Strengths of Conjugate AcidBase Pairs Acid strength increases
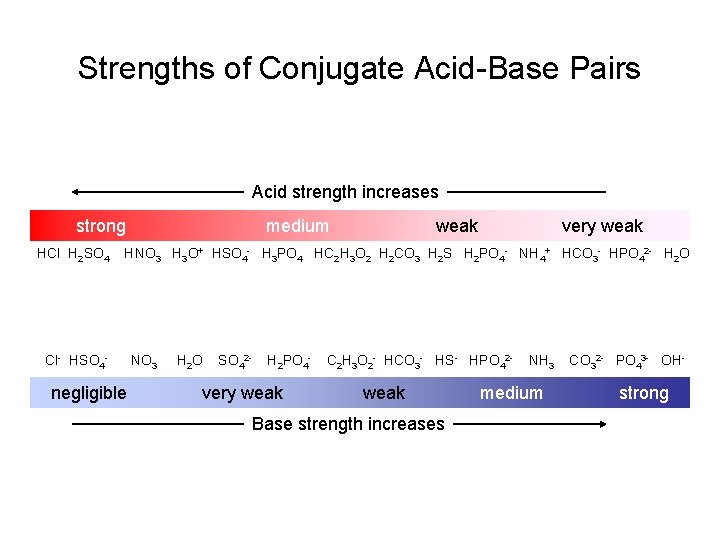
Strengths of Conjugate Acid-Base Pairs Acid strength increases strong HCl H 2 SO 4 medium weak very weak HNO 3 H 3 O+ HSO 4 - H 3 PO 4 HC 2 H 3 O 2 H 2 CO 3 H 2 S H 2 PO 4 - NH 4+ HCO 3 - HPO 42 - H 2 O Cl- HSO 4 - negligible NO 3 H 2 O SO 42 - H 2 PO 4 - very weak C 2 H 3 O 2 - HCO 3 - HS- HPO 42 - weak Base strength increases NH 3 medium CO 32 - PO 43 - OH- strong
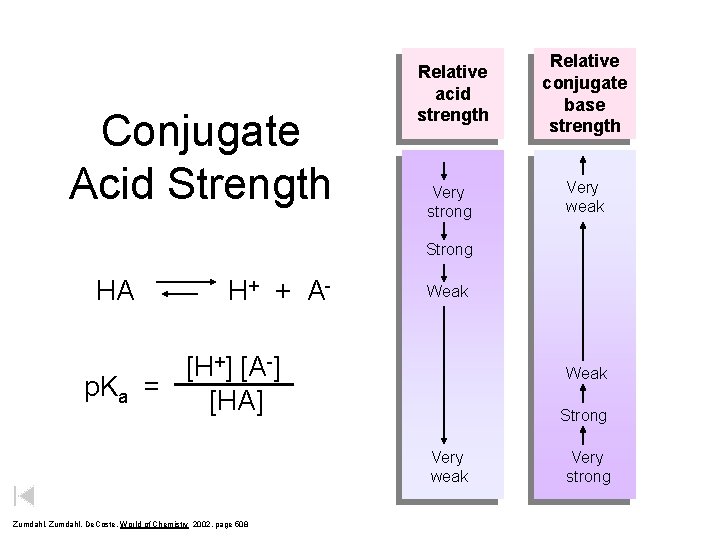
Conjugate Acid Strength Relative acid strength Relative conjugate base strength Very strong Very weak Strong HA H + + A- Weak [H+] [A-] p. Ka = [HA] Weak Strong Very weak Zumdahl, De. Coste, World of Chemistry 2002, page 508 Very strong

Solutions of Acids and Bases: The Leveling Effect • No acid stronger than H 3 O+ and no base stronger than OH– can exist in aqueous solution, leading to the phenomenon known as the leveling effect. • Any species that is a stronger acid than the conjugate acid of water (H 3 O+) is leveled to the strength of H 3 O+ in aqueous solution because H 3 O+ is the strongest acid that can exist in equilibrium with water. • In aqueous solution, any base stronger than OH– is leveled to the strength of OH– because OH– is the strongest base that can exist in equilibrium with water • Any substance whose anion is the conjugate base of a compound that is a weaker acid than water is a strong base that reacts quantitatively with water to form hydroxide ion

