatedly do. Excellence, therefore, is not an act, but a habit” Aristotle

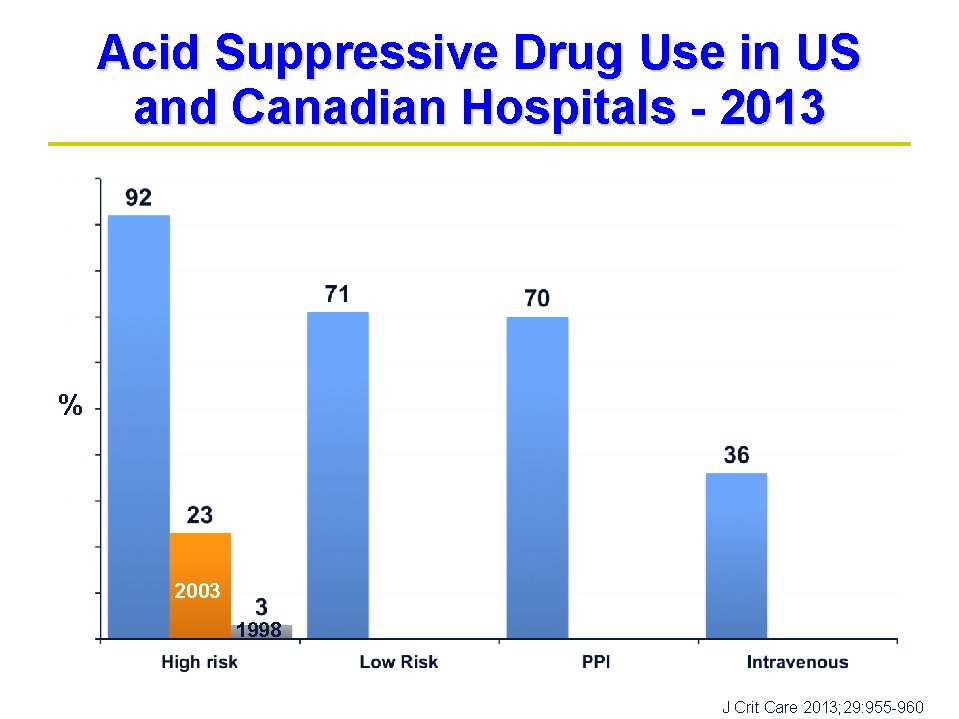
Acid Suppressive Drug Use in US and Canadian Hospitals - 2013 % 2003 1998 J Crit Care 2013; 29: 955 -960

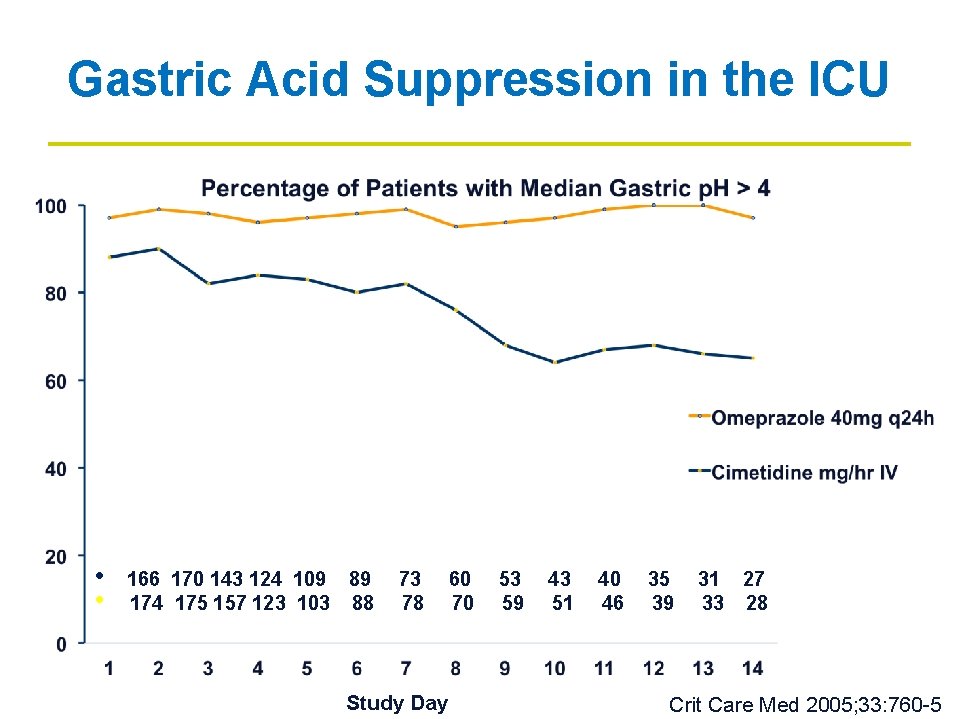
Gastric Acid Suppression in the ICU % # of Patients • • 166 170 143 124 109 89 73 60 53 43 40 35 31 27 174 175 157 123 103 88 78 70 59 51 46 39 33 28 Study Day Crit Care Med 2005; 33: 760 -5

Proton Pump Inhibitor Gastric Acid Suppression: Oral vs IV Olsen KM, Devlin J Alimen Pcol Ther 2011

Medical ICU Case Mix Heterogeneous vs. Homogeneous Gastric Acid Tube Feedings Sepsis AKI Mechanical Ventilation GI Blood Flow HOMOGENEOUS Trauma Atherosclerotic Vascular Disease Surgery Hypotension Age

Pathophysiology of Stress Ulcers Critical Illness Hypovolemia Increased catecholamines Cardiac output Increased vasoconstriction Reduced HCO 3 secretion Proinflammatory cytokine release Splanchnic hypoperfusion Reduced mucosal blood flow Decreased GI motility Acute Stress Ulcer Adapted from Mutlu GM, et al. Chest. 2001; 119: 1222– 1241. Adapted from Silen W. Hosp Pract. 1980; 15: 93– 100. Acid backdiffusion

