STOICHIOMETRY TUTORIAL Paul Gilletti 1 Instructions This is































- Slides: 49

STOICHIOMETRY TUTORIAL Paul Gilletti 1

Instructions: This is a work along tutorial. Each time you click the mouse or touch the space bar on your computer, one step of the problem solving occurs. Pressing the PAGE UP key will backup the steps. Get a pencil and paper, a periodic table and a calculator, and let’s get to work. 2

(1 -2 -3) General Approach For Problem Solving: 1. Clearly identify the Goal or Goals and the UNITS involved. (What are you trying to do? ) 2. Determine what is given and the UNITS. 3. Use conversion factors (which are really ratios) and their UNITS to CONVERT what is given into what is desired. 3

Table of Contents: Click on each tab to view problem types. View Complete Slide Show Sample problem 1 Sample problem 2 Converting grams to moles Mole to Mole Conversions Gram-Mole and Gram-Gram Problems Solution Stoichiometry Problems Limiting/Excess/ Reactant and Theoretical Yield Problems : 4

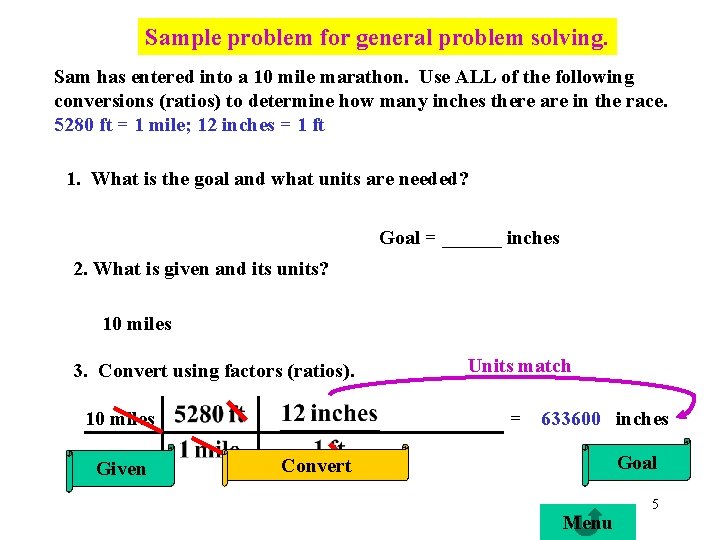
Sample problem for general problem solving. Sam has entered into a 10 mile marathon. Use ALL of the following conversions (ratios) to determine how many inches there are in the race. 5280 ft = 1 mile; 12 inches = 1 ft 1. What is the goal and what units are needed? Goal = ______ inches 2. What is given and its units? 10 miles 3. Convert using factors (ratios). 10 miles Given Units match = 633600 inches Goal Convert Menu 5

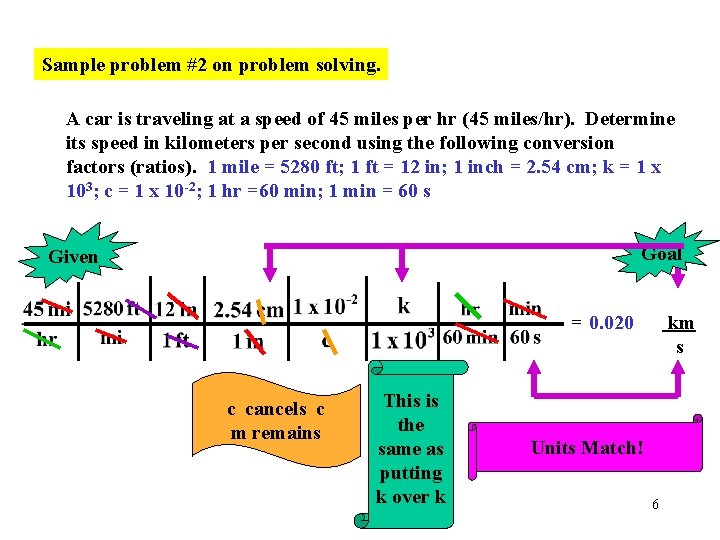
Sample problem #2 on problem solving. A car is traveling at a speed of 45 miles per hr (45 miles/hr). Determine its speed in kilometers per second using the following conversion factors (ratios). 1 mile = 5280 ft; 1 ft = 12 in; 1 inch = 2. 54 cm; k = 1 x 103; c = 1 x 10 -2; 1 hr =60 min; 1 min = 60 s Goal Given = 0. 020 c cancels c m remains This is the same as putting k over k km s Units Match! 6

