Stoichiometry Stoichiometry The study of quantitative relationships between
Stoichiometry

Stoichiometry • The study of quantitative relationships between amounts of reactants used and products formed • Stoichiometry is based on the Law of Conservation of Matter Example: 2 H 2 + O 2 2 H 2 O reads stoichiometrically as “ 2 moles of hydrogen + 1 mole of oxygen make 2 moles of H 2 O”

Steps for Solving Stoichiometry 1. Write & balance chemical equation 2. Determine mole ratio using coefficients from balanced equation 3. Write your conversion factors and check and cancel units 4. Calculate 5. Adjust for significant figures & include units

How many moles of CO 2 are created from the combustion of 5. 0 moles of propane (C 3 H 8)? . Step 1: Write & balance equation C 3 H 8 +O 2 H 2 O + CO 2 C 3 H 8 +5 O 2 4 H 2 O + 3 CO 2 Step 2: Determine “given”, “asked for”, and mole ratio 1 mole C 3 H 8 = 3 moles CO 2

Step 3: Write your conversion factors and check/cancel units 5. 0 mol C 3 H 8 3 mol CO 2 1 mol C 3 H 8 Step 4: Calculate 5. 0 X 3 = 15 Step 5: Sig Figs and units 15 moles CO 2

Mole-Mass Problem Moles A Moles B Mass B
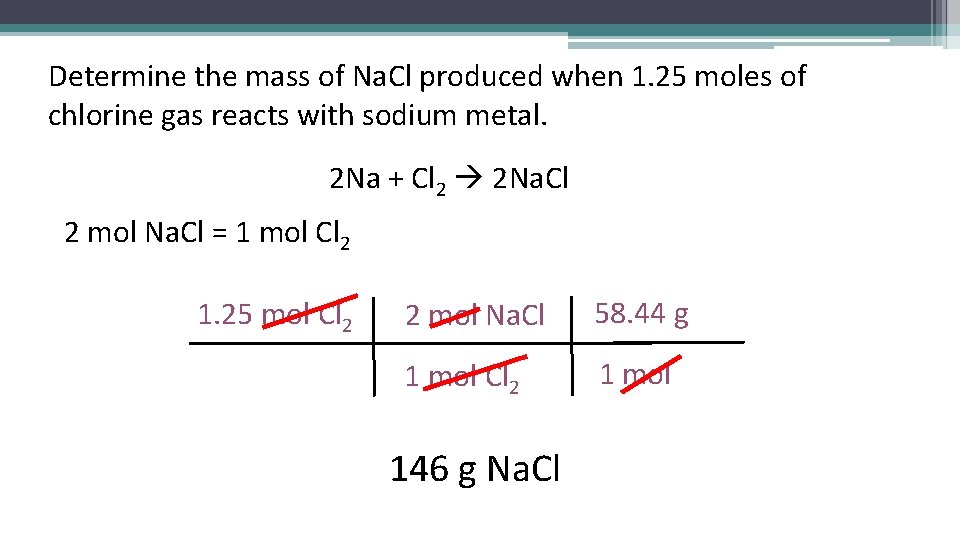
Determine the mass of Na. Cl produced when 1. 25 moles of chlorine gas reacts with sodium metal. 2 Na + Cl 2 2 Na. Cl 2 mol Na. Cl = 1 mol Cl 2 1. 25 mol Cl 2 2 mol Na. Cl 58. 44 g 1 mol Cl 2 1 mol 146 g Na. Cl

Mass-Mass Problems Mass A Moles A Moles B Mass B
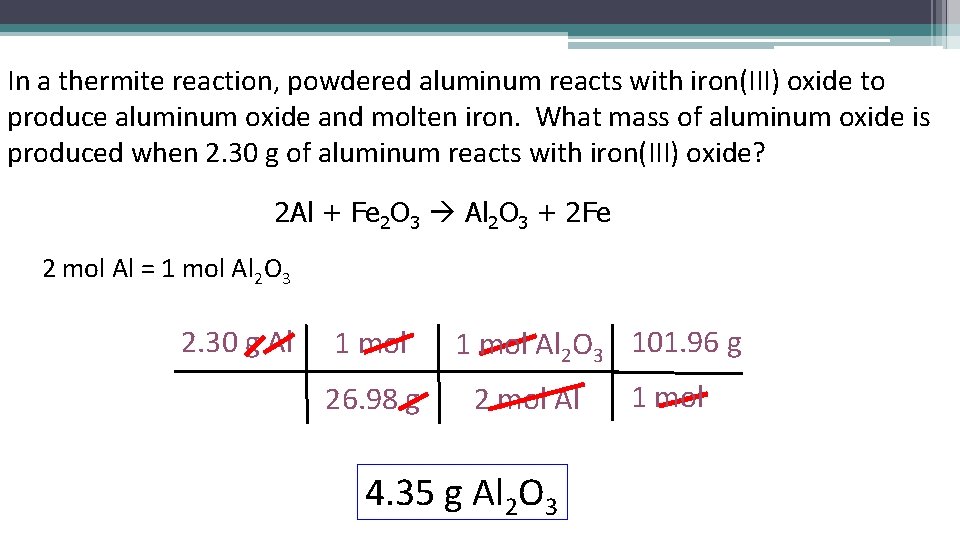
In a thermite reaction, powdered aluminum reacts with iron(III) oxide to produce aluminum oxide and molten iron. What mass of aluminum oxide is produced when 2. 30 g of aluminum reacts with iron(III) oxide? 2 Al + Fe 2 O 3 Al 2 O 3 + 2 Fe 2 mol Al = 1 mol Al 2 O 3 2. 30 g Al 1 mol 26. 98 g 1 mol Al 2 O 3 101. 96 g 2 mol Al 4. 35 g Al 2 O 3 1 mol