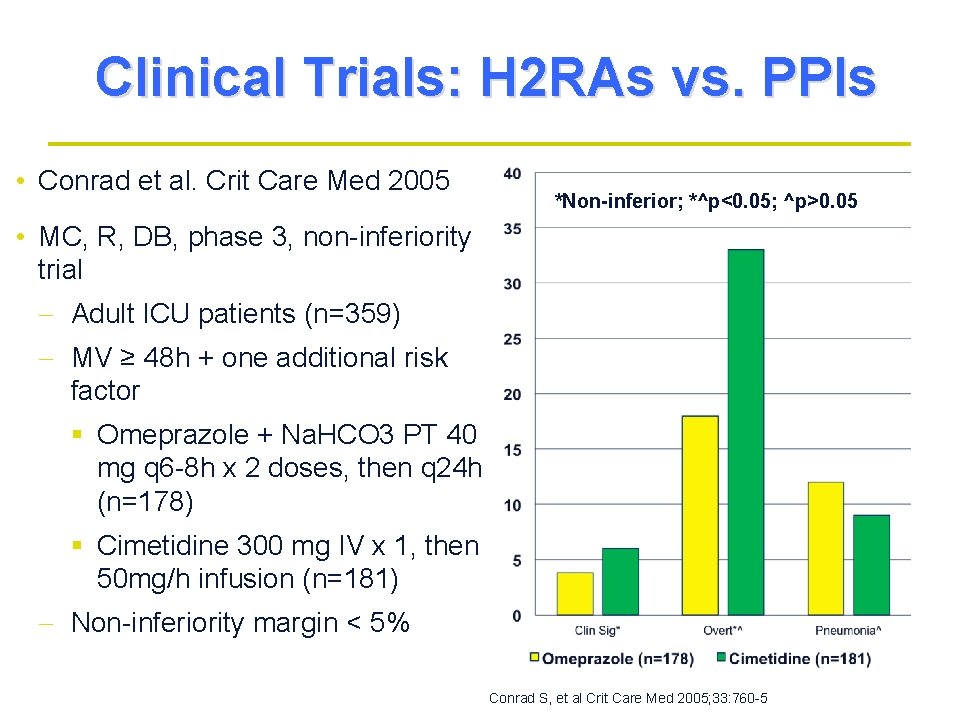
Clinical Trials: H 2 RAs vs. PPIs • Conrad et al. Crit Care Med 2005 *Non-inferior; *^p<0. 05; ^p>0. 05 • MC, R, DB, phase 3, non-inferiority trial - Adult ICU patients (n=359) - MV ≥ 48 h + one additional risk factor § Omeprazole + Na. HCO 3 PT 40 mg q 6 -8 h x 2 doses, then q 24 h (n=178) § Cimetidine 300 mg IV x 1, then 50 mg/h infusion (n=181) - Non-inferiority margin < 5% Conrad S, et al Crit Care Med 2005; 33: 760 -5

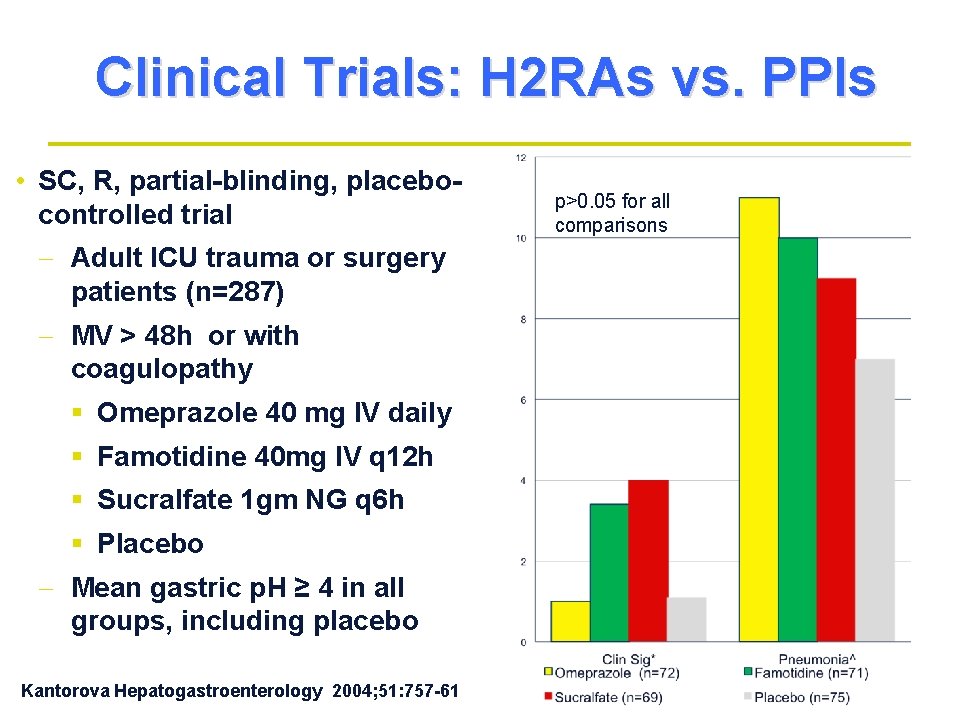
Clinical Trials: H 2 RAs vs. PPIs • SC, R, partial-blinding, placebocontrolled trial - Adult ICU trauma or surgery patients (n=287) - MV > 48 h or with coagulopathy § Omeprazole 40 mg IV daily § Famotidine 40 mg IV q 12 h § Sucralfate 1 gm NG q 6 h § Placebo - Mean gastric p. H ≥ 4 in all groups, including placebo Kantorova Hepatogastroenterology 2004; 51: 757 -61 p>0. 05 for all comparisons

RCTs: PPIs vs. H 2 RA • 14 randomized controlled trials (RCTs) • Wide differences in study design • Many non-inferiority trials • Different risk factors • Different definitions of bleeding and significant bleeding

