Stoichiometry Stoichiometry of Hamburgers Recipe for a bacon

Stoichiometry!!!

Stoichiometry of Hamburgers! Recipe for a bacon double cheeseburger is: ¬ 1 hamburger bun (top & bottom) ¬ 2 hamburger patties ¬ 2 slices of cheese ¬ 4 strips of bacon

Stoichiometry of a Hamburger Recipe for a bacon double cheeseburger is: ¬ 1 hamburger bun ¬ 2 hamburger patties ¬ 2 slices of cheese ¬ 4 strips of bacon Based on this recipe; 1. If I have five bacon double cheeseburgers; a) How many hamburger buns do I have? b) How many hamburger patties do I have? c) How many slices of cheese do I have? d) How many strips of bacon do I have 2. How many bacon double cheeseburgers can you make if you start with; a) 1 bun, 2 patties, 2 slices of cheese, 4 strips bacon b) 2 buns, 4 patties, 4 slices of cheese, 8 strips of bacon c) 1 dozen buns, 2 dozen patties, 2 dozen slices of cheese, 4 dozen bacon strips d) 1 mole buns, 2 mole patties, 2 mole slices of cheese, 4 mole bacon strips e) 10 buns, 20 patties, 2 slices of cheese, 40 strips of bacon 3

And for dessert? 4

Mock Apple Pie! Filling 2 cups water 3/4 cup granulated sugar 2 tsp. cream of tartar 30 Ritz Crackers 1 ready-to-use refrigerated pie crust (1/2 of 425 -g pkg. ) 1 Tbsp. lemon juice 1 tsp. ground cinnamon Topping 25 Ritz Crackers, crushed (about 1 cup) 1/2 cup packed brown sugar 1/2 tsp. ground cinnamon 5

Chocolate Chip Cookies!! 1 cup butter 1/2 cup white sugar 1 cup packed brown sugar 1 teaspoon vanilla extract 2 eggs 2 1/2 cups all-purpose flour 1 teaspoon baking soda 1 teaspoon salt 2 cups semisweet chocolate chips Makes 3 dozen How many eggs are needed to make 3 dozen cookies? How much butter is needed for the amount of chocolate chips used? How many eggs would we need to make 9 dozen cookies? How much brown sugar would I need if I had 1 cup white sugar?

Cookies and Chemistry…Huh!? !? � Just like chocolate chip cookies have recipes, chemists have recipes as well. • Instead of calling them recipes, we call them reaction equations • Furthermore, instead of using cups and teaspoons, we use mole • Lastly, instead of eggs, butter, sugar, etc. we use chemical compounds as ingredients.

Recipes, Balanced Equations and Stoichiometry �A recipe and a chemical equation are virtually the SAME THING!!! �You need an accurate recipe to make cookies, pies, Hungarian goulash… whatever. �For stoichiometry, you MUST start with a BALANCED CHEMICAL EQUATION! �A recipe is based on ratios (eggs to milk to flour, etc. ), and so is a balance equation. 8

Stoichiometry �Stoichiometry is the study of quantitative relationships between amounts of reactants and products formed by a chemical reaction. It’s the RATIOS of Reactants and Products! ou MUST have a BALANCED CHEMICAL EQUATIO 2 ___H 2 + ___O 2 2 ___H 2 O For this reaction, 2 moles of hydrogen gas reacts with 1 mole of oxygen gas to create 2 moles of water molecules.

