STOICHIOMETRY UNIT 1 Background Information 2 Converting grams

STOICHIOMETRY UNIT 1

Background Information 2
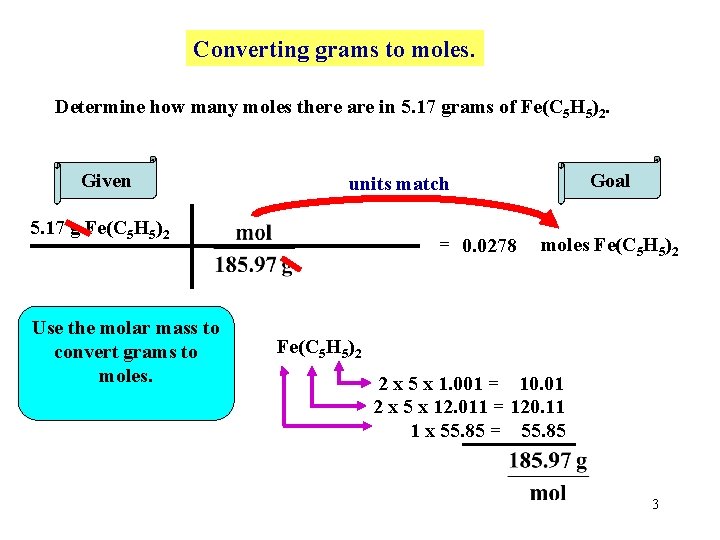
Converting grams to moles. Determine how many moles there are in 5. 17 grams of Fe(C 5 H 5)2. Given 5. 17 g Fe(C 5 H 5)2 Use the molar mass to convert grams to moles. Goal units match = 0. 0278 moles Fe(C 5 H 5)2 2 x 5 x 1. 001 = 10. 01 2 x 5 x 12. 011 = 120. 11 1 x 55. 85 = 55. 85 3

Stoichiometry

What is Stoichiometry? • Stoichiometry (Stoy-ki-ah-me-tree) is the calculation of relative amounts of reactants and products in a chemical reaction. • In other words: – Figuring out how many products you can make with the reactants you start with.
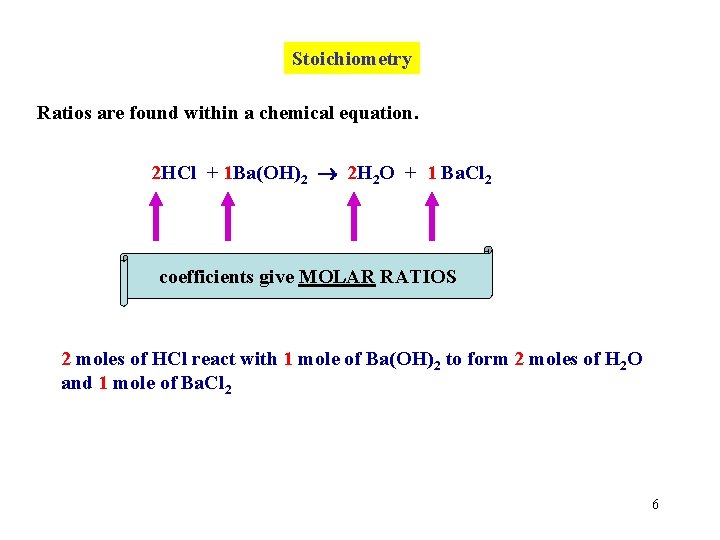
Stoichiometry Ratios are found within a chemical equation. 2 HCl + 1 Ba(OH)2 2 H 2 O + 1 Ba. Cl 2 coefficients give MOLAR RATIOS 2 moles of HCl react with 1 mole of Ba(OH)2 to form 2 moles of H 2 O and 1 mole of Ba. Cl 2 6
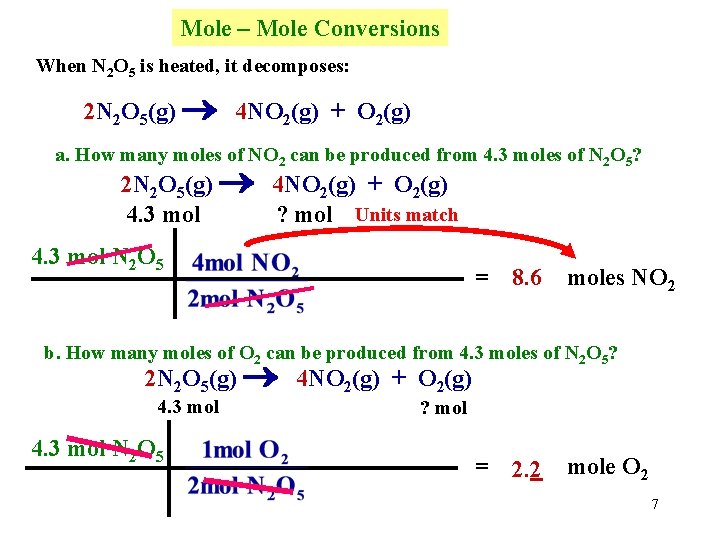
Mole – Mole Conversions When N 2 O 5 is heated, it decomposes: 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) a. How many moles of NO 2 can be produced from 4. 3 moles of N 2 O 5? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 4. 3 mol ? mol Units match 4. 3 mol N 2 O 5 = 8. 6 moles NO 2 b. How many moles of O 2 can be produced from 4. 3 moles of N 2 O 5? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 4. 3 mol N 2 O 5 ? mol = 2. 2 mole O 2 7

