Stoichiometry Stoichiometry The Proportional relationship between two or

Stoichiometry

Stoichiometry The Proportional relationship between two or more substances during a chemical reaction.

4 Simple rules 1. Balance the Equation 2. Convert to moles 3. Use the mole ratio 4. Convert to desired units

4 Simple rules 1. Balance the Equation You need to know what you start with and what you need to end with.
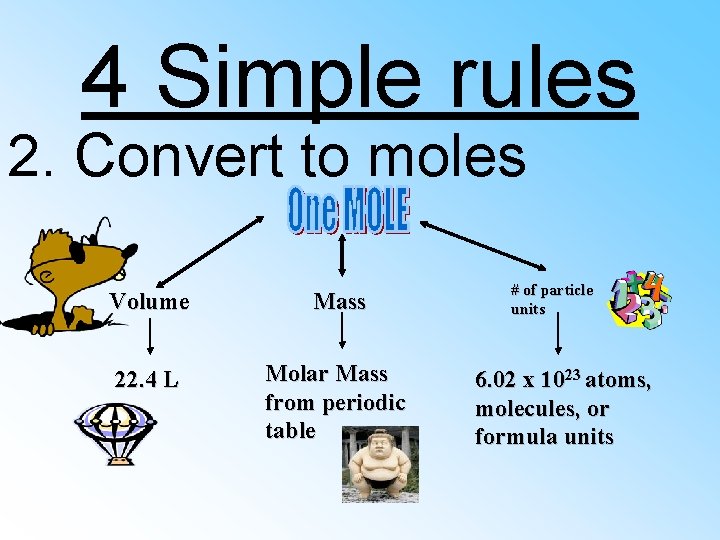
4 Simple rules 2. Convert to moles Volume Mass 22. 4 L Molar Mass from periodic table # of particle units 6. 02 x 1023 atoms, molecules, or formula units

4 Simple rules 3. Use the mole ratio Where The mole does ratio the mole comes ratio from the come coefficients from? in a balanced Chemical Equation

4 Simple rules 3. Use the mole ratio Fe 2 O + 2 Li Li 2 O + 2 Fe What is the ratio between Fe and Fe 2 O?

4 Simple rules 3. Use the mole ratio Fe 2 O + 2 Li Li 2 O + 2 Fe What is the ratio between Fe and Li?

4 Simple rules 3. Use the mole ratio The coefficients can represent # of moles or # of molecules.

4 Simple rules 4. Convert to desired units If grams are desired use mass from P. T. If you need liters use 22. 4 L If you need atoms or molecules 6. 02 x 1023 If moles are what is needed you are already done.
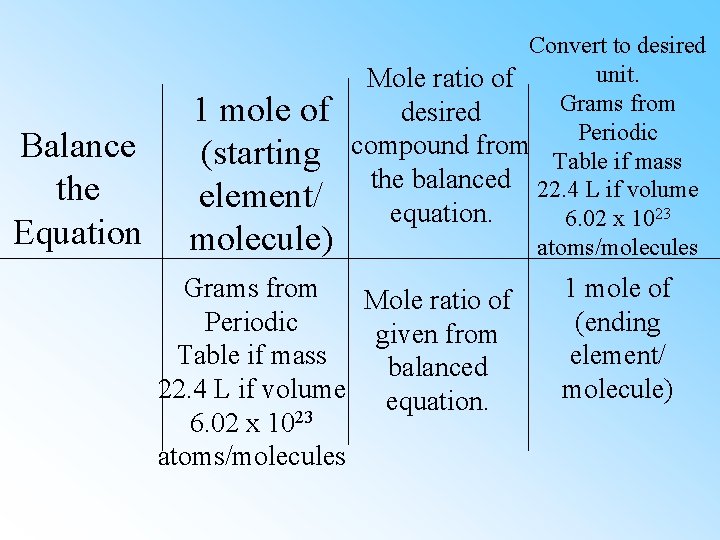
Balance the Equation 1 mole of (starting element/ molecule) Convert to desired unit. Mole ratio of Grams from desired Periodic compound from Table if mass the balanced 22. 4 L if volume equation. 6. 02 x 1023 atoms/molecules Grams from Mole ratio of Periodic given from Table if mass balanced 22. 4 L if volume equation. 6. 02 x 1023 atoms/molecules 1 mole of (ending element/ molecule)
- Slides: 11