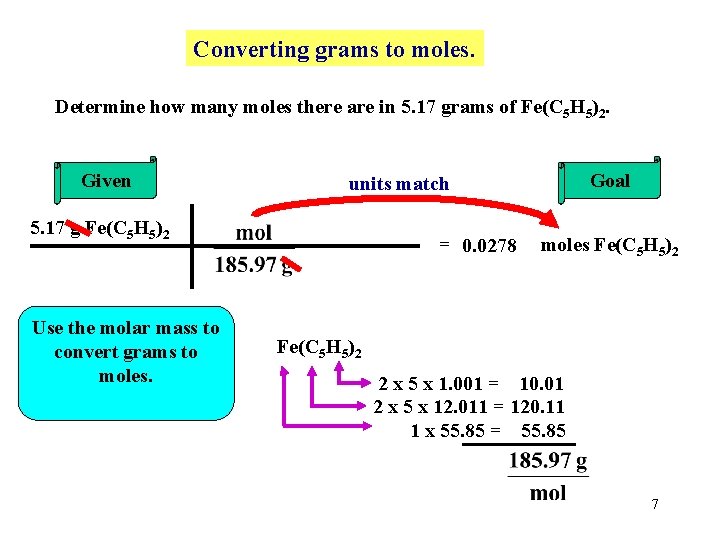
Converting grams to moles. Determine how many moles there are in 5. 17 grams of Fe(C 5 H 5)2. Given 5. 17 g Fe(C 5 H 5)2 Use the molar mass to convert grams to moles. Goal units match = 0. 0278 moles Fe(C 5 H 5)2 2 x 5 x 1. 001 = 10. 01 2 x 5 x 12. 011 = 120. 11 1 x 55. 85 = 55. 85 7

Stoichiometry (more working with ratios) Ratios are found within a chemical equation. 2 HCl + 1 Ba(OH)2 2 H 2 O + 1 Ba. Cl 2 coefficients give MOLAR RATIOS 2 moles of HCl react with 1 mole of Ba(OH)2 to form 2 moles of H 2 O and 1 mole of Ba. Cl 2 8

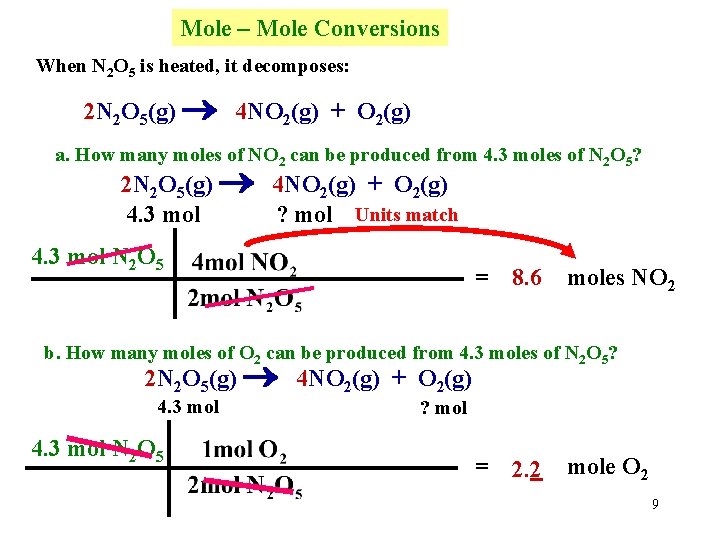
Mole – Mole Conversions When N 2 O 5 is heated, it decomposes: 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) a. How many moles of NO 2 can be produced from 4. 3 moles of N 2 O 5? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 4. 3 mol ? mol Units match 4. 3 mol N 2 O 5 = 8. 6 moles NO 2 b. How many moles of O 2 can be produced from 4. 3 moles of N 2 O 5? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 4. 3 mol N 2 O 5 ? mol = 2. 2 mole O 2 9

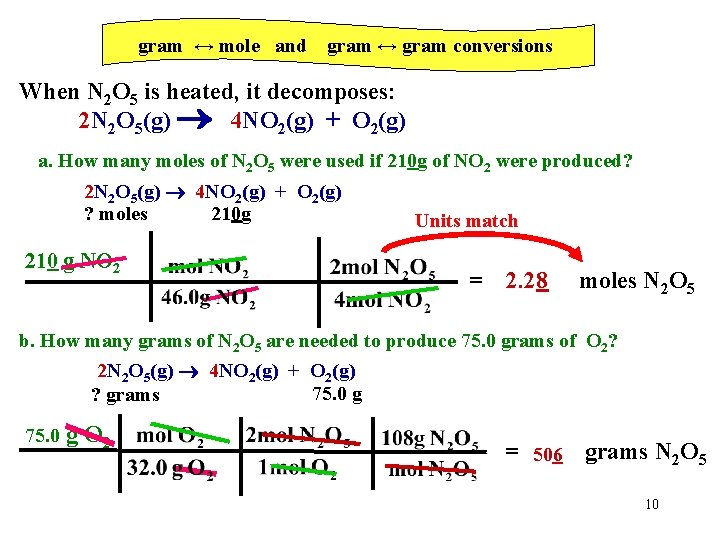
gram ↔ mole and gram ↔ gram conversions When N 2 O 5 is heated, it decomposes: 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) a. How many moles of N 2 O 5 were used if 210 g of NO 2 were produced? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) ? moles 210 g 210 g NO 2 Units match = 2. 28 moles N 2 O 5 b. How many grams of N 2 O 5 are needed to produce 75. 0 grams of O 2? 2 N 2 O 5(g) 4 NO 2(g) + O 2(g) 75. 0 g ? grams 75. 0 g O 2 = 506 grams N 2 O 5 10