Stoichiometry- Moles 2 2 ___H 2(g) + ___O 2 (g) ___H 2 O (l) What is the ratio of hydrogen gas to oxygen gas? What is the ratio of oxygen gas to water? 2: 1 1: 2 What is the ratio of hydrogen gas to water? What is the ratio of water to oxygen gas? 2: 2 2: 1 10
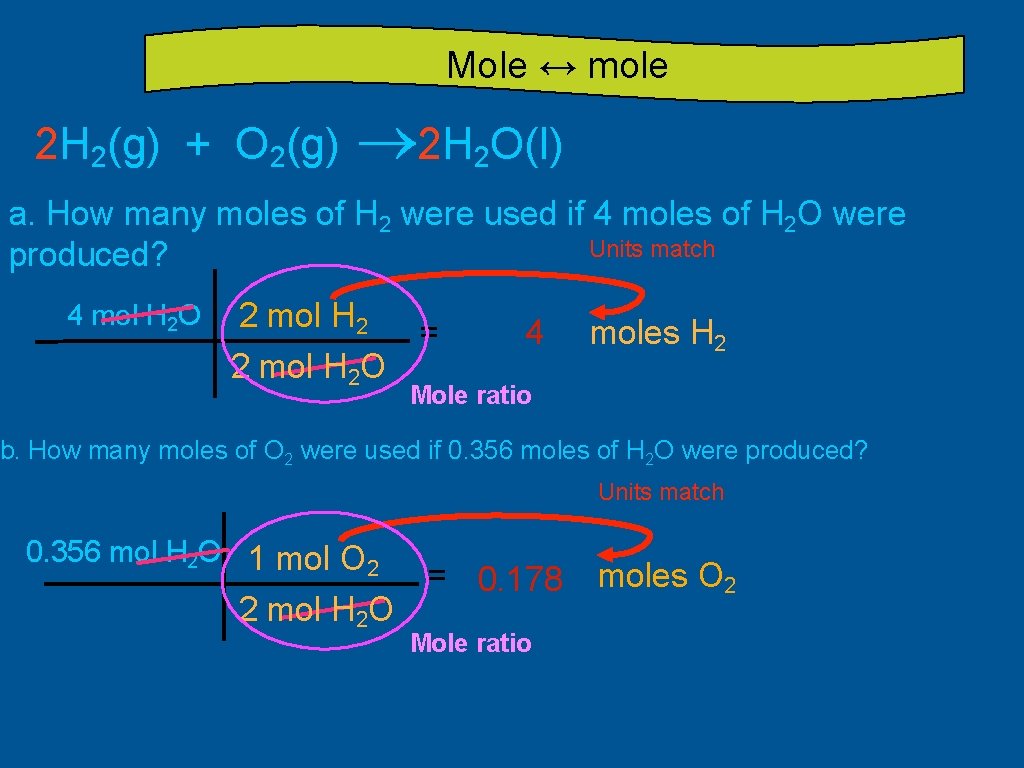
Mole ↔ mole 2 H 2(g) + O 2(g) ® 2 H 2 O(l) a. How many moles of H 2 were used if 4 moles of H 2 O were Units match produced? 4 mol H 2 O 2 mol H 2 = 2 mol H 2 O 4 moles H 2 Mole ratio b. How many moles of O 2 were used if 0. 356 moles of H 2 O were produced? Units match 0. 356 mol H 2 O 1 mol O 2 2 mol H 2 O = 0. 178 Mole ratio moles O 2
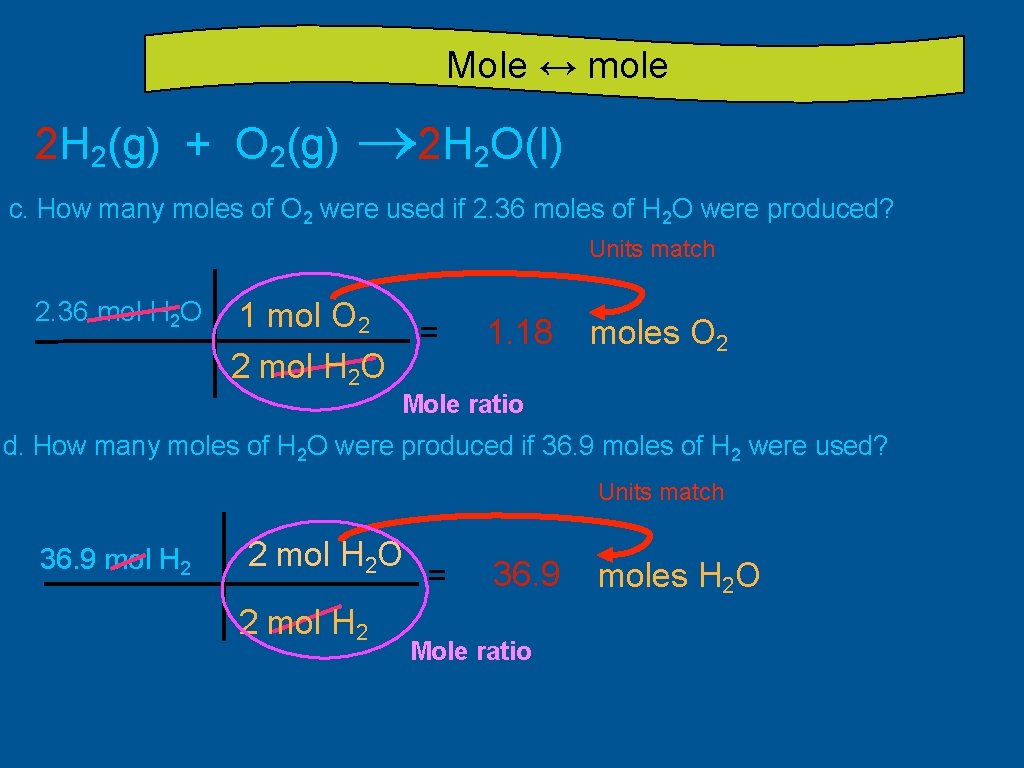
Mole ↔ mole 2 H 2(g) + O 2(g) ® 2 H 2 O(l) c. How many moles of O 2 were used if 2. 36 moles of H 2 O were produced? Units match 2. 36 mol H 2 O 1 mol O 2 = 2 mol H 2 O 1. 18 moles O 2 Mole ratio d. How many moles of H 2 O were produced if 36. 9 moles of H 2 were used? Units match 36. 9 mol H 2 2 mol H 2 O 2 mol H 2 = 36. 9 Mole ratio moles H 2 O

