Stoichiometry Stoichiometry Needs a balanced equation Use the

Stoichiometry

Stoichiometry • Needs a balanced equation • Use the balanced equation to predict ending and / or starting amounts • Coefficients are now mole ratios

In terms of Moles • The coefficients tell us how many moles of each kind. • Mole ratio - conversion ratio that relates the amounts in moles of any two substances in a chemical reaction. • Molar mass - mass, in grams, of one mole of a substance.

Review: Molar Mass A substance’s molar mass (molecular weight) is the mass in grams of one mole of the compound. CO 2 = 44. 01 grams per mole H 2 O = 18. 02 grams per mole Ca(OH)2 = 74. 10 grams per mole

Mole Ratios 2 Al 2 O 3(l) 4 Al(s) + 3 O 2(g) Mole Ratios 2 mol Al 2 O 3 : 4 mol Al 2 O 3 : 3 mol O 2 4 mol Al : 3 mol O 2 Mole Ratio (Fraction) 2 mol Al 2 O 3 4 mol Al 2 O 3 3 mol O 2 4 mol Al 3 mol O 2
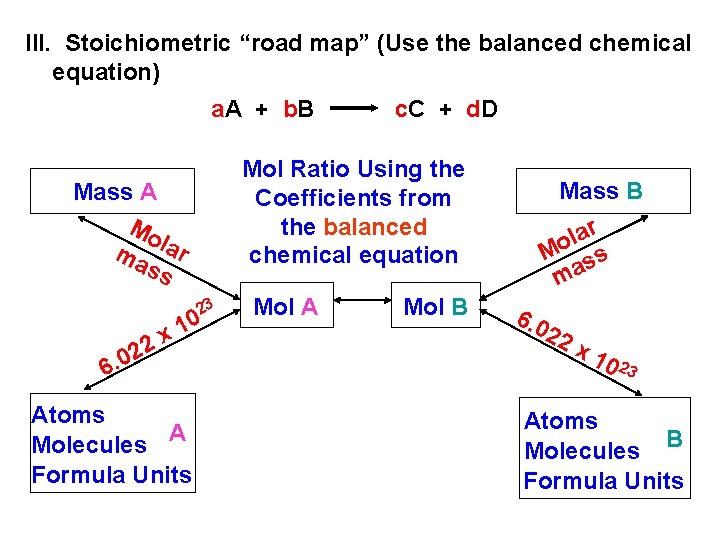
III. Stoichiometric “road map” (Use the balanced chemical equation) a. A + b. B c. C + d. D Mol Ratio Using the Coefficients from the balanced chemical equation Mass A Mo ma lar ss 23 0 1 x 2 2 0. 6 Atoms A Molecules Formula Units Mol A Mol B Mass B r a l o M ss ma 6. 0 22 x 10 23 Atoms B Molecules Formula Units
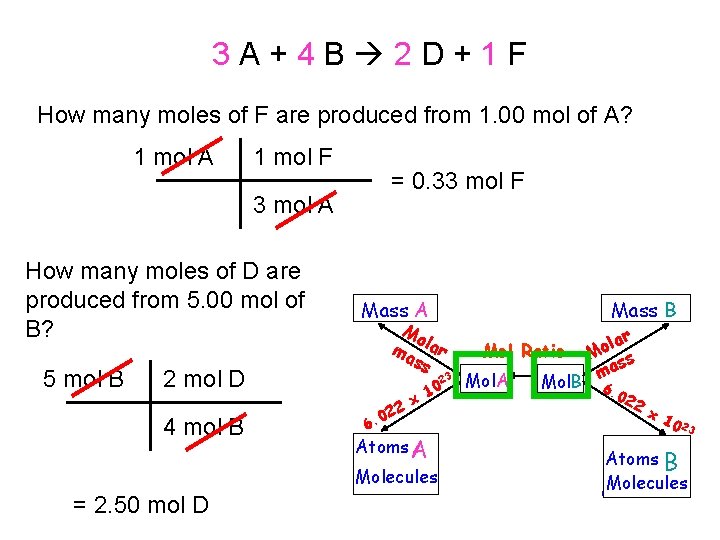