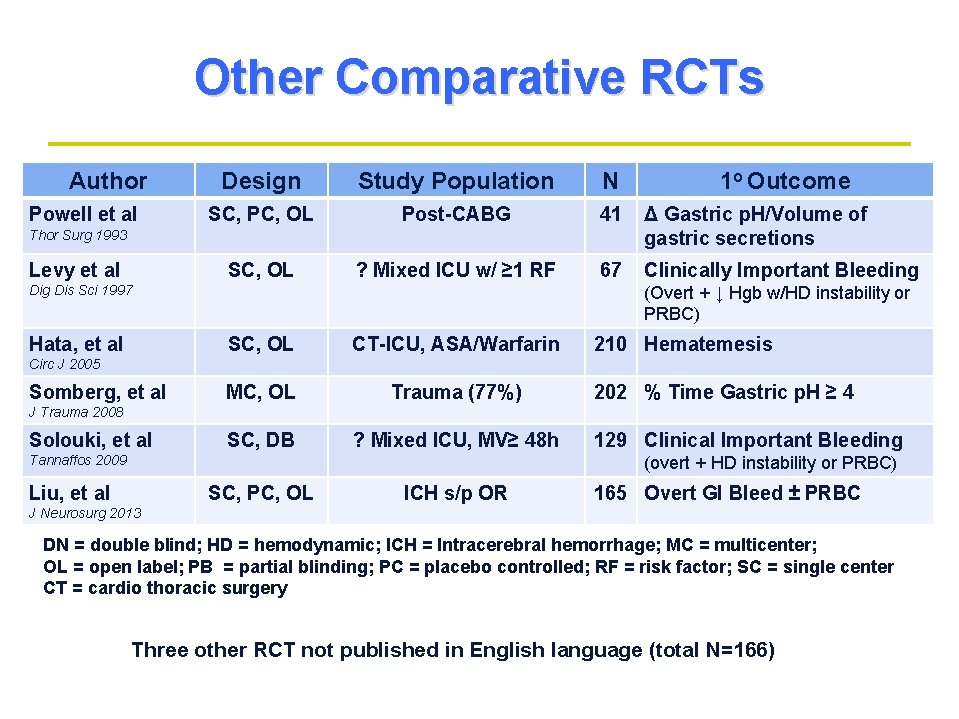
Other Comparative RCTs Author Powell et al Design Study Population N SC, PC, OL Post-CABG 41 Δ Gastric p. H/Volume of gastric secretions SC, OL ? Mixed ICU w/ ≥ 1 RF 67 Clinically Important Bleeding Thor Surg 1993 Levy et al Dig Dis Sci 1997 Hata, et al 1 o Outcome (Overt + ↓ Hgb w/HD instability or PRBC) SC, OL CT-ICU, ASA/Warfarin MC, OL Trauma (77%) SC, DB ? Mixed ICU, MV≥ 48 h 210 Hematemesis Circ J 2005 Somberg, et al 202 % Time Gastric p. H ≥ 4 J Trauma 2008 Solouki, et al Tannaffos 2009 129 Clinical Important Bleeding (overt + HD instability or PRBC) Liu, et al SC, PC, OL ICH s/p OR 165 Overt GI Bleed ± PRBC J Neurosurg 2013 DN = double blind; HD = hemodynamic; ICH = Intracerebral hemorrhage; MC = multicenter; OL = open label; PB = partial blinding; PC = placebo controlled; RF = risk factor; SC = single center CT = cardio thoracic surgery Three other RCT not published in English language (total N=166)

Meta-Analyses Pongprasobchai • et al. 2009 • 3 RCT, n=569 Clinically important bleed = OR 0. 42 (0. 2, 0. 91) • No difference in nosocomial pneumonia Lin, et al 2010 • 7 RCT (2 as abstracts only), n=936 • Upper GI bleed – Risk difference -0. 04 (-0. 09, 0. 01); I 2=66% • No difference in nosocomial pneumonia or ICU mortality Barkun, et al. 2012 • 13 RCT (5 as abstracts only). N=1587 • Clinically significant bleeding – OR 0. 3 (0. 17, 0. 54); I 2=0% • No difference in pneumonia, ICU length of stay, ICU mortality J Med Assoc Thai 2009; 92; 632; Crit Care Med 2010; 38: 1197; Am J Gastroenterol 2012; 107:

Crit Care Med 2013; 41: 693 Characteristic Criteria Types of studies included Randomized, Parallel Control Study population Adult patients, Medical or surgical ICU Intervention Any PPI therapy Control Any H 2 RA therapy Primary outcome GI bleed (clinically important vs. overt) Secondary outcome Pneumonia, ICU-mortality, ICU LOS, C. difficile Methodologic quality Cochrane risk of bias tool Analysis Random effect model, a prior subgroup analysis, statistical heterogeneity with I 2 statistic 14 randomized controlled trials (n=1720)

Clinically Important Bleeding ND ND NNT = 78 Risk Difference -0. 026 (00. 54, 0. 001) Test for heterogeneity: I 2 = 52% A= only available as abstract; ND = no definition provided for clinically important bleed NNT = number needed to treat Crit Care Med 2013; 41: 693