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? What is the first thing I must do to answer this question? GET THE FORMULA FOR EACH SUBSTANCE!! What do I do next? Balance the equation!

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® Ag 2 S 16 Ag + S 8 ® 8 Ag 2 S a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? WHY balance the equation? To get the mole ratios

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® Ag 2 S 16 Ag + S 8 ® 8 Ag 2 S a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? What mole ratio is used in this problem? 1 S 8 8 Ag 2 S 1 S 8

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® Ag 2 S 16 Ag + S 8 ® 8 Ag 2 S a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? WHY this ratio? 1 S 8 8 Ag 2 S S 8 needs to be on top because that is what the question is asking for.

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® Ag 2 S 16 Ag + S 8 ® 8 Ag 2 S a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? 1 S 8 8 Ag 2 S Now What? Put starting amounts WITH label in correct spot

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® 16 Ag + S 8 ® Ag 2 S 8 Ag 2 S a. How many moles of S 8 were used if 36 moles of Ag 2 S were produced? Units match 36 mol Ag 2 S 1 mol S 8 8 mol Ag 2 S = 4. 5 Mole ratio moles S 8

You Try!! Silver metal reacts with sulfur solid (S 8) to create silver I sulfide Ag + S 8 ® 16 Ag + S 8 ® Ag 2 S 8 Ag 2 S b. How many moles of Ag 2 S were produced if 7 moles of Ag were Units match used? 7 mol Ag 8 mol Ag 2 S = 3. 5 16 mol Ag Mole ratio moles Ag 2 S