3 A+4 B 2 D+1 F How many moles of F are produced from 1. 00 mol of A? 1 mol A 1 mol F = 0. 33 mol F 3 mol A How many moles of D are produced from 5. 00 mol of B? 5 mol B Mass A Mo ma lar ss 2 mol D 4 mol B 23 22 0 6. x 10 Atoms A Molecules = 2. 50 mol D Mass B ar l o Mol Ratio M s as m Mol. A Mol. B 6. 02 2 x 1 0 23 Atoms B Molecules
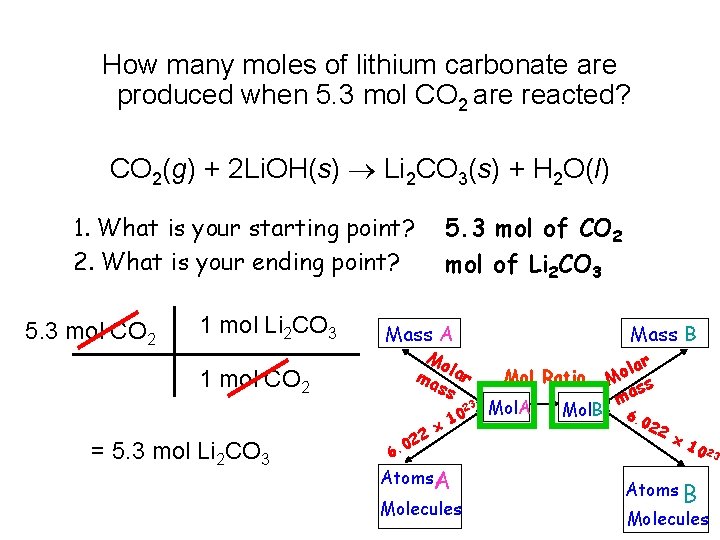
How many moles of lithium carbonate are produced when 5. 3 mol CO 2 are reacted? CO 2(g) + 2 Li. OH(s) Li 2 CO 3(s) + H 2 O(l) 1. What is your starting point? 2. What is your ending point? 5. 3 mol CO 2 1 mol Li 2 CO 3 5. 3 mol of CO 2 mol of Li 2 CO 3 Mass A Mo ma lar ss 1 mol CO 2 23 = 5. 3 mol Li 2 CO 3 0 6. 22 x 10 Atoms A Molecules Mass B Mol Ratio Mol. A Mol. B lar o M ss ma 6. 0 22 x 1 0 23 Atoms B Molecules
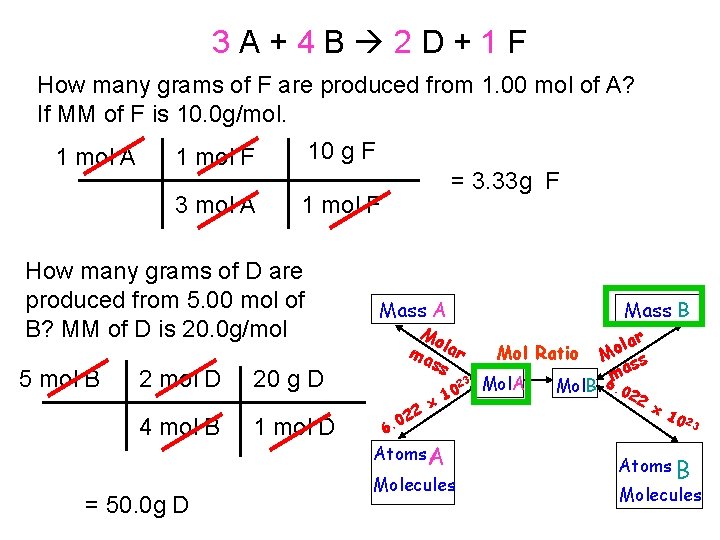
3 A+4 B 2 D+1 F How many grams of F are produced from 1. 00 mol of A? If MM of F is 10. 0 g/mol. 1 mol A 1 mol F 10 g F 3 mol A 1 mol F How many grams of D are produced from 5. 00 mol of B? MM of D is 20. 0 g/mol 5 mol B 2 mol D 4 mol B = 50. 0 g D = 3. 33 g F Mass A Mo ma lar ss 20 g D 1 mol D 23 0 6. 22 x 10 Atoms A Molecules Mass B Mol Ratio Mol. A lar o M ss a 6 m Mol. B. 02 2 x 10 23 Atoms B Molecules
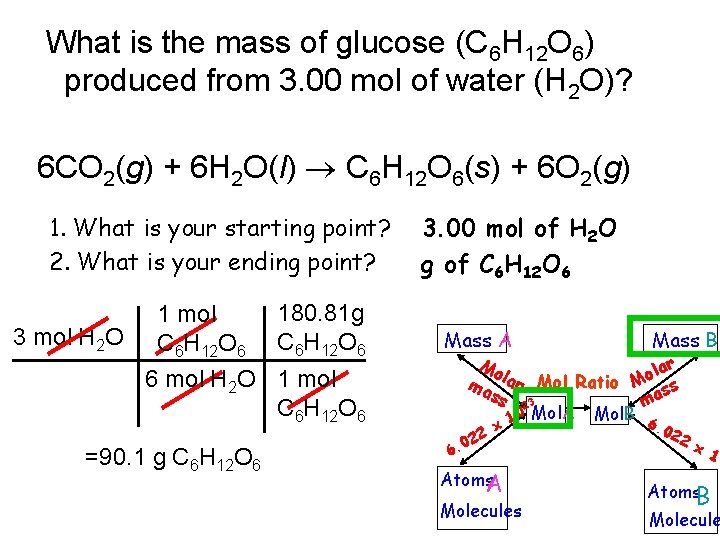