Weak Acids (p. Ka) Weak Acids – dissociate incompletely (~20%) Strong Acids – dissociate completely (~100%) A(g) + 2 B(g) 3 C(g) + D(g) Equilibrium constant (Keq) = Keq = Le. Chatelier’s Principle (lu-SHAT-el-YAY’s)
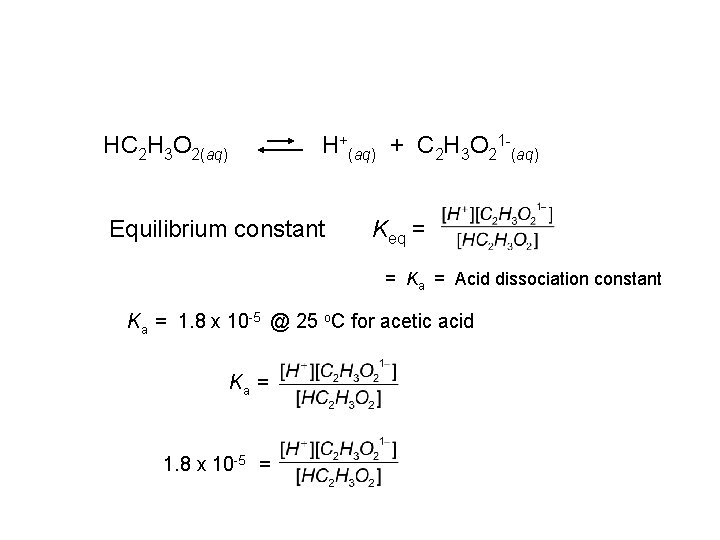
HC 2 H 3 O 2(aq) H+(aq) + C 2 H 3 O 21 -(aq) Equilibrium constant Keq = = Ka = Acid dissociation constant Ka = 1. 8 x 10 -5 @ 25 o. C for acetic acid Ka = 1. 8 x 10 -5 =
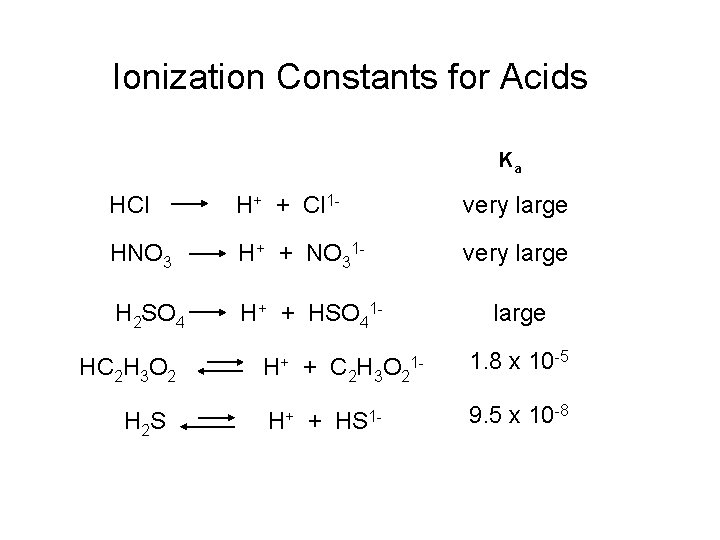
Ionization Constants for Acids Ka HCl H+ + Cl 1 - very large HNO 3 H+ + NO 31 - very large H 2 SO 4 H+ + HSO 41 - HC 2 H 3 O 2 H 2 S large H+ + C 2 H 3 O 21 - 1. 8 x 10 -5 H+ 9. 5 x 10 -8 + HS 1 -
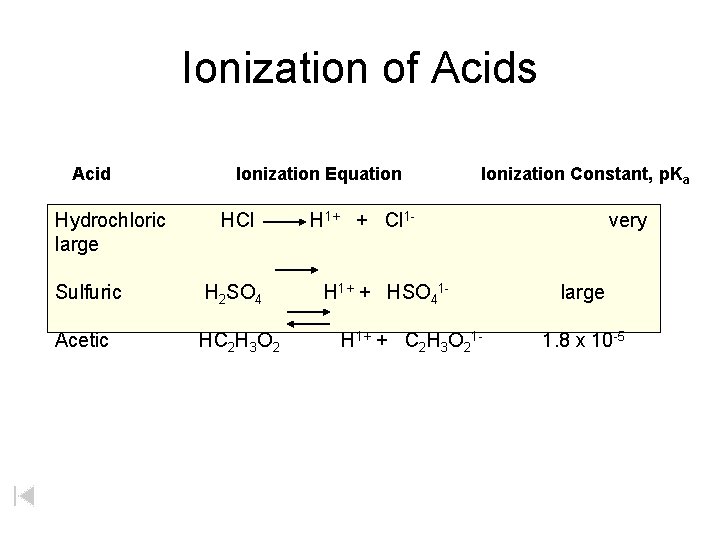
Ionization of Acids Acid Hydrochloric large Ionization Equation HCl Sulfuric H 2 SO 4 Acetic HC 2 H 3 O 2 Ionization Constant, p. Ka H 1+ + Cl 1 H 1+ + HSO 41 H 1+ + C 2 H 3 O 21 - very large 1. 8 x 10 -5
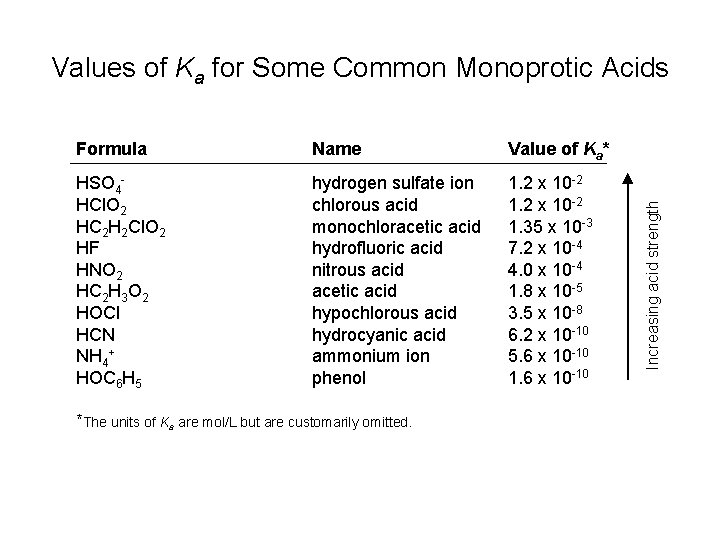
Formula Name Value of Ka* HSO 4 HCl. O 2 HC 2 H 2 Cl. O 2 HF HNO 2 HC 2 H 3 O 2 HOCl HCN NH 4+ HOC 6 H 5 hydrogen sulfate ion chlorous acid monochloracetic acid hydrofluoric acid nitrous acid acetic acid hypochlorous acid hydrocyanic acid ammonium ion phenol 1. 2 x 10 -2 1. 35 x 10 -3 7. 2 x 10 -4 4. 0 x 10 -4 1. 8 x 10 -5 3. 5 x 10 -8 6. 2 x 10 -10 5. 6 x 10 -10 1. 6 x 10 -10 *The units of Ka are mol/L but are customarily omitted. Increasing acid strength Values of Ka for Some Common Monoprotic Acids