Influence of Risk of Bias Crit Care Med 2013; 41: 693

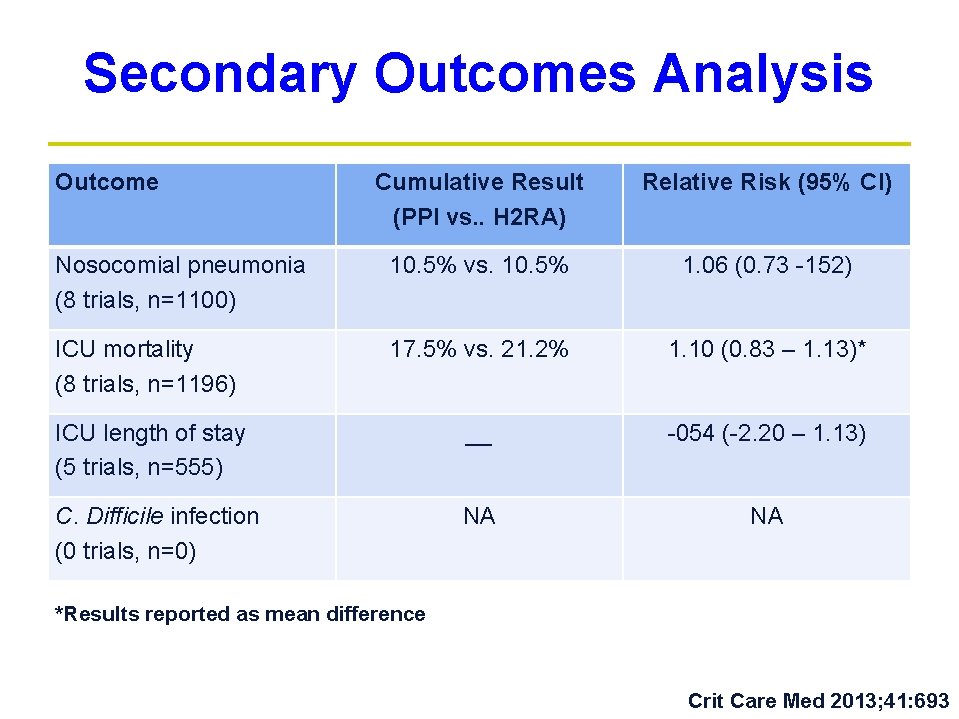
Secondary Outcomes Analysis Outcome Cumulative Result (PPI vs. . H 2 RA) Relative Risk (95% CI) Nosocomial pneumonia (8 trials, n=1100) 10. 5% vs. 10. 5% 1. 06 (0. 73 -152) ICU mortality (8 trials, n=1196) 17. 5% vs. 21. 2% 1. 10 (0. 83 – 1. 13)* ICU length of stay (5 trials, n=555) __ -054 (-2. 20 – 1. 13) C. Difficile infection (0 trials, n=0) NA NA *Results reported as mean difference Crit Care Med 2013; 41: 693

One More Meta-Analysis • Intensive Care Med 2014; 40: 11 -20 • 20 trials included; H 2 RA (20 trials) and PPI (two trials) • Conventional MA revealed a reduction in bleeding with SUP • Sequential trial analysis indicated No Significance • Like all other MA, no mortality benefit

Raises the question, “Is acid suppression necessary in critically ill patients”?

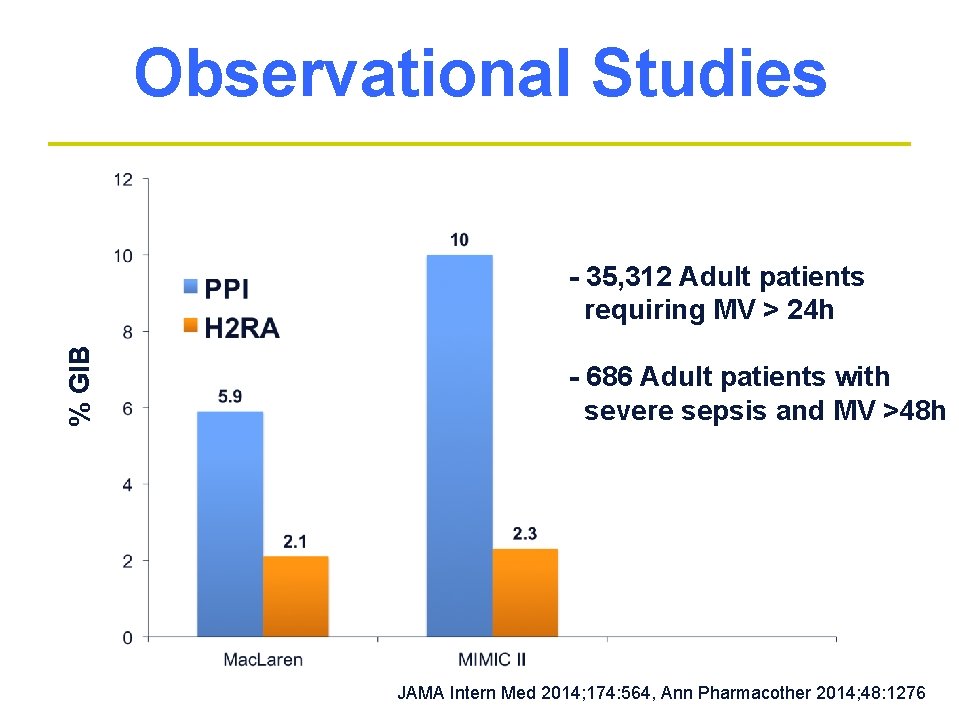
Observational Studies % GIB - 35, 312 Adult patients requiring MV > 24 h - 686 Adult patients with severe sepsis and MV >48 h JAMA Intern Med 2014; 174: 564, Ann Pharmacother 2014; 48: 1276

Withholding Pantoprazole for Stress Ulcer Prophylaxis in Critically Ill Patients: A Pilot Randomized Clinical Trial and Meta-Analysis Patient Criteria Interventions/Outcomes • > 18 years of age • Primary: withholding SUP • Invasive MV anticipated for > 48 hours or at least 72 hours prior to randomization • Randomized, blinded, placebo controlled • No active GI bleed or increased risk of bleeding - 2 antiplatelet drugs • Clinical Outcomes - Clinically important GI bleed - Ventilator associated pneumonia (VAP) • Palliative care or decision to withdraw life support - C. difficile infection • Pregnancy - ICU length of stay • Receipt of > two DDE of prophylaxis with H 2 RA or PPI - Hospital length of stay • - ICU mortalitry C