Gram to Gram Conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + 3 H 2(g) First write a balanced equation. 11

Gram to Gram Conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + ? grams 3. 45 g 3 H 2(g) Now let’s get organized. Write the information below the substances. 12

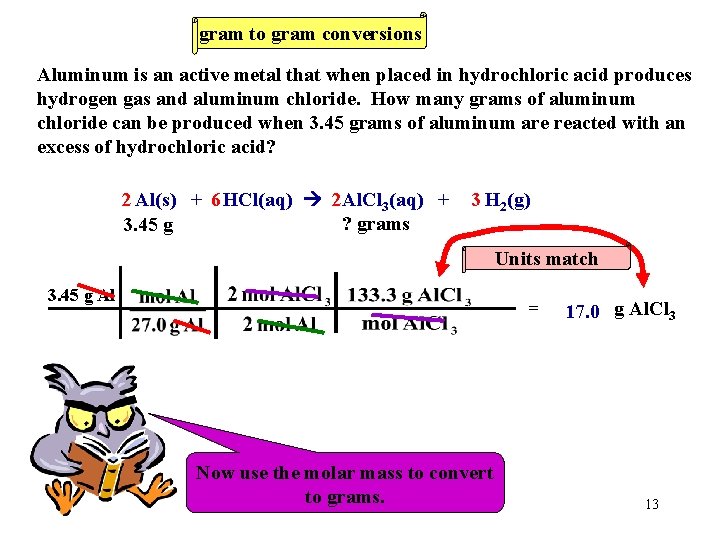
gram to gram conversions Aluminum is an active metal that when placed in hydrochloric acid produces hydrogen gas and aluminum chloride. How many grams of aluminum chloride can be produced when 3. 45 grams of aluminum are reacted with an excess of hydrochloric acid? 2 Al(s) + 6 HCl(aq) 2 Al. Cl 3(aq) + ? grams 3. 45 g 3 H 2(g) Units match 3. 45 g Al = Now We must Now use Let’s the always use work molar thethe convert molar mass problem. ratio. to toconvert moles. to grams. 17. 0 g Al. Cl 3 13

14

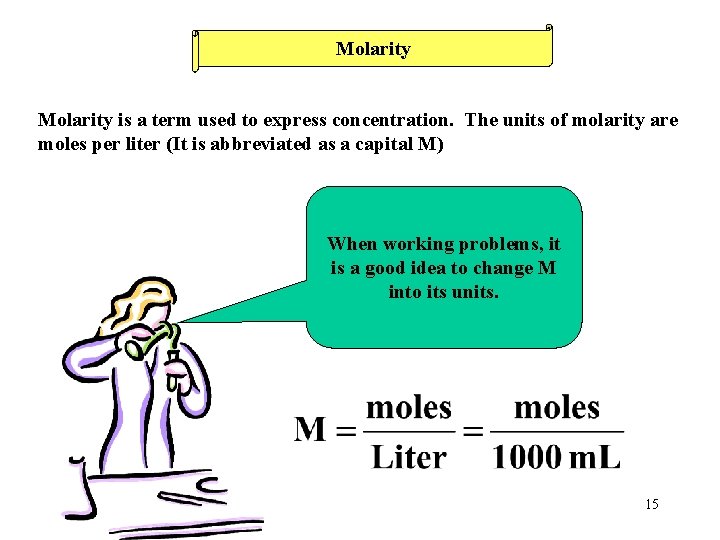
Molarity is a term used to express concentration. The units of molarity are moles per liter (It is abbreviated as a capital M) When working problems, it is a good idea to change M into its units. 15

16

Solutions A solution is prepared by dissolving 3. 73 grams of Al. Cl 3 in water to form 200. 0 m. L solution. A 10. 0 m. L portion of the solution is then used to prepare 100. 0 m. L of solution. Determine the molarity of the final solution. What type of problem(s) is this? Molarity followed by dilution. 17