What is the mass of glucose (C 6 H 12 O 6) produced from 3. 00 mol of water (H 2 O)? 6 CO 2(g) + 6 H 2 O(l) C 6 H 12 O 6(s) + 6 O 2(g) 1. What is your starting point? 2. What is your ending point? 180. 81 g 1 mol 3 mol H 2 O C H O C 6 H 12 O 6 6 12 6 6 mol H 2 O 1 mol C 6 H 12 O 6 =90. 1 g C 6 H 12 O 6 3. 00 mol of H 2 O g of C 6 H 12 O 6 Mass A Mass B Mo lar o l a ma r Mol Ratio M ss ss 3 ma 2 10 Mol. A Mol. B 6 22 0 6. x Atoms. A Molecules . 02 2 x 10 Atoms. B Molecule

6 CO 2(g) + 6 H 2 O(l) C 6 H 12 O 6(s) + 6 O 2(g) What is the mass of oxygen (O 2) produced from 2. 50 mol of water (H 2 O)? 1. What is your starting point? 2. What is your ending point? 2. 50 mol of H 2 O g of O 2 2. 5 mol H 2 O 6 mol O 2 32. 0 g O 2 6 mol H 2 O 1 mol O 2 Mass A Mo ma lar ss 23 =80. 0 g O 2 0 6. 22 x 10 Atoms A Molecules Mass B Mol Ratio Mol. A Mol. B lar o M ss ma 6. 0 22 x 1 0 23 Atoms B Molecules
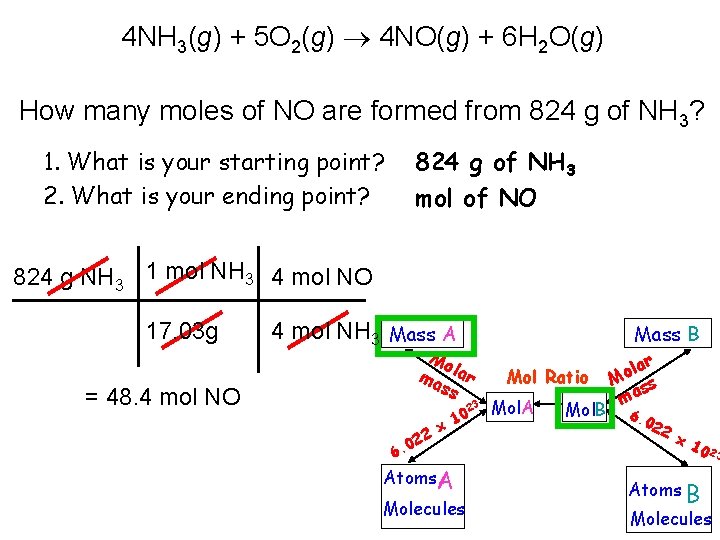
4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) How many moles of NO are formed from 824 g of NH 3? 1. What is your starting point? 2. What is your ending point? 824 g of NH 3 mol of NO 824 g NH 3 1 mol NH 3 4 mol NO 17. 03 g 4 mol NH 3 Mass A Mo ma lar ss = 48. 4 mol NO 23 22 0 6. x 10 Atoms A Molecules Mass B Mol Ratio Mol. A Mol. B lar o M ss ma 6. 0 22 x 1 0 23 Atoms B Molecules
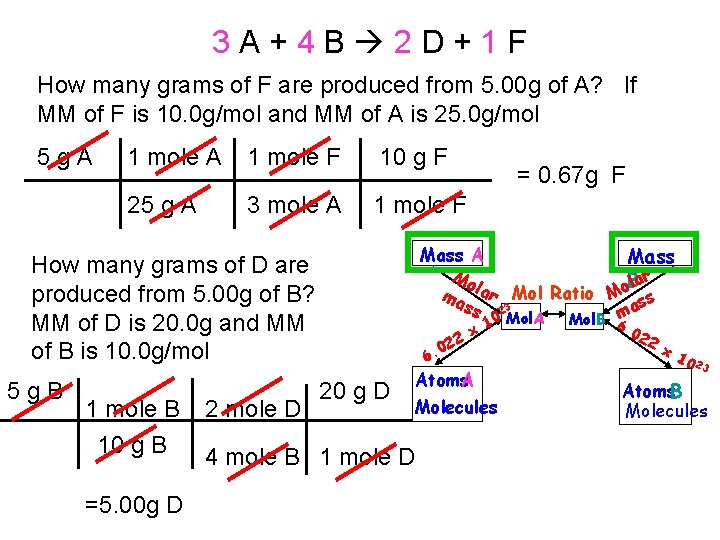