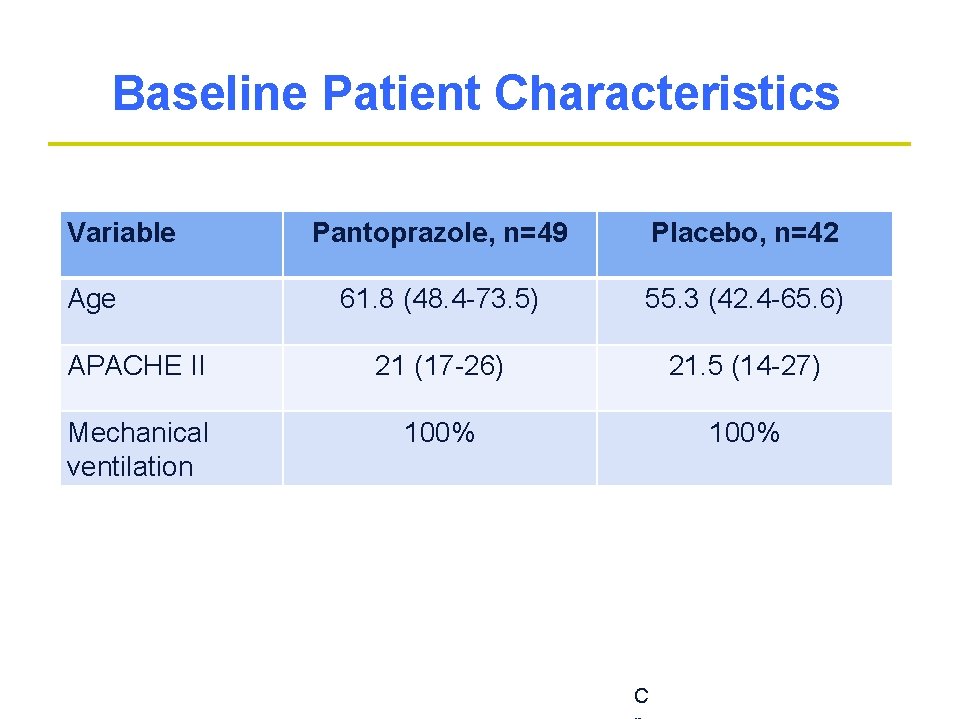
Baseline Patient Characteristics Variable Pantoprazole, n=49 Placebo, n=42 61. 8 (48. 4 -73. 5) 55. 3 (42. 4 -65. 6) APACHE II 21 (17 -26) 21. 5 (14 -27) Mechanical ventilation 100% Age C

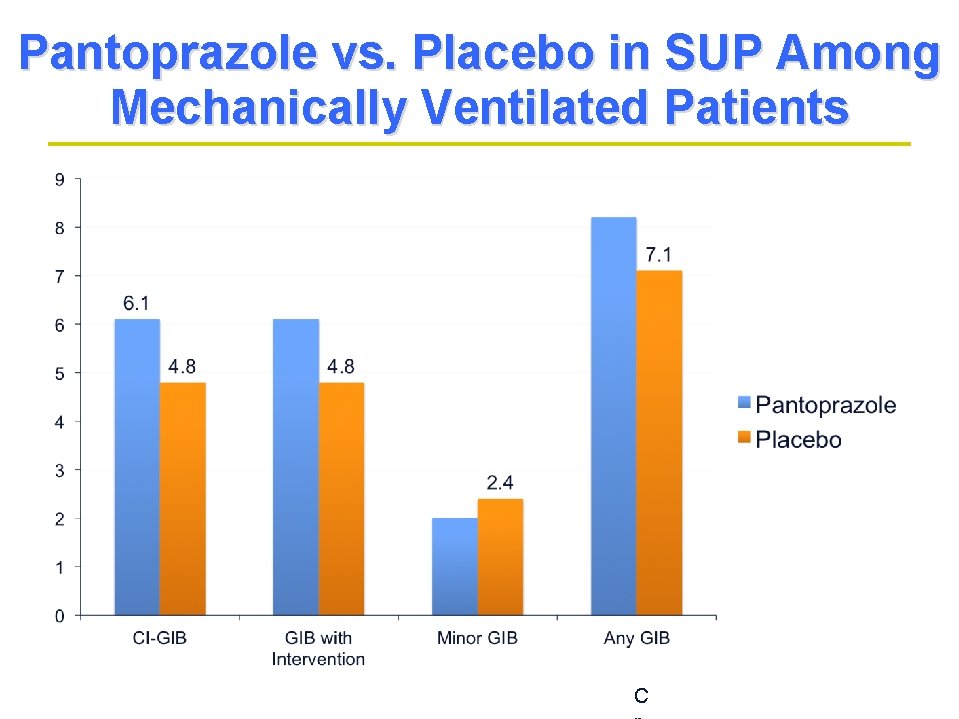
Pantoprazole vs. Placebo in SUP Among Mechanically Ventilated Patients C

Meta-Analysis Clinically Important Gastrointestinal Bleeding C


Consequences of Overuse • Clostridium difficile exists in two forms - Acid-sensitive vegetative form - Acid-resistant spore form • Vegetative form typically destroyed in acid environment • Altering gastric acidity may predispose patient to acquiring enteric infections • Over 30 trials have evaluated PPIs and C. difficile; 2 retrospective trials in the ICU; no RCTs