Mass-Mass Stoichiometry Problems Putting It All Together 8
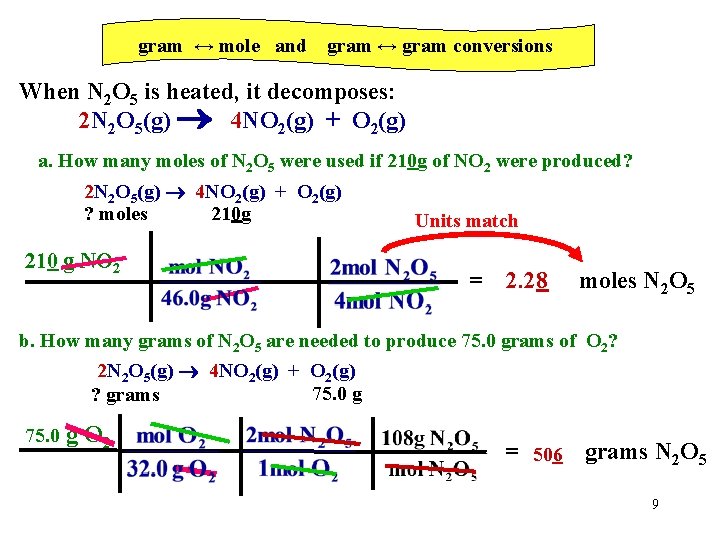
gram ↔ mole and gram ↔ gram conversions When N 2 O 5 is heated, it decomposes: 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) a. How many moles of N 2 O 5 were used if 210 g of NO 2 were produced? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) ? moles 210 g 210 g NO 2 Units match = 2. 28 moles N 2 O 5 b. How many grams of N 2 O 5 are needed to produce 75. 0 grams of O 2? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 75. 0 g ? grams 75. 0 g O 2 = 506 grams N 2 O 5 9

Gram to Gram Conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + 3 H 2(g) First write a balanced equation. 10

Gram to Gram Conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + ? grams 3. 45 g 3 H 2(g) Now let’s get organized. Write the information below the substances. 11
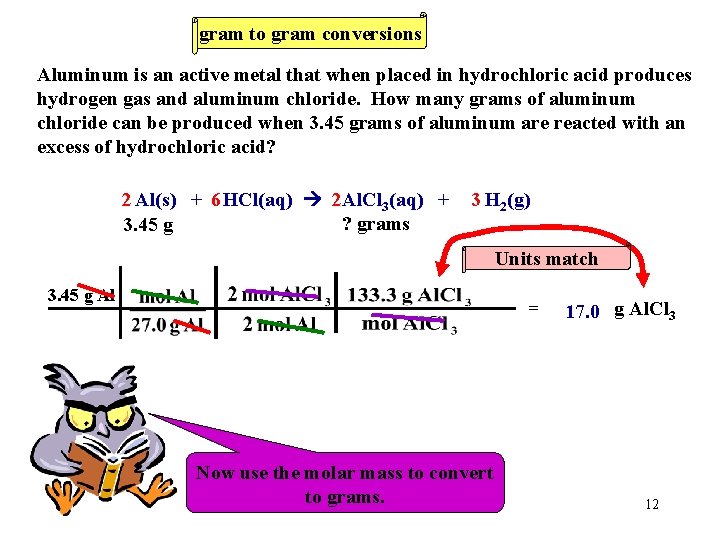
gram to gram conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + ? grams 3. 45 g 3 H 2(g) Units match 3. 45 g Al = Now We must Now use Let’s the always use work molar thethe convert molar mass problem. ratio. to toconvert moles. to grams. 17. 0 g Al. Cl 3 12