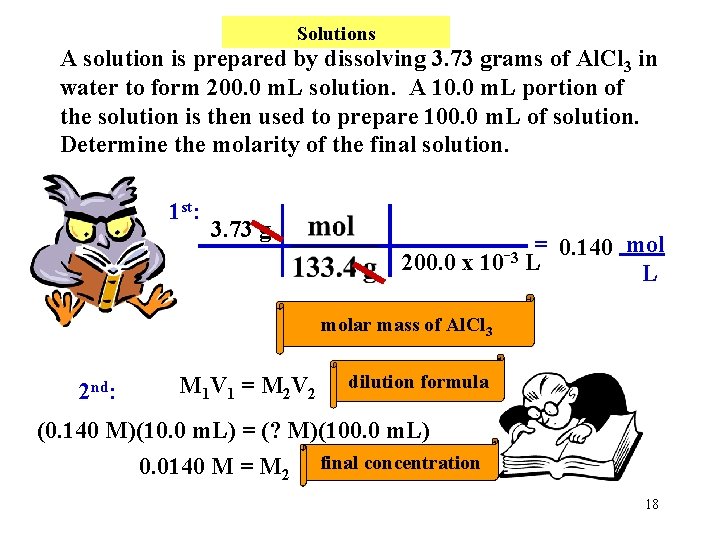
Solutions A solution is prepared by dissolving 3. 73 grams of Al. Cl 3 in water to form 200. 0 m. L solution. A 10. 0 m. L portion of the solution is then used to prepare 100. 0 m. L of solution. Determine the molarity of the final solution. 1 st: 3. 73 g = 0. 140 mol 3 200. 0 x 10 L L molar mass of Al. Cl 3 2 nd: M 1 V 1 = M 2 V 2 dilution formula (0. 140 M)(10. 0 m. L) = (? M)(100. 0 m. L) 0. 0140 M = M 2 final concentration 18

19

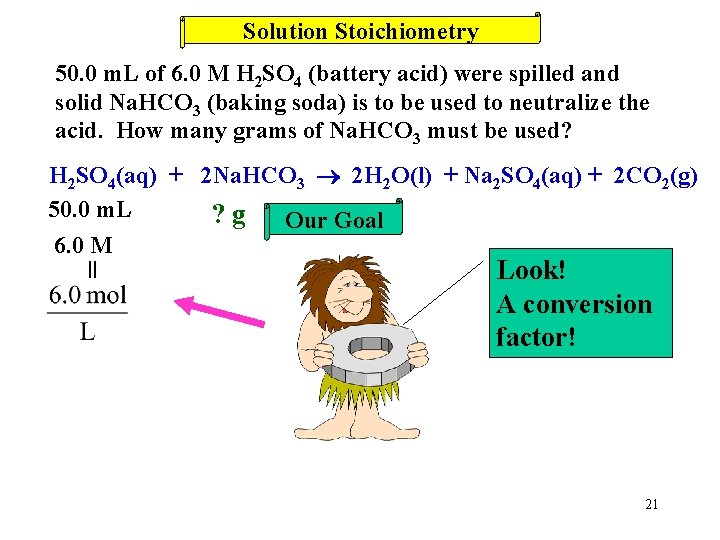
Solution Stoichiometry 50. 0 m. L of 6. 0 M H 2 SO 4 (battery acid) were spilled and solid Na. HCO 3 (baking soda) is to be used to neutralize the acid. How many grams of Na. HCO 3 must be used? H 2 SO 4(aq) + 2 Na. HCO 3 2 H 2 O(l) + Na 2 SO 4(aq) + 2 CO 2(g) 20

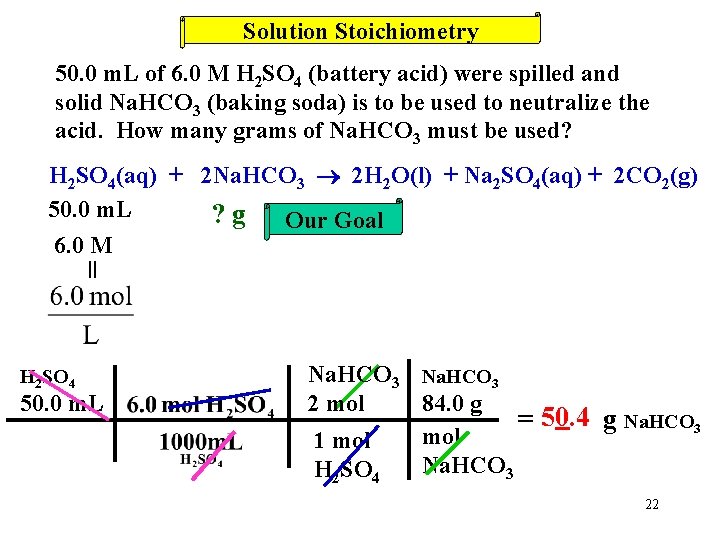
Solution Stoichiometry 50. 0 m. L of 6. 0 M H 2 SO 4 (battery acid) were spilled and solid Na. HCO 3 (baking soda) is to be used to neutralize the acid. How many grams of Na. HCO 3 must be used? H 2 SO 4(aq) + 2 Na. HCO 3 2 H 2 O(l) + Na 2 SO 4(aq) + 2 CO 2(g) 50. 0 m. L ? g Our Goal 6. 0 M = Look! A conversion factor! 21