3 A+4 B 2 D+1 F How many grams of F are produced from 5. 00 g of A? If MM of F is 10. 0 g/mol and MM of A is 25. 0 g/mol 5 g. A 1 mole F 10 g F 25 g A 3 mole A 1 mole F Mass A Mass Mo Blar l o a ma r Mol Ratio M ss ss 23 a 0 Mol. A Mol. B m How many grams of D are produced from 5. 00 g of B? MM of D is 20. 0 g and MM of B is 10. 0 g/mol 5 g. B 1 mole B 10 g B =5. 00 g D 2 mole D = 0. 67 g F 22 0 6. 20 g D 4 mole B 1 mole D x 1 Atoms. A Molecules 6. 0 22 x 1 0 23 Atoms. B Molecules
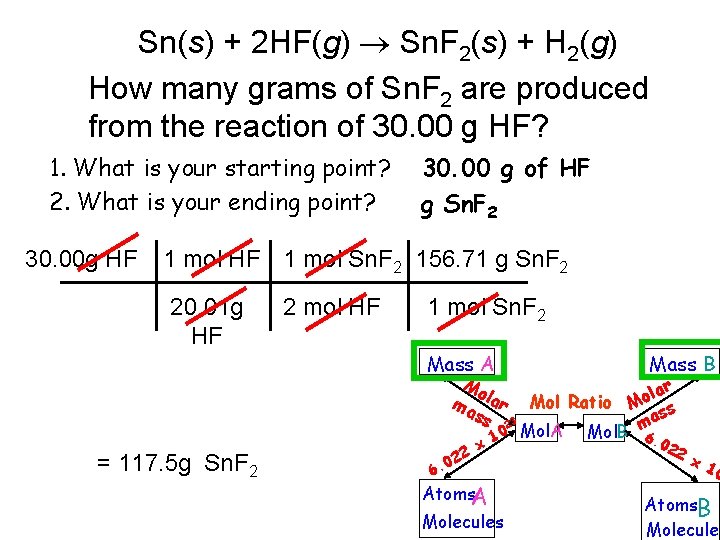
Sn(s) + 2 HF(g) Sn. F 2(s) + H 2(g) How many grams of Sn. F 2 are produced from the reaction of 30. 00 g HF? 1. What is your starting point? 2. What is your ending point? 30. 00 g HF 30. 00 g of HF g Sn. F 2 1 mol HF 1 mol Sn. F 2 156. 71 g Sn. F 2 20. 01 g HF 2 mol HF 1 mol Sn. F 2 Mass A Mo ma lar ss = 117. 5 g Sn. F 2 22 0 6. lar o Mol Ratio M s as 23 m 0 Mol. A Mol. B x 1 Atoms. A Mass B Molecules 6. 0 22 x 1 0 Atoms. B Molecules

Working a Stoichiometry Problem 6. 50 grams of aluminum reacts with an excess of oxygen. How many grams of aluminum oxide are formed? 1. Identify reactants and products and write the balanced equation. 4 Al + 3 O 2 2 Al 2 O 3 a. Every reaction needs a yield sign! b. What are the reactants? c. What are the products? d. What are the balanced coefficients?