Stoichiometry Part II Mr. Mole

Stoichiometry- Grams 2 2___H O (l) ___H (g) + ___O (g) 2 2 2 What is the molar mass of the hydrogen gas in this reaction? 2. 02 g What is the molar mass of water? 18. 02 g What is the molar mass of oxygen gas? 32 g
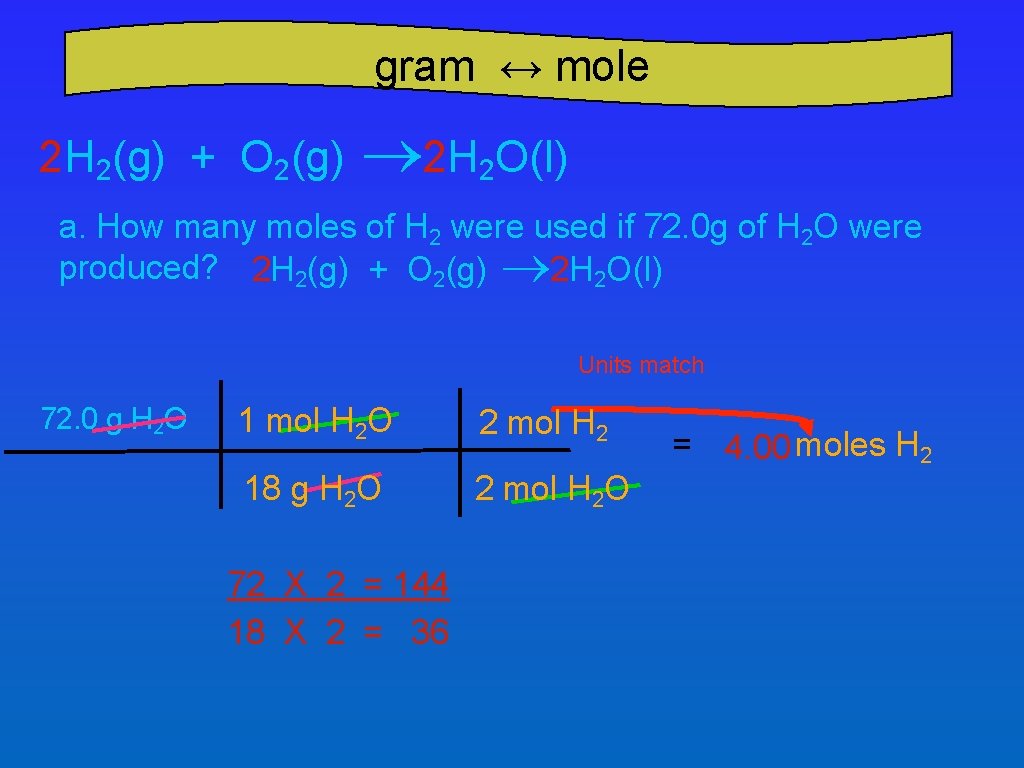
gram ↔ mole 2 H 2(g) + O 2(g) ® 2 H 2 O(l) a. How many moles of H 2 were used if 72. 0 g of H 2 O were produced? 2 H 2(g) + O 2(g) ® 2 H 2 O(l) Units match 72. 0 g H 2 O 1 mol H 2 O 2 mol H 2 18 g H 2 O 2 mol H 2 O 72 X 2 = 144 18 X 2 = 36 = 4. 00 moles H 2
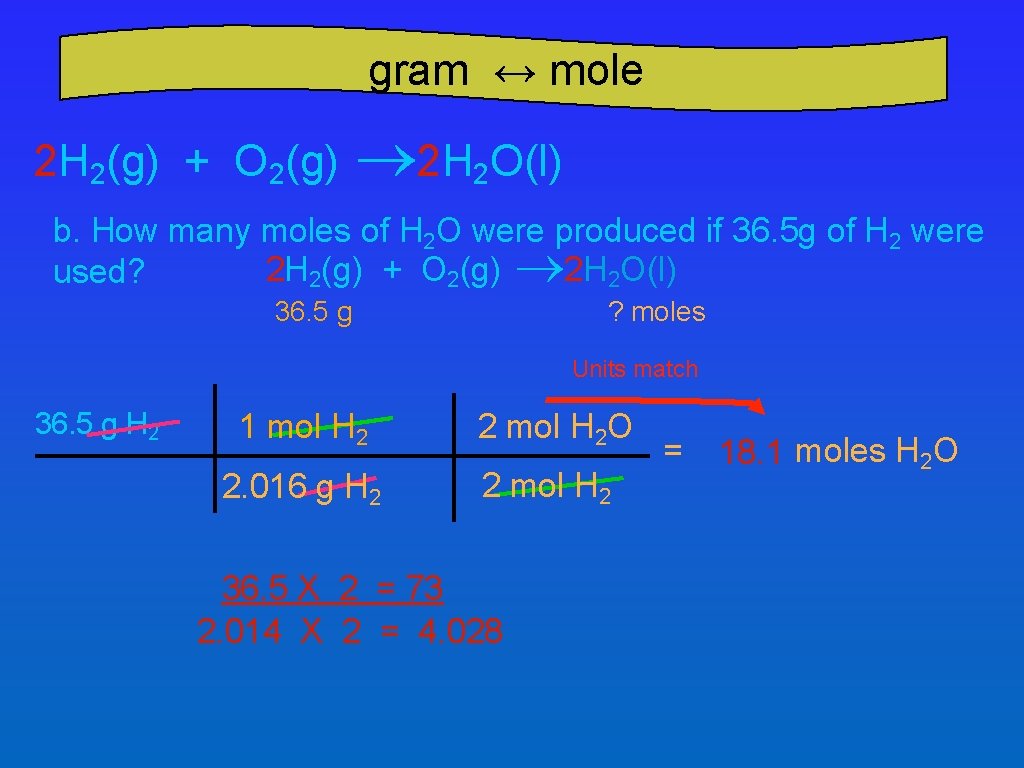
gram ↔ mole 2 H 2(g) + O 2(g) ® 2 H 2 O(l) b. How many moles of H 2 O were produced if 36. 5 g of H 2 were 2 H 2(g) + O 2(g) ® 2 H 2 O(l) used? ? moles 36. 5 g Units match 36. 5 g H 2 1 mol H 2 2 mol H 2 O 2. 016 g H 2 2 mol H 2 36. 5 X 2 = 73 2. 014 X 2 = 4. 028 = 18. 1 moles H 2 O