Solution Stoichiometry 50. 0 m. L of 6. 0 M H 2 SO 4 (battery acid) were spilled and solid Na. HCO 3 (baking soda) is to be used to neutralize the acid. How many grams of Na. HCO 3 must be used? H 2 SO 4(aq) + 2 Na. HCO 3 2 H 2 O(l) + Na 2 SO 4(aq) + 2 CO 2(g) 50. 0 m. L ? g Our Goal 6. 0 M = H 2 SO 4 50. 0 m. L Na. HCO 3 84. 0 g 2 mol = 50. 4 g Na. HCO 3 mol 1 mol Na. HCO 3 H 2 SO 4 22

23

Solution Stoichiometry: Determine how many m. L of 0. 102 M Na. OH solution are needed to neutralize 35. 0 m. L of 0. 125 M H 2 SO 4 solution. 2 1 2 SO 4 ____Na. OH + ____H 2 2 O ____H + 1 2 SO 4 ____Na First write a balanced Equation. 24

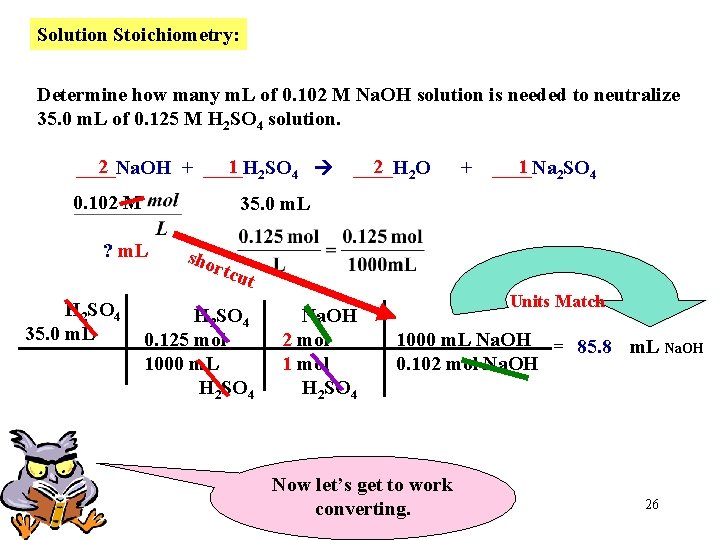
Solution Stoichiometry: Determine how many m. L of 0. 102 M Na. OH solution is needed to neutralize 35. 0 m. L of 0. 125 M H 2 SO 4 solution. 2 1 2 SO 4 ____Na. OH + ____H 0. 102 M 2 2 O ____H + 1 2 SO 4 ____Na 35. 0 m. L ? m. L Our Goal Since 1 L = 1000 m. L, we can use this to save on the number of conversions Now, let’s get organized. Place numerical Information and accompanying UNITS below each compound. 25

Solution Stoichiometry: Determine how many m. L of 0. 102 M Na. OH solution is needed to neutralize 35. 0 m. L of 0. 125 M H 2 SO 4 solution. 2 1 2 SO 4 ____Na. OH + ____H 0. 102 M + 1 2 SO 4 ____Na 35. 0 m. L ? m. L H 2 SO 4 35. 0 m. L 2 2 O ____H sho rtcu t H 2 SO 4 0. 125 mol 1000 m. L H 2 SO 4 Na. OH 2 mol 1 mol H 2 SO 4 Units Match 1000 m. L Na. OH = 85. 8 m. L Na. OH 0. 102 mol Na. OH Now let’s get to work converting. 26