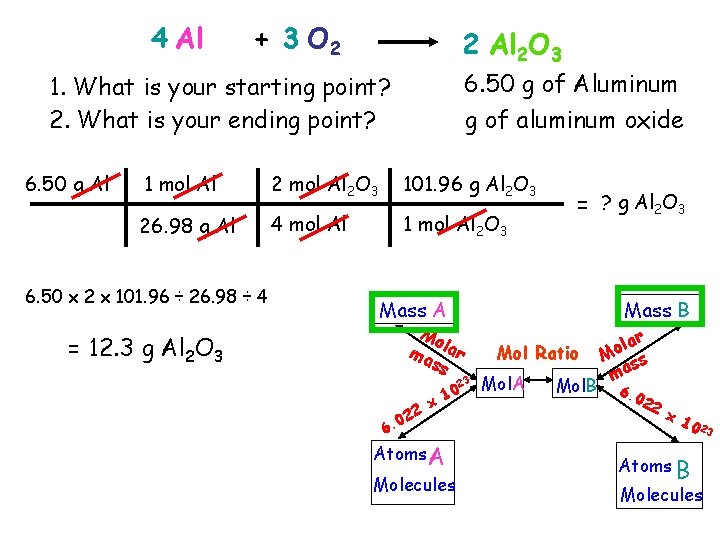
4 Al + 3 O 2 2 Al 2 O 3 6. 50 g of Aluminum g of aluminum oxide 1. What is your starting point? 2. What is your ending point? 6. 50 g Al 1 mol Al 2 O 3 101. 96 g Al 2 O 3 26. 98 g Al 4 mol Al 1 mol Al 2 O 3 6. 50 x 2 x 101. 96 ÷ 26. 98 ÷ 4 = ? g Al 2 O 3 Mass A Mo ma lar ss = 12. 3 g Al 2 O 3 23 0 6. 22 x 10 Atoms A Molecules Mass B Mol Ratio Mol. A Mol. B lar o M ss ma 6. 0 22 x 1 0 23 Atoms B Molecules
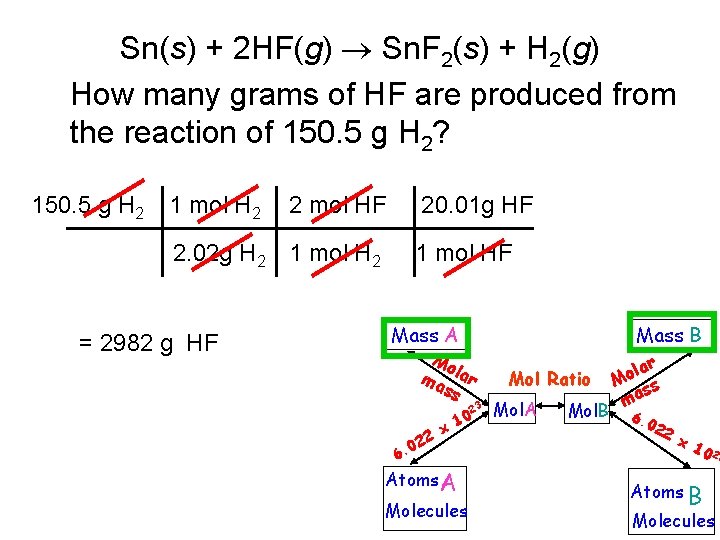
Sn(s) + 2 HF(g) Sn. F 2(s) + H 2(g) How many grams of HF are produced from the reaction of 150. 5 g H 2? 150. 5 g H 2 1 mol H 2 2 mol HF 2. 02 g H 2 1 mol H 2 = 2982 g HF 20. 01 g HF 1 mol HF Mass A Mo ma lar ss 23 0 6. 22 x 10 Atoms A Molecules Mass B Mol Ratio Mol. A Mol. B lar o M ss ma 6. 0 22 x 1 0 23 Atoms B Molecules

II. Limiting Reagent A. Stoichiometric amounts: The proportions indicated in balanced the _____ rxn. B. Most reactions do not have stoichiometric amounts. Generally, one reactant will be _____ before the depleted other. The reactant that is depleted first is known as the limiting reagent (LR) __________. The reactant that is left at the excess reagent (ER) end of the reaction is called the __________. C. Analogy: How to make a cheese sandwich. 2 slices of bread + 1 slice of cheese → 1 cheese sandwich If you have 8 slices of bread and 6 slices of cheese, how many sandwiches can you make? __ (theoretical yield) 4 bread What is the limiting reagent? ______ cheese What is the excess reagent? _______ How much of the excess reagent is left at the end of the 2 slices cheese rxn? _______

D. Theoretical yield: The amount of product in limiting reagent grams that forms if all of the ________ has reacted. (This number is CALCULATED on paper! Units are in grams only!) E. Actual yield: The amount of product (in grams) that is actually made (This number is from the EXPERIMENT). F. Percent yield: The comparison of the actual yield to theoretical yield.