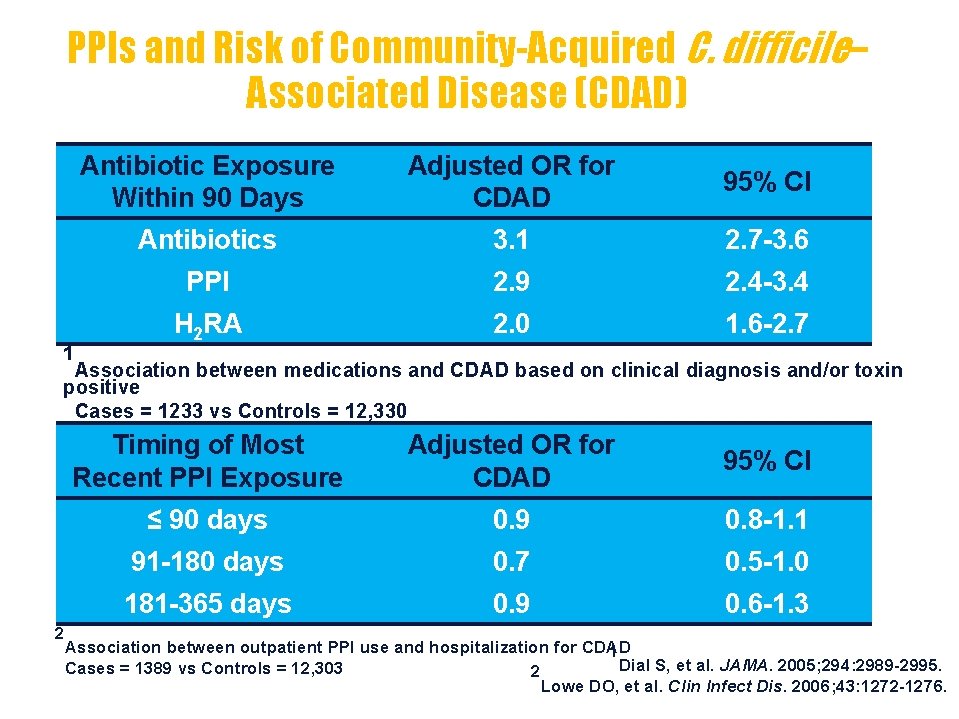
PPIs and Risk of Community-Acquired C. difficile– Associated Disease (CDAD) 1 Antibiotic Exposure Within 90 Days Adjusted OR for CDAD 95% CI Antibiotics 3. 1 2. 7 -3. 6 PPI 2. 9 2. 4 -3. 4 H 2 RA 2. 0 1. 6 -2. 7 Association between medications and CDAD based on clinical diagnosis and/or toxin positive Cases = 1233 vs Controls = 12, 330 2 Timing of Most Recent PPI Exposure Adjusted OR for CDAD 95% CI ≤ 90 days 0. 9 0. 8 -1. 1 91 -180 days 0. 7 0. 5 -1. 0 181 -365 days 0. 9 0. 6 -1. 3 Association between outpatient PPI use and hospitalization for CDAD 1 Dial S, et al. JAMA. 2005; 294: 2989 -2995. Cases = 1389 vs Controls = 12, 303 2 Lowe DO, et al. Clin Infect Dis. 2006; 43: 1272 -1276.

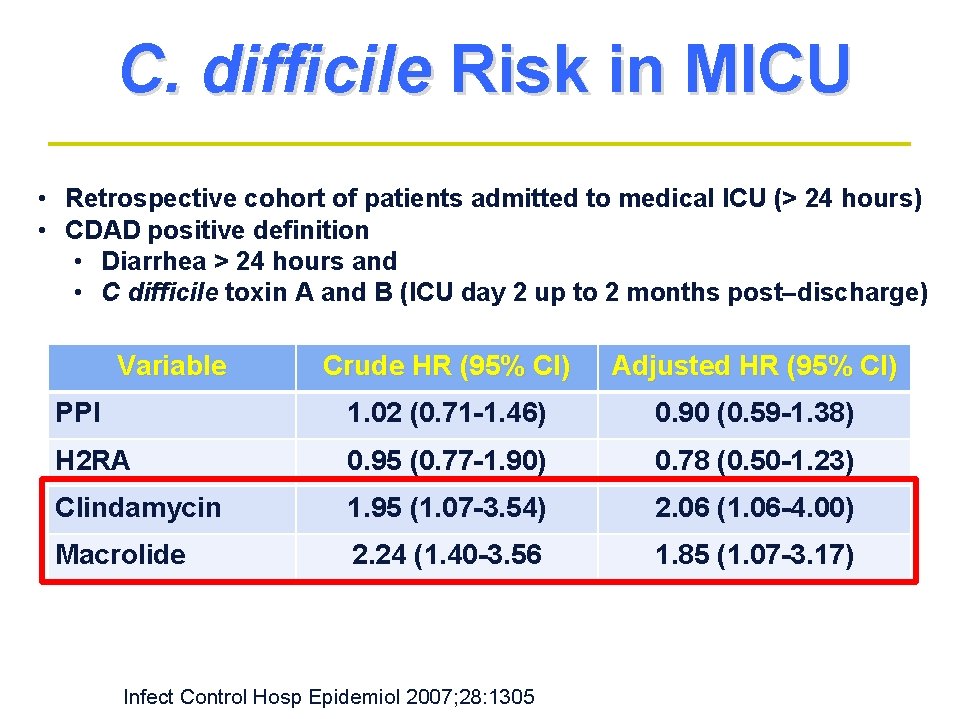
C. difficile Risk in MICU • Retrospective cohort of patients admitted to medical ICU (> 24 hours) • CDAD positive definition • Diarrhea > 24 hours and • C difficile toxin A and B (ICU day 2 up to 2 months post–discharge) Variable Crude HR (95% CI) Adjusted HR (95% CI) PPI 1. 02 (0. 71 -1. 46) 0. 90 (0. 59 -1. 38) H 2 RA 0. 95 (0. 77 -1. 90) 0. 78 (0. 50 -1. 23) Clindamycin 1. 95 (1. 07 -3. 54) 2. 06 (1. 06 -4. 00) Macrolide 2. 24 (1. 40 -3. 56 1. 85 (1. 07 -3. 17) Infect Control Hosp Epidemiol 2007; 28: 1305