Sample 1) One gram of concentrated sulfuric acid (H 2 SO 4) is diluted to a 1. 0 dm 3 volume with water. What is the molar concentration of the hydrogen ion in this solution? What is the p. H? Solution) First determine the number of moles of H 2 SO 4 x mol H 2 SO 4 = 1 g H 2 SO 4 H+ + HSO 41 - 1 mol H 2 SO 4 98 g H 2 SO 4 & = 0. 010 mol H 2 SO 4 HSO 41 - H+ + SO 42 - OVERALL: H 2 SO 4 0. 010 M 2 H+ + SO 42 - in dilute solutions. . . occurs ~100% 0. 020 M p. H = - log [H+] substitute into equation p. H = - log [0. 020 M] p. H = 1. 69

A volume of 5. 71 cm 3 of pure acetic acid, HC 2 H 3 O 2, is diluted with water at 25 o. C to form a solution with a volume of 1. 0 dm 3. What is the molar concentration of the hydrogen ion, H+, in this solution? (The density of pure acetic acid is 1. 05 g/cm 3. ) Step 1) Find the mass of the acid Mass of acid = density of acid x volume of acid = 1. 05 g/cm 3 x 5. 71 cm 3 = 6. 00 g Step 2) Find the number of moles of acid. (From the formula of acetic acid, you can calculate that the molar mass of acetic acid is 60 g / mol). x mol acetic acid = 6. 00 g HC 2 H 3 O 2 Molarity: M = mol / L Substitute into equation Step 3) Find the [H+] Ka = = 0. 10 mol acetic acid (in 1 L) M = 0. 10 mol / 1 L M = 0. 1 molar HC 2 H 3 O 2
![HC 2 H 3 O 2 Step 3) Find the 0. 1 M [H+] HC 2 H 3 O 2 Step 3) Find the 0. 1 M [H+]](http://slidetodoc.com/presentation_image/5ba30e8731425b56a619cb908144ceb7/image-11.jpg)
HC 2 H 3 O 2 Step 3) Find the 0. 1 M [H+] weak acid H+ + C 2 H 3 O 21 - 0. 1? M Ka = 1. 8 x 10 -5 @ 25 o. C for acetic acid Ka = 1. 8 x 10 -5 How do the concentrations of H+ and C 2 H 3 O 21 - compare? = Substitute into equation: p. H = - log[H+] x 2 = 1. 8 x 10 -6 M x = 1. 3 x 10 -3 molar p. H = - log [1. 3 x 10 -3 M] = [H+] p. H = 2. 9