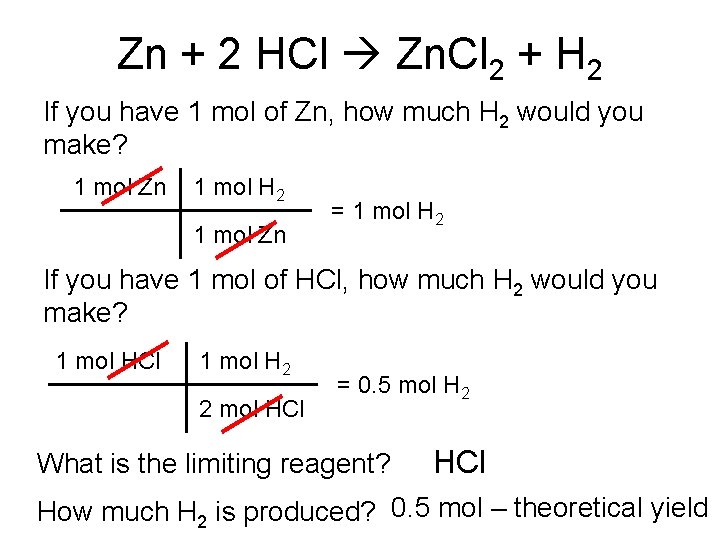
Zn + 2 HCl Zn. Cl 2 + H 2 If you have 1 mol of Zn, how much H 2 would you make? 1 mol Zn 1 mol H 2 1 mol Zn = 1 mol H 2 If you have 1 mol of HCl, how much H 2 would you make? 1 mol HCl 1 mol H 2 2 mol HCl = 0. 5 mol H 2 What is the limiting reagent? HCl How much H 2 is produced? 0. 5 mol – theoretical yield
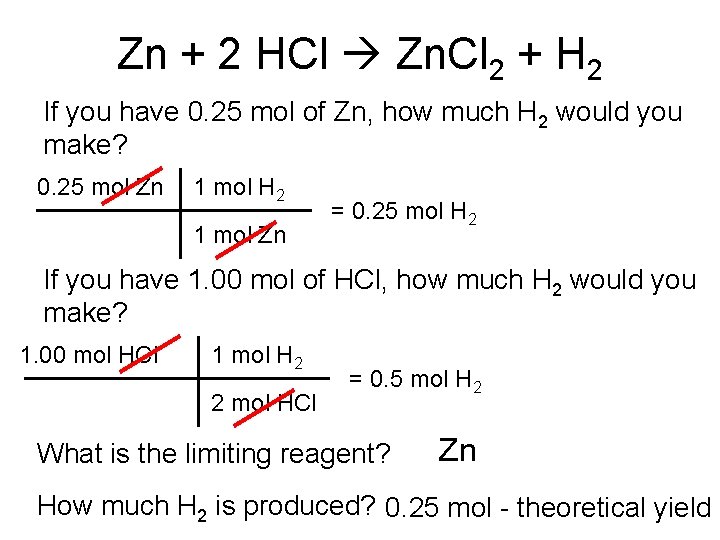
Zn + 2 HCl Zn. Cl 2 + H 2 If you have 0. 25 mol of Zn, how much H 2 would you make? 0. 25 mol Zn 1 mol H 2 1 mol Zn = 0. 25 mol H 2 If you have 1. 00 mol of HCl, how much H 2 would you make? 1. 00 mol HCl 1 mol H 2 2 mol HCl = 0. 5 mol H 2 What is the limiting reagent? Zn How much H 2 is produced? 0. 25 mol - theoretical yield
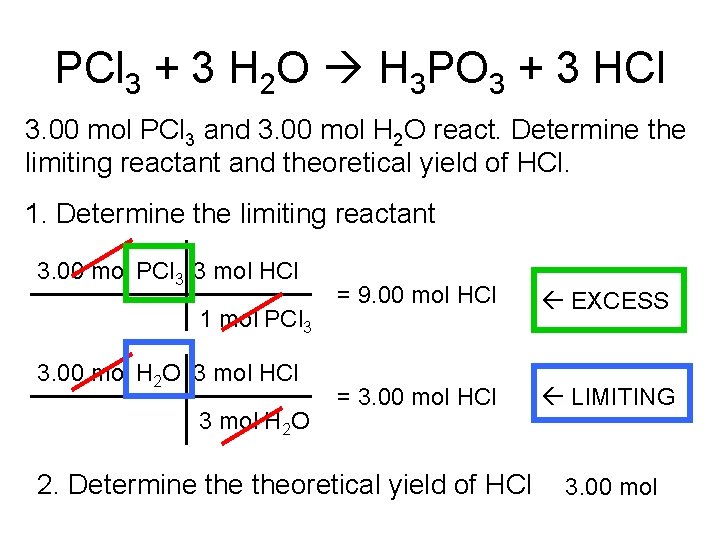
PCl 3 + 3 H 2 O H 3 PO 3 + 3 HCl 3. 00 mol PCl 3 and 3. 00 mol H 2 O react. Determine the limiting reactant and theoretical yield of HCl. 1. Determine the limiting reactant 3. 00 mol PCl 3 3 mol HCl 1 mol PCl 3 3. 00 mol H 2 O 3 mol HCl 3 mol H 2 O = 9. 00 mol HCl EXCESS = 3. 00 mol HCl LIMITING 2. Determine theoretical yield of HCl 3. 00 mol
