Stoichiometry Using the Balanced Equation What does the

Stoichiometry Using the Balanced Equation

What does the balanced equation really mean? 2 H 2 + O 2 2 H 2 O Particles: 2 molecules H 2 + 1 molecule O 2 2 molecules H 2 O Moles: 2 moles H 2 + 1 mole O 2 2 moles H 2 O Which is more useful? moles

2 H 2 + O 2 2 H 2 O Like a recipe, the balanced equation tells you in what ratios the ingredients must be mixed. Therefore, for every 2 moles of H 2 you will need 1 mole of O 2. Mathematically, this can be written 2 moles H 2 = 1 mole O 2 What can you do with equalities? Dimensional Analysis

2 H 2 + O 2 2 H 2 O How many conversion factors (called mole ratios) can be written from this 1 equation? 2 moles H 2 = 1 mole O 2 2 moles H 2 = 2 mole H 2 O 1 mole O 2 = 2 moles H 2 O
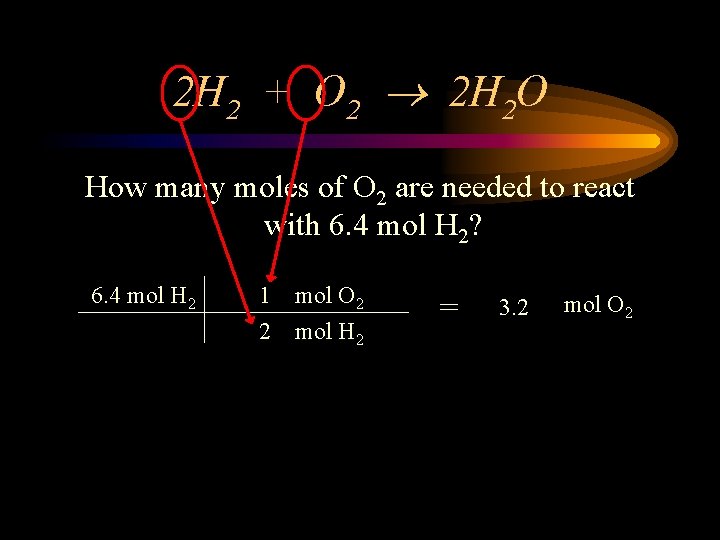
2 H 2 + O 2 2 H 2 O How many moles of O 2 are needed to react with 6. 4 mol H 2? 6. 4 mol H 2 1 2 mol O 2 mol H 2 = 3. 2 mol O 2

2 H 2 + O 2 2 H 2 O How many moles of H 2 O are produced from 6. 4 mol H 2? 6. 4 mol H 2 2 2 mol H 2 O mol H 2 = 6. 4 mol H 2 O

Multi-step Stoichiometry EVERY stoiciometry problem involves moving from 1 substance to another! How many molecules of H 2 does it take to make 647 g of H 2 O? 2 H 2 + O 2 2 H 2 O ? molecules 647 g The only way to move from 1 substance to another is the mole ratio!

Multi-step Stoichiometry You have three tools for stoichiometry problems! Tools of Stoichiometry 1. 2. 3. 4. Mole Ratio Molar Mass Avogadro's # Volume balanced equation coefficients periodic table mass = 1 mol 6. 02 1023 particles = 1 mol particles 22. 4 L at STP moles A moles B mass A (g) 1 mole A particles A 1 mole A 22. 4 L of A Every stoichiometry problem uses the mole ratio!!!

Multi-step Stoichiometry To answer the original question, you need to use all 3 tools. How many molecules of H 2 does it take to make 647 g of H 2 O? 2 H 2 + O 2 2 H 2 O ? molecules 647 g This problem not only involves changing substances (mole ratio), but also mass (molar mass) and molecules (Avogadro’s number).