27

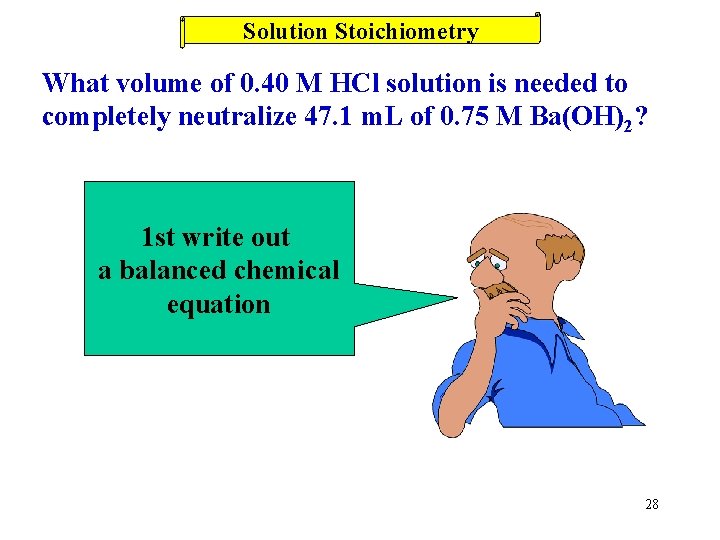
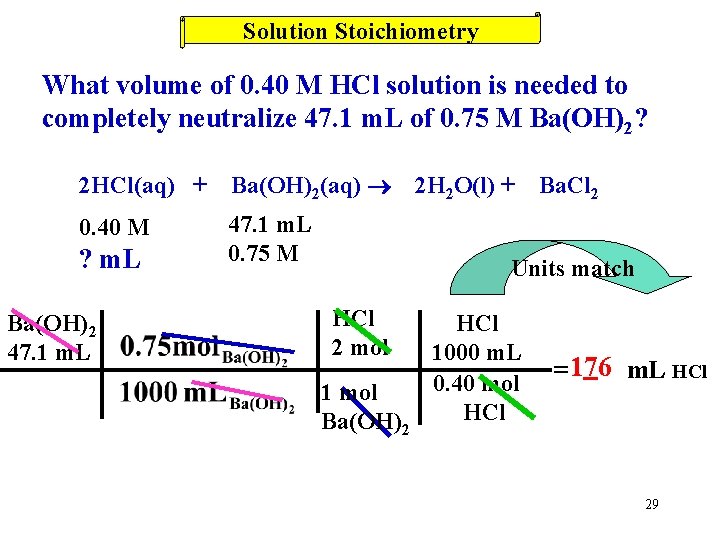
Solution Stoichiometry What volume of 0. 40 M HCl solution is needed to completely neutralize 47. 1 m. L of 0. 75 M Ba(OH)2? 1 st write out a balanced chemical equation 28

Solution Stoichiometry What volume of 0. 40 M HCl solution is needed to completely neutralize 47. 1 m. L of 0. 75 M Ba(OH)2? 2 HCl(aq) + Ba(OH)2(aq) 0. 40 M 47. 1 m. L 0. 75 M ? m. L Ba(OH)2 47. 1 m. L 2 H 2 O(l) + Ba. Cl 2 Units match HCl 2 mol 1 mol Ba(OH)2 HCl 1000 m. L 0. 40 mol HCl = 176 m. L HCl 29

30

31

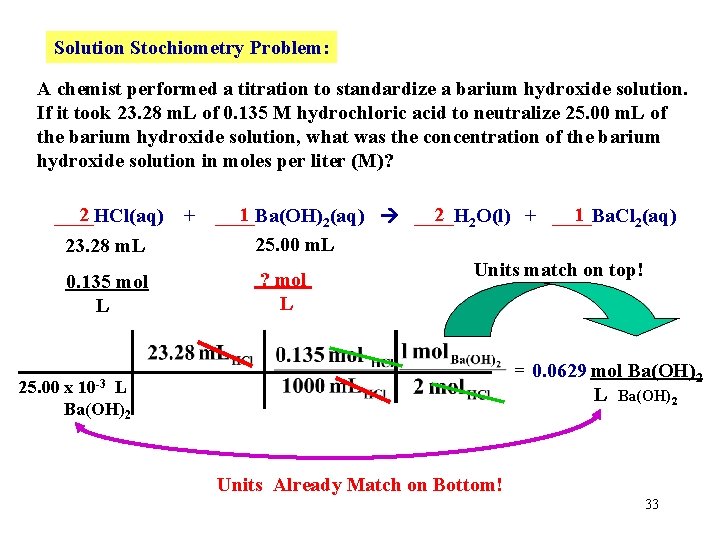
Solution Stochiometry Problem: A chemist performed a titration to standardize a barium hydroxide solution. If it took 23. 28 m. L of 0. 135 M hydrochloric acid to neutralize 25. 00 m. L of the barium hydroxide solution, what was the concentration of the barium hydroxide solution in moles per liter (M)? 2 ____HCl(aq) 23. 28 m. L 0. 135 mol L + 1 2 2 O(l) + ____Ba. Cl 1 ____Ba(OH) 2(aq) ____H 2(aq) 25. 00 m. L ? mol L First write a balanced chemical reaction. 32