Next Topics 13
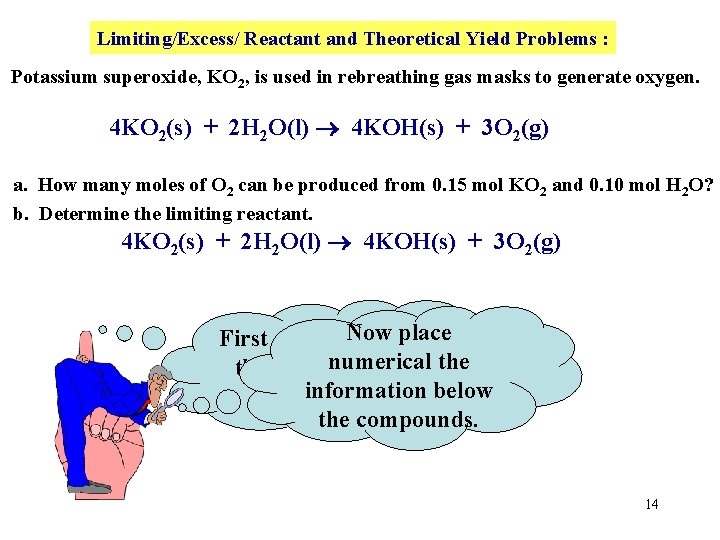
Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) Nowthe place First copy down numerical the BALANCED information below equation! the compounds. 14
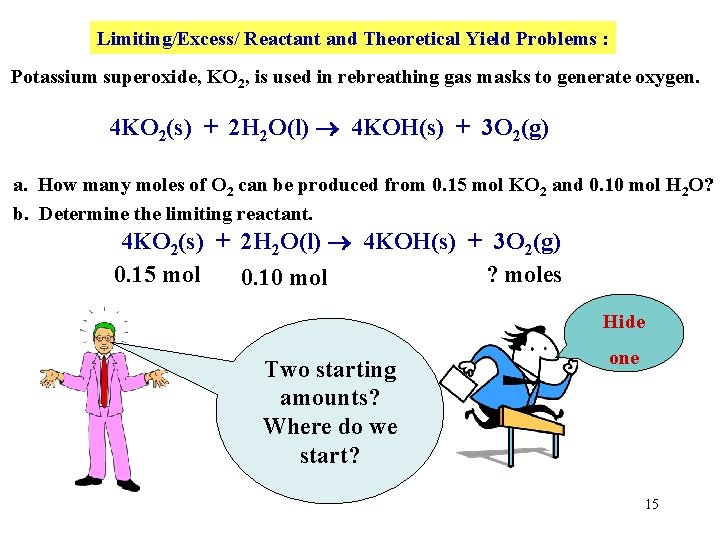
Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles 0. 10 mol Hide Two starting amounts? Where do we start? one 15

Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles Hide 0. 10 mol Based on: 0. 15 mol KO 2 = 0. 1125 mol O 2 16

Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles Hide 0. 10 mol Based on: 0. 15 mol KO 2 Based on: 0. 10 mol H 2 O H 2 O = 0. 1125 mol O 2 = 0. 150 mol O 2 17
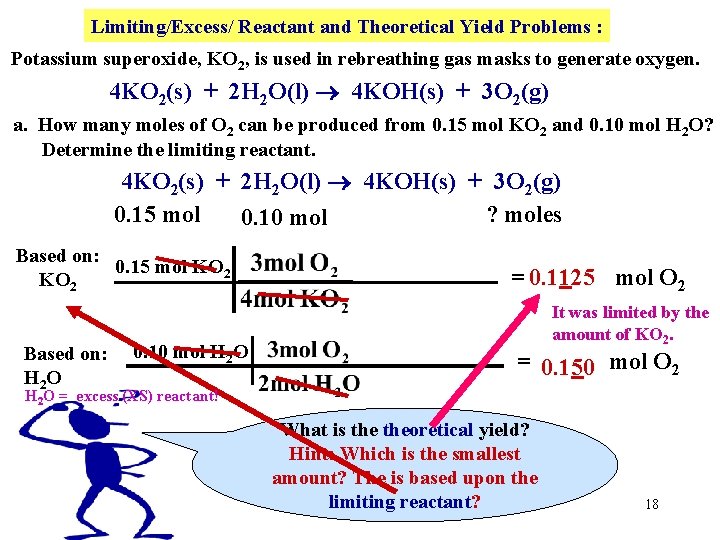
Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles 0. 10 mol Based on: 0. 15 mol KO 2 Based on: H 2 O 0. 10 mol H 2 O = 0. 1125 mol O 2 It was limited by the amount of KO 2. = 0. 150 mol O 2 H 2 O = excess (XS) reactant! What is theoretical yield? Hint: Which is the smallest amount? The is based upon the limiting reactant? 18

Theoretical yield vs. Actual yield Suppose theoretical yield for an experiment was calculated to be 19. 5 grams, and the experiment was performed, but only 12. 3 grams of product were recovered. Determine the % yield. Theoretical yield = 19. 5 g based on limiting reactant Actual yield = 12. 3 g experimentally recovered 19

Limiting/Excess Reactant Problem with % Yield 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 g 47. 0 one g Hide Based on: 120. 0 g KO 2 = 40. 51 g O 2 20

Limiting/Excess Reactant Problem with % Yield 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 47. 0 g Hideg Based on: 120. 0 g KO 2 = 40. 51 g O 2 Based on: 47. 0 g H 2 O H 2 O = 125. 3 g O 2 Question if only 35. 2 g of O 2 were recovered, what was the percent yield? 21

If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 g 47. 0 g Based on: 120. 0 g KO 2 = 40. 51 g O 2 Based on: 47. 0 g H 2 O H 2 O = 125. 3 g O 2 Determine how many grams of Water were left over. The Difference between the above amounts is directly RELATED to the XS H 2 O. 125. 3 - 40. 51 = 84. 79 g of O 2 that could have been formed from the XS water. 84. 79 g O 2 = 31. 83 g XS H 2 O 22
- Slides: 22