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? What is the first thing I must do to answer this question? GET THE FORMULA FOR EACH SUBSTANCE! What do I do next? Balance the equation!

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 2 Al + 3 Br 2 ® 2 Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? WHY the equation? To get the mole ratios

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 2 Al + 3 Br 2 ® 2 Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? WHY the equation? To get the mole ratios 3 Br 2 2 Al. Br 3 3 Br 2

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 2 Al + 3 Br 2 ® 2 Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? 2 Al. Br 3 3 Br 2 2 Al. Br 3 needs to be on top because that is what the question is asking for.

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 2 Al + 3 Br 2 ® 2 Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? What else do I need to solve this problem? Convert grams to moles

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 2 Al + 3 Br 2 ® 2 Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? How do I do that? Look up starting substances on periodic table get molar mass.

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? 1 mol Br 2 Now what? 159. 808 g Br 2 Set it up! AND use your gram/mole ratio with mole/mole ratio
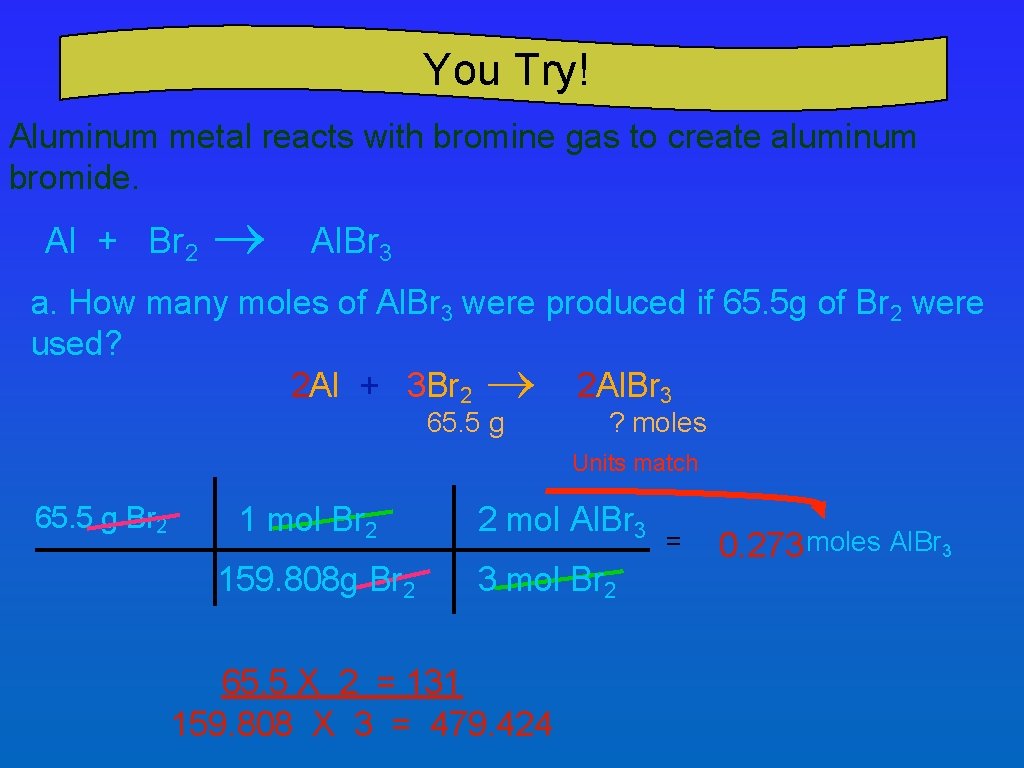
You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 a. How many moles of Al. Br 3 were produced if 65. 5 g of Br 2 were used? 2 Al + 3 Br 2 ® 2 Al. Br 3 65. 5 g ? moles Units match 65. 5 g Br 2 1 mol Br 2 159. 808 g Br 2 2 mol Al. Br 3 3 mol Br 2 65. 5 X 2 = 131 159. 808 X 3 = 479. 424 = 0. 273 moles Al. Br 3

