Stoichiometry with Chemical Reactions Stoichiometry Unit Objectives n

Stoichiometry with Chemical Reactions

Stoichiometry Unit Objectives n Solve reaction stoichiometry problems: ¨ Mole- Mole ¨ Mass – Mole ¨ Mass-Mass ¨ Mass Volume

Introduction In any stoichiometry problem we start with a given quantity of a reactant or product. We use conversion factors and unit analysis to determine the quantities of the unknowns that react or form. n Conversion factors: n 1. Molar Mass : ________ n 2. Mole Ratio : _______ n 3. Molar Volume of a Gas: _______ n
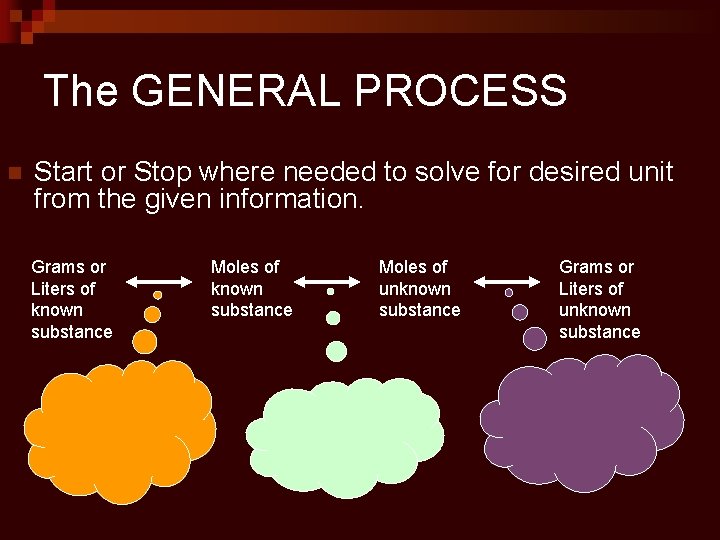
The GENERAL PROCESS n Start or Stop where needed to solve for desired unit from the given information. Grams or Liters of known substance Moles of unknown substance Grams or Liters of unknown substance

Mole to Mole n Mole Ratio: Mole ratio comes from: __________ n Example: Find all mole ratios for the reaction of the combustion of ethane. n ___C 2 H 6 + ___O 2 ___CO 2 + ___H 2 O n
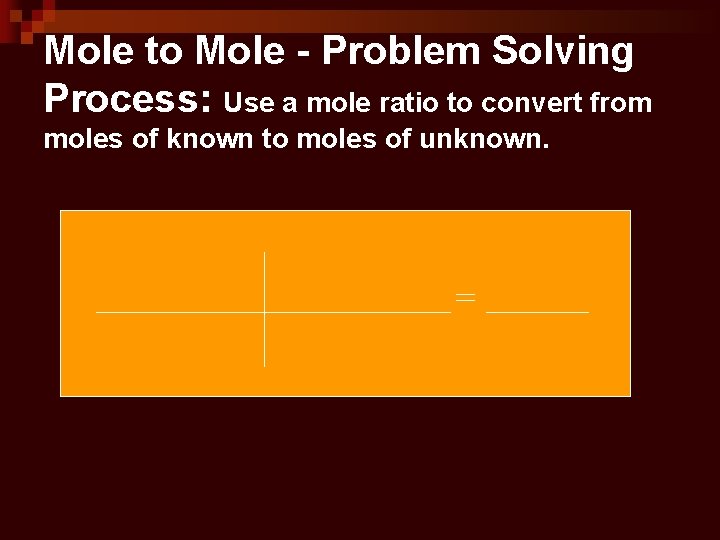
Mole to Mole - Problem Solving Process: Use a mole ratio to convert from moles of known to moles of unknown.

Mole-Mole Example: n How many moles of CO 2 will be produced from the combustion of 0. 575 mol of C 2 H 6?

Mole to Mass Problems: Start with a given number of moles, convert to unknown number of grams. Problem Solving Process:

Mole to Mass Example n In photosynthesis, plants use the sun’s energy to produce glucose and oxygen from water and carbon dioxide. How many grams of glucose are produced when 5. 25 moles of Carbon Dioxide react with excess water?