Solution Stochiometry Problem: A chemist performed a titration to standardize a barium hydroxide solution. If it took 23. 28 m. L of 0. 135 M hydrochloric acid to neutralize 25. 00 m. L of the barium hydroxide solution, what was the concentration of the barium hydroxide solution in moles per liter (M)? 2 ____HCl(aq) 23. 28 m. L 0. 135 mol L + 1 2 2 O(l) + ____Ba. Cl 1 ____Ba(OH) 2(aq) ____H 2(aq) 25. 00 m. L Units match on top! ? mol L = 0. 0629 mol Ba(OH)2 L Ba(OH)2 10 -3 25. 00 x L Ba(OH)2 Units Already Match on Bottom! 33

34

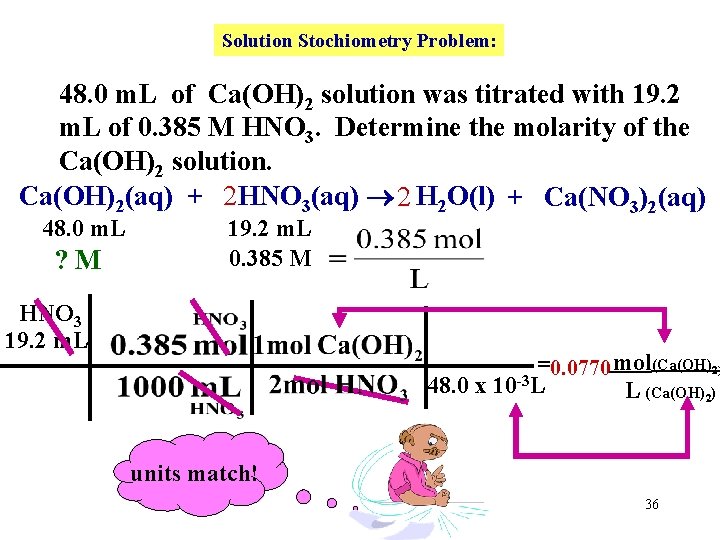
Solution Stochiometry Problem: 48. 0 m. L of Ca(OH)2 solution was titrated with 19. 2 m. L of 0. 385 M HNO 3. Determine the molarity of the Ca(OH)2 solution. We must first write a balanced equation. 35

Solution Stochiometry Problem: 48. 0 m. L of Ca(OH)2 solution was titrated with 19. 2 m. L of 0. 385 M HNO 3. Determine the molarity of the Ca(OH)2 solution. Ca(OH)2(aq) + 2 HNO 3(aq) 2 H 2 O(l) + Ca(NO 3)2(aq) 48. 0 m. L ? M 19. 2 m. L 0. 385 M HNO 3 19. 2 m. L =0. 0770 mol(Ca(OH)2) 48. 0 x 10 -3 L L (Ca(OH)2) units match! 36

37

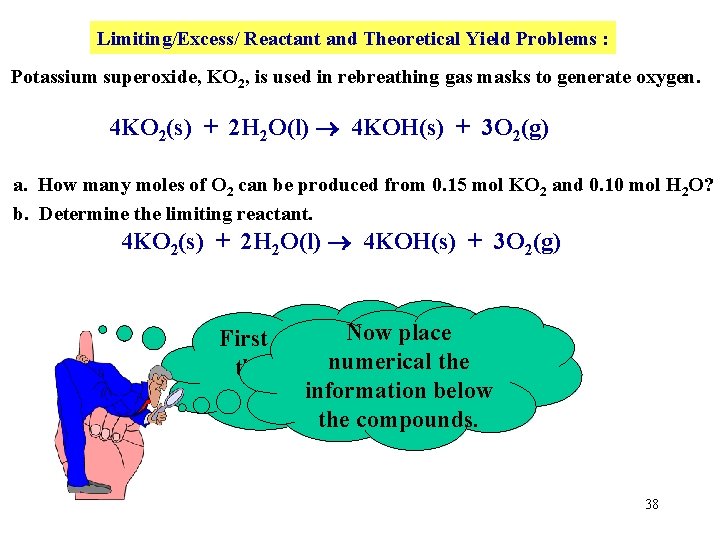
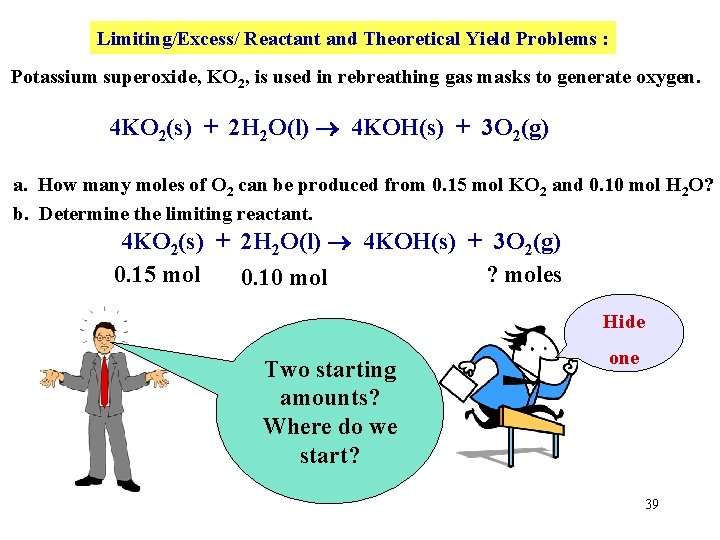
Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) Nowthe place First copy down numerical the BALANCED information below equation! the compounds. 38