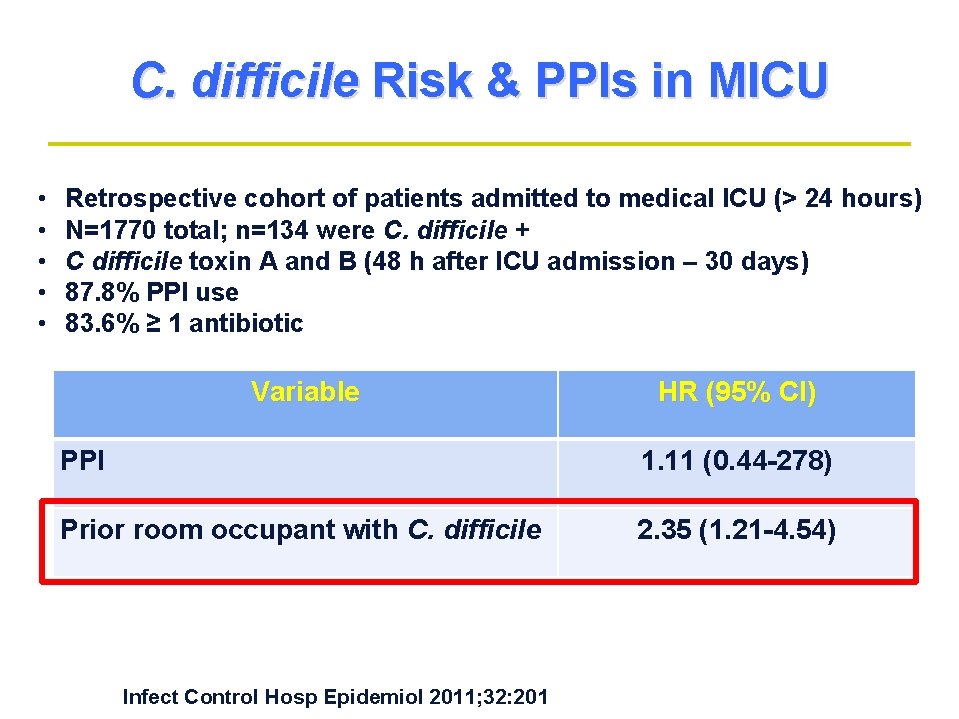
C. difficile Risk & PPIs in MICU • • • Retrospective cohort of patients admitted to medical ICU (> 24 hours) N=1770 total; n=134 were C. difficile + C difficile toxin A and B (48 h after ICU admission – 30 days) 87. 8% PPI use 83. 6% ≥ 1 antibiotic Variable HR (95% CI) PPI 1. 11 (0. 44 -278) Prior room occupant with C. difficile 2. 35 (1. 21 -4. 54) Infect Control Hosp Epidemiol 2011; 32: 201

PPIs and Risk of Community. Acquired CDAD Ü 317 cases of community-acquired C. difficile–associated disease treated with oral vancomycin and 3167 controls Odds ratio for C. difficile– associated disease 95% CI Antibiotic 8. 2 6. 1 – 11. 0 PPI 3. 5 2. 3 – 5. 2 Exposure within 90 days • Questions remain: – Epidemiologic studies have limitations – C. difficile–associated disease involves a complex interaction between host, pathogen, and environment Dial S, et al. CMAJ. 2006; 175: 745 -748. Cunningham R. CMAJ. 2006; 175: 757 -758.

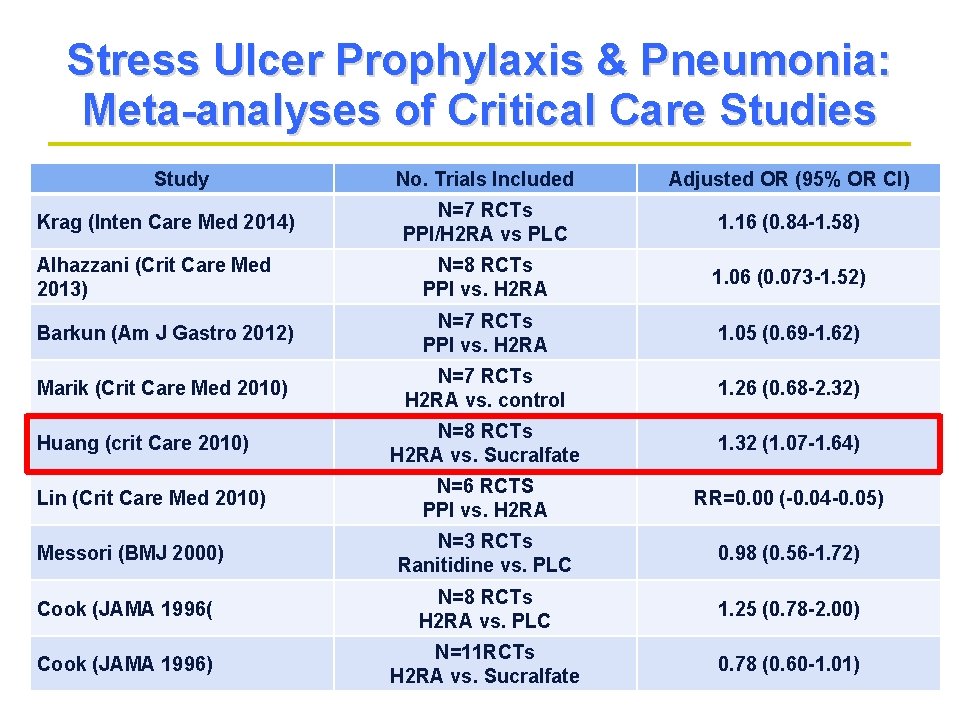
Stress Ulcer Prophylaxis & Pneumonia: Meta-analyses of Critical Care Studies Study No. Trials Included Adjusted OR (95% OR CI) N=7 RCTs PPI/H 2 RA vs PLC 1. 16 (0. 84 -1. 58) Alhazzani (Crit Care Med 2013) N=8 RCTs PPI vs. H 2 RA 1. 06 (0. 073 -1. 52) Barkun (Am J Gastro 2012) N=7 RCTs PPI vs. H 2 RA 1. 05 (0. 69 -1. 62) Marik (Crit Care Med 2010) N=7 RCTs H 2 RA vs. control 1. 26 (0. 68 -2. 32) N=8 RCTs H 2 RA vs. Sucralfate 1. 32 (1. 07 -1. 64) N=6 RCTS PPI vs. H 2 RA RR=0. 00 (-0. 04 -0. 05) Messori (BMJ 2000) N=3 RCTs Ranitidine vs. PLC 0. 98 (0. 56 -1. 72) Cook (JAMA 1996( N=8 RCTs H 2 RA vs. PLC 1. 25 (0. 78 -2. 00) Cook (JAMA 1996) N=11 RCTs H 2 RA vs. Sucralfate 0. 78 (0. 60 -1. 01) Krag (Inten Care Med 2014) Huang (crit Care 2010) Lin (Crit Care Med 2010)