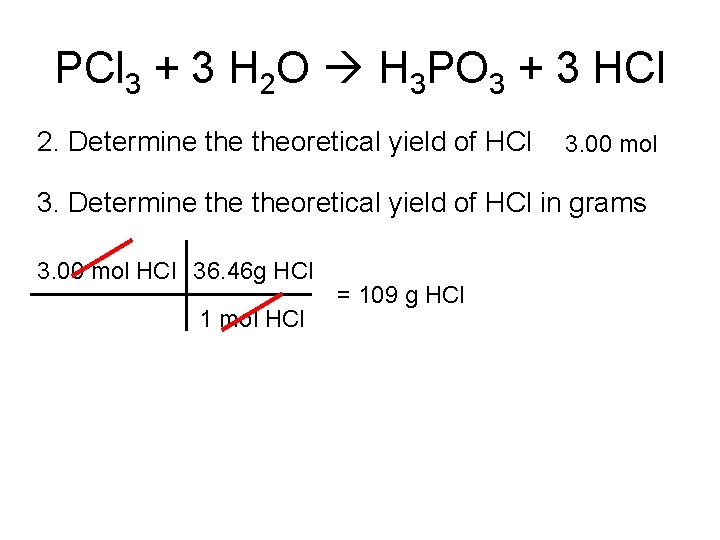
PCl 3 + 3 H 2 O H 3 PO 3 + 3 HCl 2. Determine theoretical yield of HCl 3. 00 mol 3. Determine theoretical yield of HCl in grams 3. 00 mol HCl 36. 46 g HCl 1 mol HCl = 109 g HCl
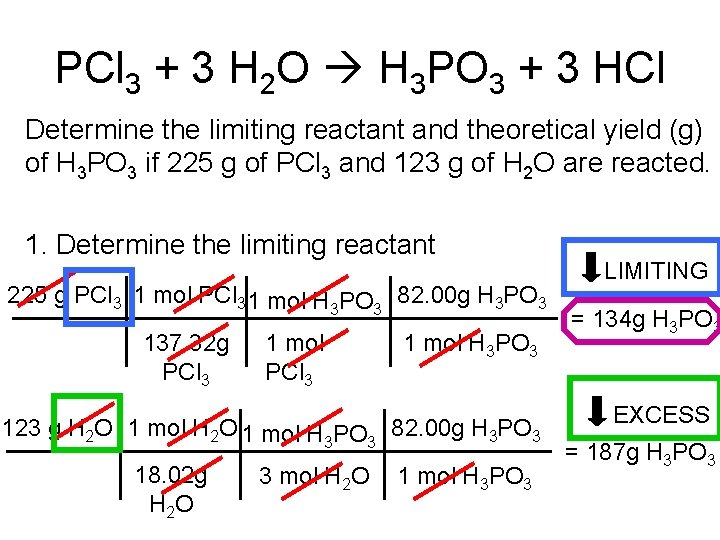
PCl 3 + 3 H 2 O H 3 PO 3 + 3 HCl Determine the limiting reactant and theoretical yield (g) of H 3 PO 3 if 225 g of PCl 3 and 123 g of H 2 O are reacted. 1. Determine the limiting reactant 225 g PCl 3 1 mol H 3 PO 3 82. 00 g H 3 PO 3 137. 32 g PCl 3 1 mol H 3 PO 3 123 g H 2 O 1 mol H 3 PO 3 82. 00 g H 3 PO 3 18. 02 g H 2 O 3 mol H 2 O 1 mol H 3 PO 3 LIMITING = 134 g H 3 PO 3 EXCESS = 187 g H 3 PO 3

PCl 3 + 3 H 2 O H 3 PO 3 + 3 HCl The theoretical yield of this reaction is 134 g H 3 PO 3. However, the actual yield from the experiment is 120 g. Calculate the percent yield. 120 g H 3 PO 4 X 100% 134 g H 3 PO 4 = 89. 6 %