Volume-Volume Problem Volume of substance A Use 1 mol = 22. 4 L as conversion factor. Volume A Moles A Moles B Volume B Use 1 mol = 22. 4 L as conversion factor. Volume of substance B

What volume of hydrogen gas, along with nitrogen gas, is necessary to produce 16. 0 L of ammonia gas (NH 3) at STP? N 2 + 3 H 2 2 NH 3 2 mol NH 3 = 3 mol H 2 16. 0 L NH 3 1 mol 3 mol H 2 22. 4 L 2 mol NH 3 1 mol 24. 0 L H 2

practice How many moles of bromine are produced when fluorine reacts with 347. 5 g of potassium bromide? F 2 + 2 KBr Br 2 + 2 KF 2 mol KBr = 1 mol Br 2 347. 5 g KBr 1 mol Br 2 119 g 2 mol KBr 1. 460 mol Br 2
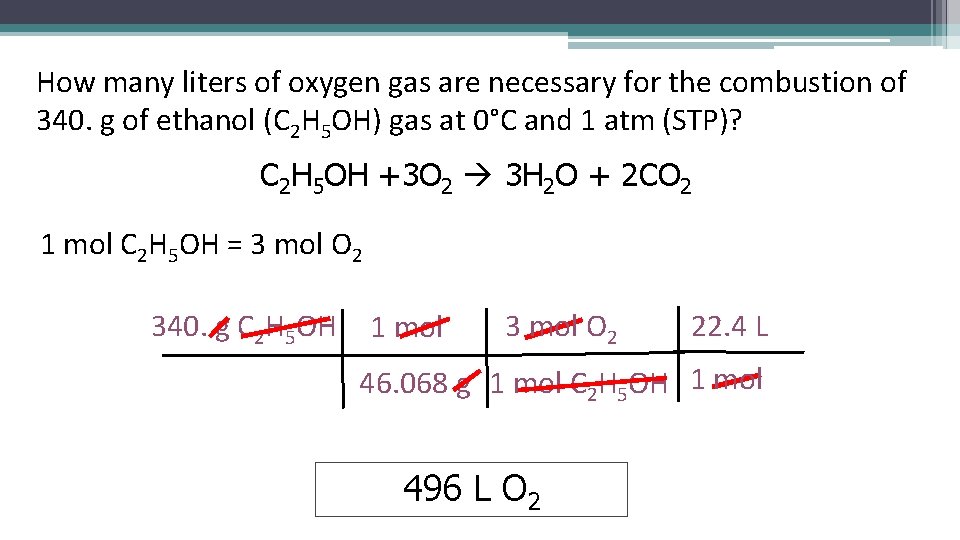
How many liters of oxygen gas are necessary for the combustion of 340. g of ethanol (C 2 H 5 OH) gas at 0°C and 1 atm (STP)? C 2 H 5 OH +3 O 2 3 H 2 O + 2 CO 2 1 mol C 2 H 5 OH = 3 mol O 2 340. g C 2 H 5 OH 1 mol 3 mol O 2 22. 4 L 46. 068 g 1 mol C 2 H 5 OH 1 mol 496 L O 2

How many atoms of silver can be produced with 2. 50 g copper reacting with silver nitrate? Cu + 2 Ag. NO 3 Cu(NO 3)2 + 2 Ag 1 mol Cu = 2 mol Ag 2. 50 g Cu 1 mol 2 mol Ag 6. 022 x 1023 atoms 63. 55 g 1 mol Cu 1 mol 4. 74 x 1022 atoms Ag
- Slides: 14