2 H 2 + O 2 2 H 2 O ? molecules 647 g First, you want to set up a dimensional analysis problem. Put your given and target in the equation. 647 g H 2 O = ? molecules H 2
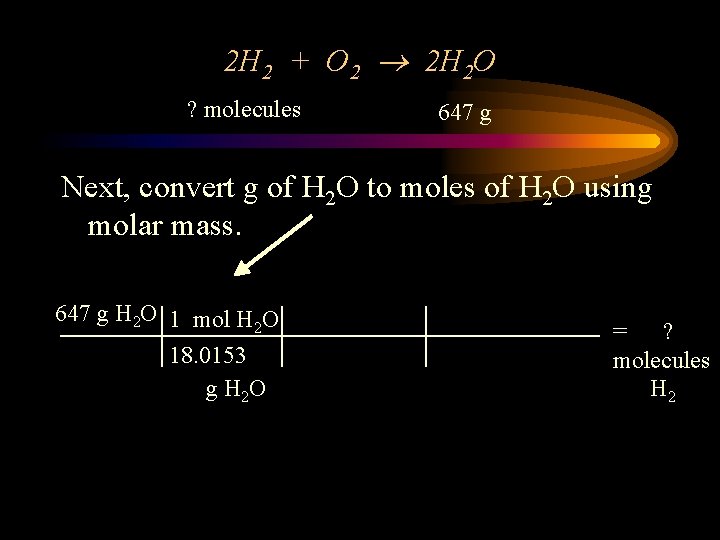
2 H 2 + O 2 2 H 2 O ? molecules 647 g Next, convert g of H 2 O to moles of H 2 O using molar mass. 647 g H 2 O 1 mol H O 2 18. 0153 g H 2 O = ? molecules H 2

2 H 2 + O 2 2 H 2 O ? molecules 647 g Next, EVERY problem must have a mole ratio step, so put that next! 647 g H 2 O 1 mol H O 2 mol H 2 2 = ? 18. 0153 g 2 mol H 2 O molecules H 2 O H 2 • You don’t want H 2 O, so put moles H 2 O in the bottom of the middle step (remember you have to be in moles to move from 1 substance to another). • You want to change to H 2, so put moles of H 2 on top (mole ratio is moles to moles). • The numbers come from the balanced equation!
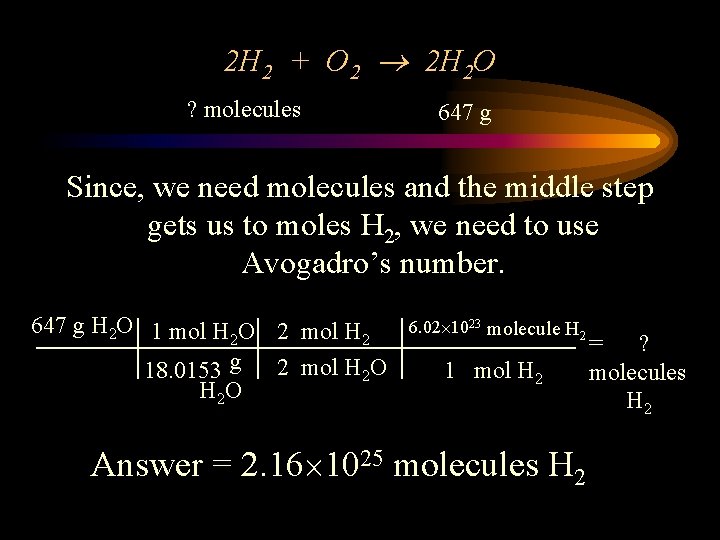
2 H 2 + O 2 2 H 2 O ? molecules 647 g Since, we need molecules and the middle step gets us to moles H 2, we need to use Avogadro’s number. 647 g H 2 O 1 mol H 2 O 2 mol H 2 18. 0153 g 2 mol H 2 O 6. 02 1023 molecule H 2 1 mol H 2 Answer = 2. 16 1025 molecules H 2 = ? molecules H 2

Calculating Stoichiometric Problems 1. Balance the equation 2. Convert given to moles 1. Set up mole ratios. 2. Use mole ratios to calculate moles of desired chemical. 3. Convert moles to target.