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 b. How many moles of Al were used if 125 g of Al. Br 3 were produced? 2 Al + 3 Br 2 ® 2 Al. Br 3 ? moles 125 g Units match 125 g Al. Br 3 1 mol Al. Br 3 266. 69 g Al. Br 3 2 mol Al. Br 3 125 X 2 = 250 266. 69 X 2 = 533. 38 = 0. 469 moles Al
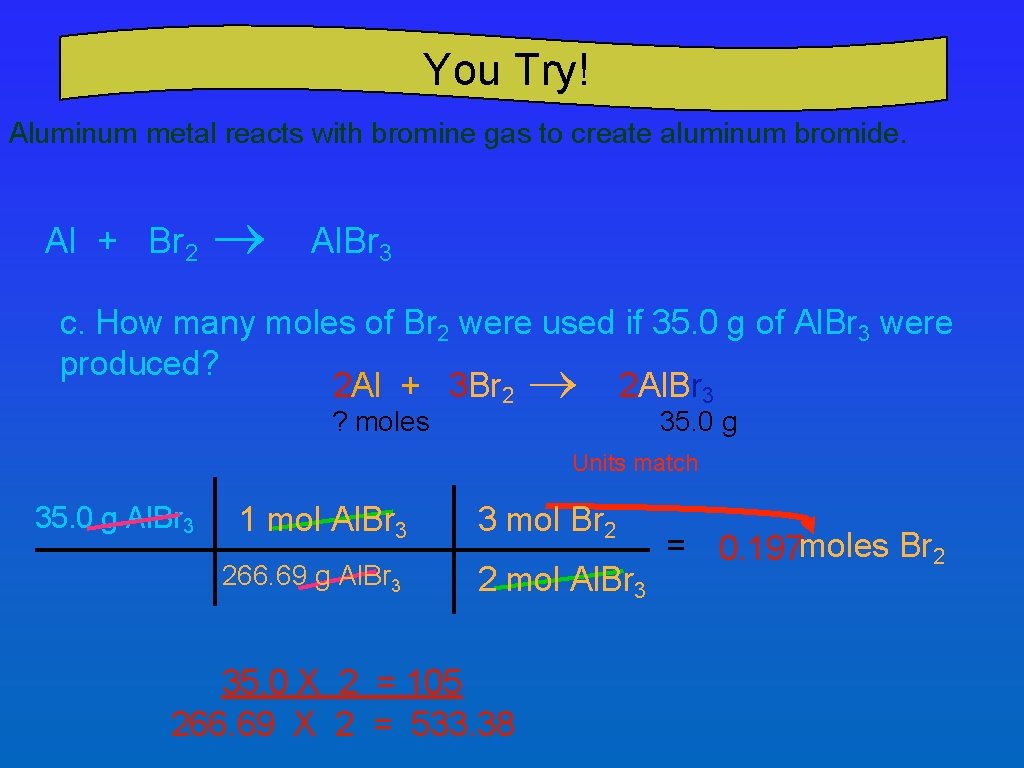
You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 c. How many moles of Br 2 were used if 35. 0 g of Al. Br 3 were produced? 2 Al + 3 Br 2 ® 2 Al. Br 3 ? moles 35. 0 g Units match 35. 0 g Al. Br 3 1 mol Al. Br 3 266. 69 g Al. Br 3 3 mol Br 2 2 mol Al. Br 3 35. 0 X 2 = 105 266. 69 X 2 = 533. 38 = 0. 197 moles Br 2

You Try! Aluminum metal reacts with bromine gas to create aluminum bromide. Al + Br 2 ® Al. Br 3 d. How many moles of Al. Br 3 were produced if 75. 0 g of Al were used? 2 Al + 3 Br ® 2 Al. Br 75. 0 g 2 3 ? moles Units match 75. 0 g Al 1 mol Al 26. 982 g Al 2 mol Al. Br 3 2 mol Al 75. 0 X 2 = 150 26. 982 X 2 = 53. 964 = 2. 78 moles Al. Br 3
- Slides: 34