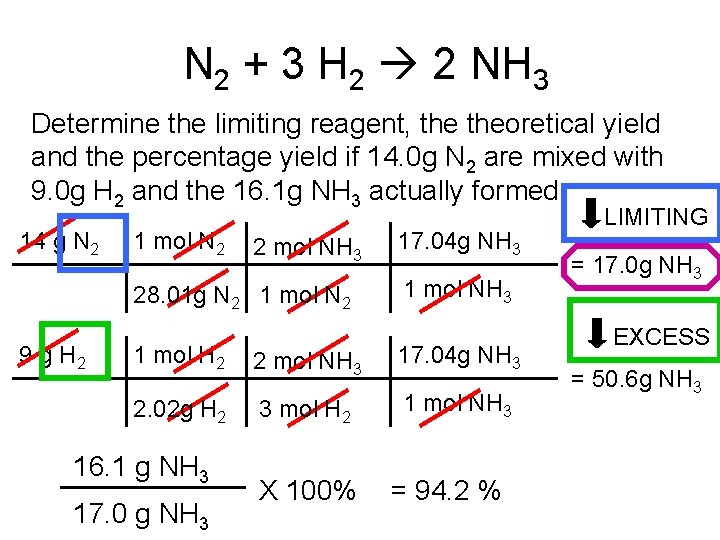
N 2 + 3 H 2 2 NH 3 Determine the limiting reagent, theoretical yield and the percentage yield if 14. 0 g N 2 are mixed with 9. 0 g H 2 and the 16. 1 g NH 3 actually formed. 14 g N 2 1 mol N 2 2 mol NH 3 28. 01 g N 2 1 mol N 2 9 g H 2 17. 04 g NH 3 1 mol H 2 2 mol NH 3 17. 04 g NH 3 2. 02 g H 2 3 mol H 2 1 mol NH 3 16. 1 g NH 3 17. 0 g NH 3 X 100% = 94. 2 % LIMITING = 17. 0 g NH 3 EXCESS = 50. 6 g NH 3
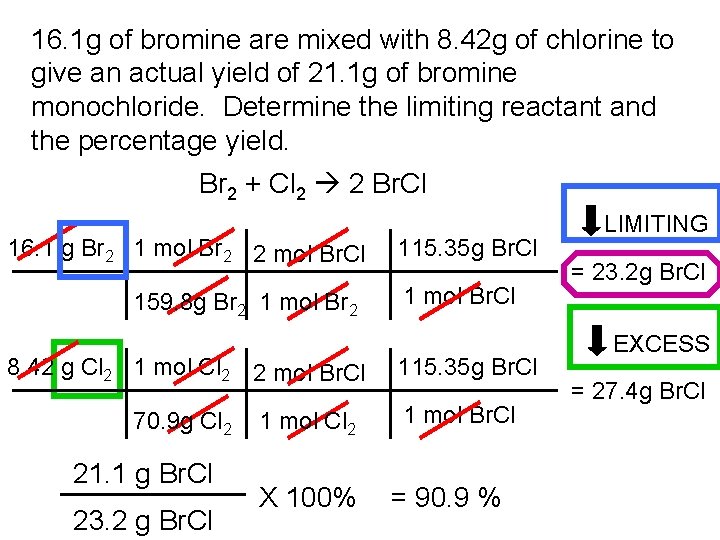
16. 1 g of bromine are mixed with 8. 42 g of chlorine to give an actual yield of 21. 1 g of bromine monochloride. Determine the limiting reactant and the percentage yield. Br 2 + Cl 2 2 Br. Cl 16. 1 g Br 2 1 mol Br 2 2 mol Br. Cl 159. 8 g Br 2 1 mol Br 2 8. 42 g Cl 2 1 mol Cl 2 2 mol Br. Cl 70. 9 g Cl 2 21. 1 g Br. Cl 23. 2 g Br. Cl 1 mol Cl 2 X 100% 115. 35 g Br. Cl 1 mol Br. Cl = 90. 9 % LIMITING = 23. 2 g Br. Cl EXCESS = 27. 4 g Br. Cl
- Slides: 27