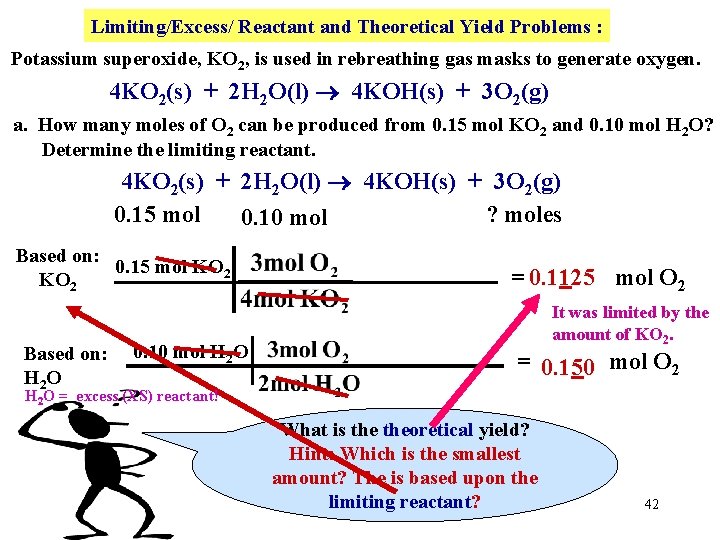
Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles 0. 10 mol Hide Two starting amounts? Where do we start? one 39

Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles Hide 0. 10 mol Based on: 0. 15 mol KO 2 = 0. 1125 mol O 2 40

Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? b. Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles Hide 0. 10 mol Based on: 0. 15 mol KO 2 Based on: 0. 10 mol H 2 O H 2 O = 0. 1125 mol O 2 = 0. 150 mol O 2 41

Limiting/Excess/ Reactant and Theoretical Yield Problems : Potassium superoxide, KO 2, is used in rebreathing gas masks to generate oxygen. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) a. How many moles of O 2 can be produced from 0. 15 mol KO 2 and 0. 10 mol H 2 O? Determine the limiting reactant. 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) 0. 15 mol ? moles 0. 10 mol Based on: 0. 15 mol KO 2 Based on: H 2 O 0. 10 mol H 2 O = 0. 1125 mol O 2 It was limited by the amount of KO 2. = 0. 150 mol O 2 H 2 O = excess (XS) reactant! What is theoretical yield? Hint: Which is the smallest amount? The is based upon the limiting reactant? 42

Theoretical yield vs. Actual yield Suppose theoretical yield for an experiment was calculated to be 19. 5 grams, and the experiment was performed, but only 12. 3 grams of product were recovered. Determine the % yield. Theoretical yield = 19. 5 g based on limiting reactant Actual yield = 12. 3 g experimentally recovered 43

Limiting/Excess Reactant Problem with % Yield 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 g 47. 0 one g Hide Based on: 120. 0 g KO 2 = 40. 51 g O 2 44

Limiting/Excess Reactant Problem with % Yield 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 47. 0 g Hideg Based on: 120. 0 g KO 2 = 40. 51 g O 2 Based on: 47. 0 g H 2 O H 2 O = 125. 3 g O 2 Question if only 35. 2 g of O 2 were recovered, what was the percent yield? 45

If a reaction vessel contains 120. 0 g of KO 2 and 47. 0 g of H 2 O, how many grams of O 2 can be produced? 4 KO 2(s) + 2 H 2 O(l) 4 KOH(s) + 3 O 2(g) ? g 120. 0 g 47. 0 g Based on: 120. 0 g KO 2 = 40. 51 g O 2 Based on: 47. 0 g H 2 O H 2 O = 125. 3 g O 2 Determine how many grams of Water were left over. The Difference between the above amounts is directly RELATED to the XS H 2 O. 125. 3 - 40. 51 = 84. 79 g of O 2 that could have been formed from the XS water. 84. 79 g O 2 = 31. 83 g XS H 2 O 46

47

Try this problem (then check your answer): Calculate the molarity of a solution prepared by dissolving 25. 6 grams of Al(NO 3)3 in 455 m. L of solution. After you have worked the problem, click here to see setup answer 48

49