Another Example How many grams of O 2 are needed to use up 13. 6 moles of H 2? 2 H 2 + O 2 2 H 2 O 13. 6 mol H 2 g? 1 mol O 2 31. 9988 g O 2 2 mol H 2 1 mol O 2 = 218 g O 2

12. 3 Limiting Reactants Cake Recipe 2 cups flour 1 cup butter 1 tsp. salt 1/2 cup sugar If I have 6 cups of flour, 4 cups of butter, 112 tsp. of salt, and 429 cups of sugar, how many cakes can I make?

12. 3 Limiting Reactants Cake Recipe You Have in the Cupboard 2 cups flour 1 cup butter 1 tsp. Salt 1/2 cup sugar 6 cups flour 4 cup butter 112 tsp. salt 429 cups sugar 1 cakes 2 cups flour 4 cups butter 1 cakes 1 cups butter = 3 cakes = 4 cakes

12. 3 Limiting Reactants Cake Recipe You Have in the Cupboard 2 cups flour 1 cup butter 1 tsp. Salt 1/2 cup sugar 112 tsp. salt 6 cups flour 4 cup butter 112 tsp. salt 429 cups sugar 1 cakes 1 tsp. salt 429 cups sugar 1 cakes. 5 cups sugar = 112 cakes = 858 cakes

12. 3 Limiting Reactants Flour controls or limits how many cakes we can make because after we make 3 cakes we run out of flour. It also controls how much of the other ingredients we need!

12. 3 Limiting Reactants How much butter is left over? First find how much was used and then subtract how much is left. FLOUR controls how much we use. 6 cups flour 1 cup butter 2 cups flour = 3 cups butter 4 cups butter – 3 cups butter = 1 cup butter left

12. 3 Limiting Reactants Limiting reactant - the reactant that makes the least. It controls everything about the reaction because when it is used up the reaction stops. Chemical reactions work the same way!!!!

12. 3 Limiting Reactants 2 H 2 + O 2 2 H 2 O 2. 50 g 3. 40 g How much water can be made? 2. 50 g H 2 3. 40 g O 2 1 mol H 2 2 mol H 2 O 18. 0153 g H 2 O = 22. 3 g H O 2 You must find which reactant controls 2. 01588 g H 2 2 mol H 2 1 mol H 2 O (limiting reactant) the reaction. 1 mol O 2 2 mol H 2 O 18. 0153 g H 2 O = 3. 83 g H O 2 31. 9988 1 mol O 2 1 mol H 2 O g O 2 is therefore the limiting reactant and 3. 83 g of water is produced!

12. 3 Limiting Reactants 2 H 2 + O 2 2 H 2 O 2. 50 g 3. 40 g How much H 2 is left over? 1. Find out how much H 2 was used up by the 3. 40 g O 2 2. Then subtract that amount from 2. 50 g to find out how much H 2 is left

12. 3 Limiting Reactants 2 H 2 + O 2 2 H 2 O 2. 50 g 3. 40 g How much H 2 is left over? 3. 40 g O 2 1 mol O 2 2 mol H 2 31. 9988 1 mol O 2 g O 2 2. 01588 g H 2 1 mol H 2 2. 50 g. 428 g 2. 07 g H 2 left over =. 428 g H 2

12. 4 Percent Yield When we calculated that 3. 83 g of H 2 O could be made in the previous sample problem, we didn’t actually perform the reaction and measure the resulting water. We made theoretical prediction of how much could be made. Actual Yield – do the reaction to find the yield Theoretical Yield – do the math to predict the yield

12. 4 Percent Yield When doing stoichiometric calculations, you are calculating what is known as a theoretical yield - the maximum amount that could be produced during the reaction. Under ideal conditions, this is exactly what will happen. In reality, however, the actual yield will be less than what is predicted.

12. 4 Percent Yield
- Slides: 27