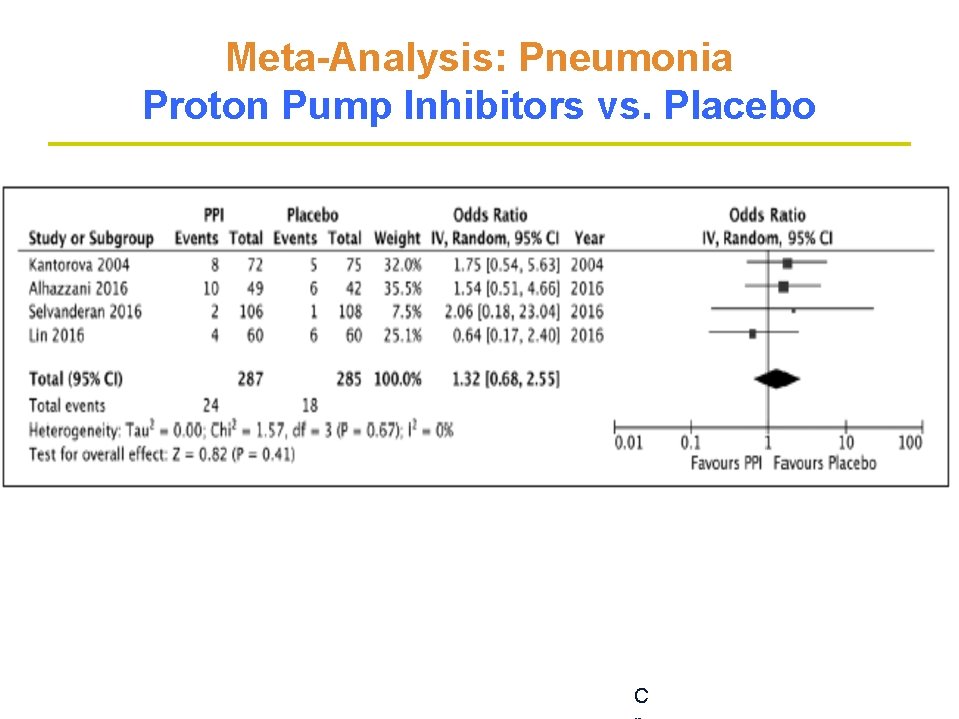
Meta-Analysis: Pneumonia Proton Pump Inhibitors vs. Placebo C

Efficacy and Safety Take Home Messages • Prevention of clinical important GI bleeds - Prevalence of CI-GIB is low - Majority of RCTs demonstrate no difference - Results of meta-analyses dependent on statistical analysis - No improvement in mortality - Treatment effect in favor of PPIs diminishes with high quality (i. e. , low risk of bias) RCTs

Efficacy and Safety Take Home Messages • Safety - Consistent findings of no statistical difference regarding nosocomial pneumonia, ICU length of stay, and ICU mortality - Observational data suggest a relationship between PPI use and C. difficile infection - No comparative data from RCTs support C. difficile associated infection - Until then, either option can be justified for SUP, although randomized trials suggest no prophylaxis is required - However, time has come that pharmacist with physicians can start limiting SUP to only the most severely ill In the ICU, less is often more

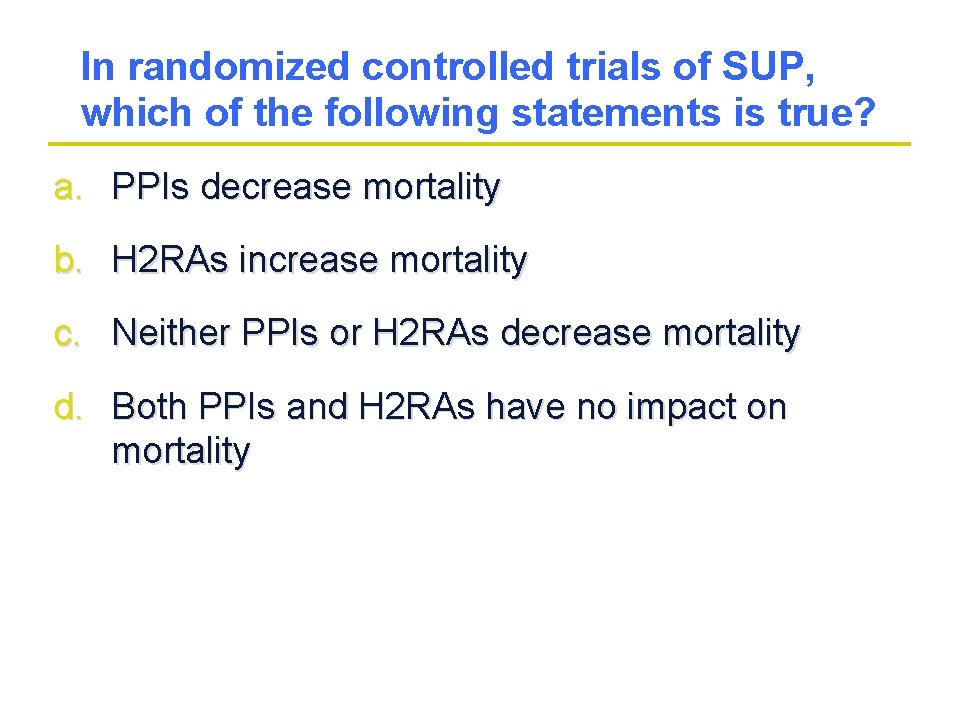
In randomized controlled trials of SUP, which of the following statements is true? a. PPIs decrease mortality b. H 2 RAs increase mortality c. Neither PPIs or H 2 RAs decrease mortality d. Both PPIs and H 2 RAs have no impact on mortality

The primary cause of stress ulcers in medical ICU patients is: a. Decreased bicarbonate secretion b. Splanchic hypoperfusion c. Hypersecretion of acid in the stomach d. Release of IL-6 into the gastric mucosa Splanchnic

Which of the following statements are true regarding the impact of PPIs on C. difficile infection in the ICU? a. Previous occupant of the room with C. difficile significantly increases the incidence b. Antibiotics generally have a lower incidence of C. difficile compared to PPIs c. PPIs have a significant impact on toxin A but not toxin B in C. difficile infection d. Prior exposure to an H 2 RA is generally required before a PPI can induce C. difficile infection