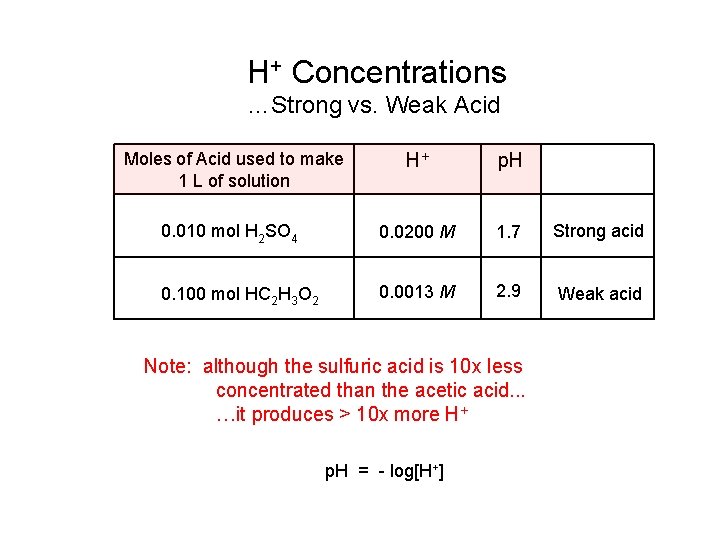
H+ Concentrations …Strong vs. Weak Acid Moles of Acid used to make 1 L of solution H+ p. H 0. 010 mol H 2 SO 4 0. 0200 M 1. 7 Strong acid 0. 100 mol HC 2 H 3 O 2 0. 0013 M 2. 9 Weak acid Note: although the sulfuric acid is 10 x less concentrated than the acetic acid. . . …it produces > 10 x more H+ p. H = - log[H+]

Practice Problems: 1 a) What is the molar hydrogen ion concentration in a 2. 00 dm 3 solution of hydrogen chloride in which 3. 65 g of HCl is dissolved? 1 b) p. H 2 a) What is the molar concentration of hydrogen ions in a solution containing 3. 20 g of HNO 3 in 250 cm 3 of solution? 2 b) p. H 3 a) An acetic acid solution is 0. 25 M. What is its molar concentration of hydrogen ions? 3 b) p. H 4) A solution of acetic acid contains 12. 0 g of HC 2 H 3 O 2 in 500 cm 3 of solution. What is the molar concentration of hydrogen ions? 1 a) 0. 0500 M 1 b) p. H = 1. 3 2 a) 0. 203 M 2 b) p. H = 0. 7 3 a) 2. 1 x 10 -3 M 3 b) p. H = 2. 7 4) 2. 7 x 10 -3 M

Weak Acids Cyanic acid is a weak monoprotic acid. If the initial concentration of cyanic + is 4. 8 x 10 -3 M, acid is 0. 150 M and the equilibrium concentration. Hof 3 O H 3+O (aq) calculate Ka for cyanic acid. 4. 8 x 10 -3 M 0. 150 M Ka = + CN 1 -(aq) H+(aq) HCN(aq) [Products] [Reactants] Ka = [4. 8 x 10 -3 M][4. 8 [CNx 1 -10 ] -3 M] [0. 150 M] 4. 8 x 10 -3 M [H 3 O+] [CN 1 -] [HCN] Ka = 1. 54 x 10 -4 How is [H 3 O+] determined? Measure p. H of solution and work backwards
- Slides: 14