Mole to Mass Example (cont. )

Mole to Mass (cont. ) n What mass of water is needed to react with the above carbon dioxide?

Mass – Mass Problems n Start with a given number of moles, convert to unknown number of grams.

Mass to Mass - Problem Solving Process

Mass to Mass Industrial solvent Carbon disulfide is produced in the following unbalanced reaction: C + SO 2 CS 2 + CO n How many grams of CS 2 and CO are formed if 12. 5 g of SO 2 react? n

Volume Problems n n n n Recall the standard molar volume of a gas: 22. 4 L/mol This is true only at STP: 1 ATM and 0 degree C Avogadro’s Law: Since a mole of any gas occupies the same volume as any other gas, a volume ratio is the same as a mole ratio. Ex: __1_ N 2 + _3__ H 2 __2_ NH 3 Mole Ratios: 1 mol N 2: 3 mole H 2: 2 mol NH 3 Volume Ratios: 1 L N 2: 3 L H 2: 2 L NH 3
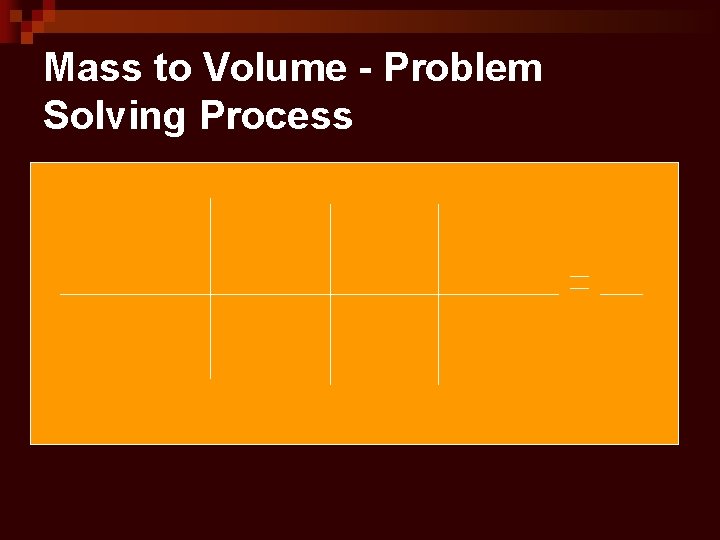
Mass to Volume - Problem Solving Process

Mass to Volume n A piece of magnesium metal with a mass of 2. 76 g is added to a solution of Hydrochloric acid. What is the volume of Hydrogen gas produced at STP?

Limiting Reactants n Stoichiometric Proportions: n Limiting Reactant: n Excess Reactant:

LR – How To – Method 1 To find the Limiting reactant in a chemical reaction: 1. Pick a reactant and assume it is the LR. 2. Calculate the amount of the other reactant needed to use up all of the LR. 3. Compare the amount needed to the amount available, for the second reactant. (given in the problem. ) If you have enough of it (more available than needed) then it is the XS reactant, and you guessed right: The first reactant is the LR ¨ If you don’t have enough of it (more needed than you are given) then you guessed wrong: the second reactant is the LR and the first is in XS. ¨

LR – How to – Method 2 n To find the Limiting Reactant in a chemical reaction: n Pick a reactant, and assume it’s the limiting reactant. Calculate the amount of ONE of the products that can be produced from that reactant. n Now pick the other reactant, and calculate the amount of the SAME product made from that reactant. n The reactant that produces the LEAST amount of PRODUCT is the limiting reactant, and this lesser amount of product is the actual amount of product made.

2 H 2 + O 2 2 H 2 O n Determine the LR of the reaction when given 1. 22 g O 2 and 1. 05 g H 2. (method 1)

H 2 + Cl 2 2 HCl n Determine the LR of the reaction when given 2. 92 g Cl 2 and 3. 65 g H 2. (method 2)

% Yield in a Chemical Reaction n % Yield = n Actual Yield = n Expected Yield = n Actual Yield is always less than expected yield and there are several reasons for this:

Actual Yield is always less than expected yield. There are several reasons for this: 1. 2. 3.

% Yield Example Problem n When 4. 9 g of Magnesium are burned in excess oxygen, 6. 5 g of Magnesium oxide are formed. What is the % yield of the chemical reaction?
- Slides: 25