Stoichiometry Stoichiometry Basics Stoichiometry is the study of

Stoichiometry

Stoichiometry Basics Stoichiometry is the study of the quantitative relationships that exist in chemical formulas and balanced chemical equations. Stoichiometry allows us to calculate the amounts of substances that are related by a balanced chemical equation.

Do You Remember? 4 Fe + 3 O 2 → 2 Fe 2 O 3 8 Fe + 6 O 2 → 4 Fe 2 O 3 4, 000 Fe + 3, 000 O 2 → 2, 000 Fe 2 O 3 4 mol Fe + 3 mol O 2 → 2 mol Fe 2 O 3 0. 24 mol Fe + 0. 18 mol O 2 → 0. 12 mol Fe 2 O 3

Interpreting Balanced Equations Balance the equation. . . Cu + 2 Ag. NO 3 → Cu(NO 3)2 + 2 Ag This means that 1 mole of copper reacts with 2 moles of silver nitrate to form 1 mole of copper (II) nitrate and 2 moles of silver.

Mole - Mole Problems Balance the equation. . . 6 Li + N 2 → 2 Li 3 N How many moles of Li 3 N can be produced if 3. 00 moles of lithium reacts completely with nitrogen?

Review Example 6 Li + N 2 → 2 Li 3 N How many moles of N 2 are required to react with 7. 2 moles of Lithium?
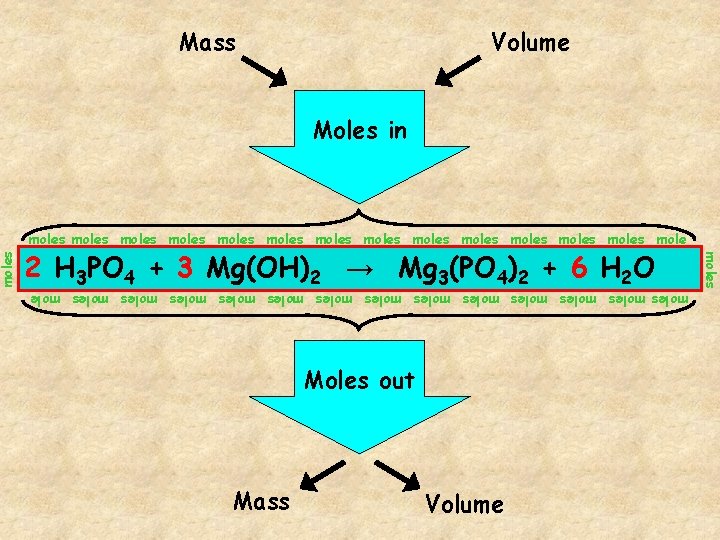
Volume Moles in moles moles moles moles mole 2 The H 3 PO Mg 3(PO )2 + gateway. 6 H 2 O balanced chemical equation is 4 the 4 + 3 Mg(OH) 2 → moles moles moles moles mole Moles out Mass Volume moles Mass
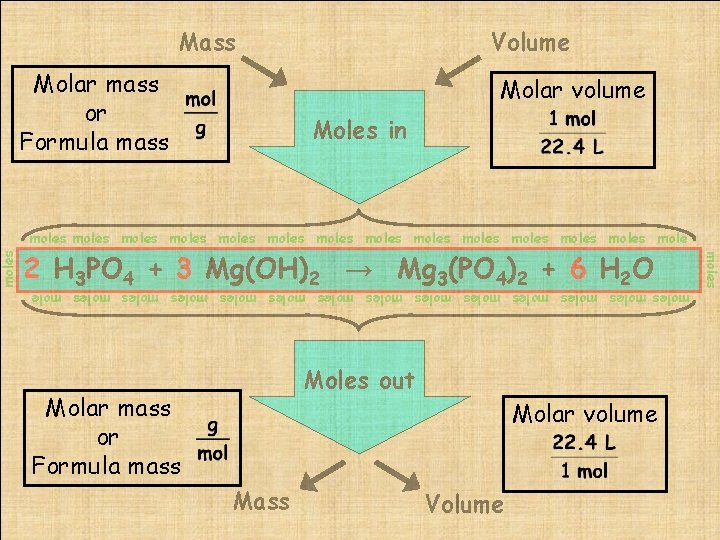
Molar mass or Formula mass Volume Molar volume Moles in moles moles moles moles mole 2 The H 3 PO Mg 3(PO )2 + gateway. 6 H 2 O balanced chemical equation is 4 the 4 + 3 Mg(OH) 2 → moles moles moles moles mole Moles out Molar mass or Formula mass Molar volume Mass Volume moles Mass

Mass - Mass Conversions How many grams of Sn. F 4 can be formed from 12. 8 g of HF? Sn + 4 HF → Sn. F 4 + 2 H 2

Mass - Volume Problems How many liters of O 2(g) are needed at STP to burn 60. 0 g of magnesium? 2 Mg + O 2 → 2 Mg. O

Volume - Volume Problems How many liters of H 2 O(g) can be formed from 48. 9 L of CH 4(g) at STP? CH 4 + 2 O 2 → CO 2 + 2 H 2 O Notice that numerically the first ratio and the third ratio cancel out. For vol/vol problems the mole ratio is sufficient.
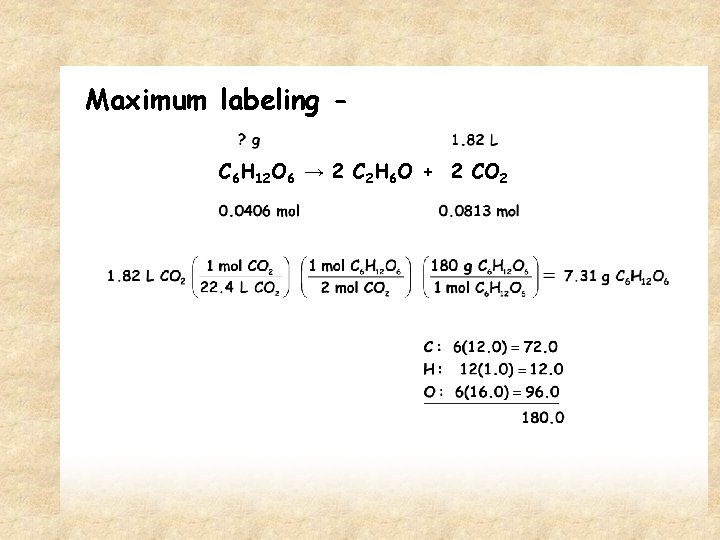
Maximum labeling C 6 H 12 O 6 → 2 C 2 H 6 O + 2 CO 2

Minimum labeling C 6 H 12 O 6 → 2 C 2 H 6 O + 2 CO 2

Limiting Reactants Under what conditions do we need to be concerned about limiting reactants? (and, … just what is a limiting reactant? )
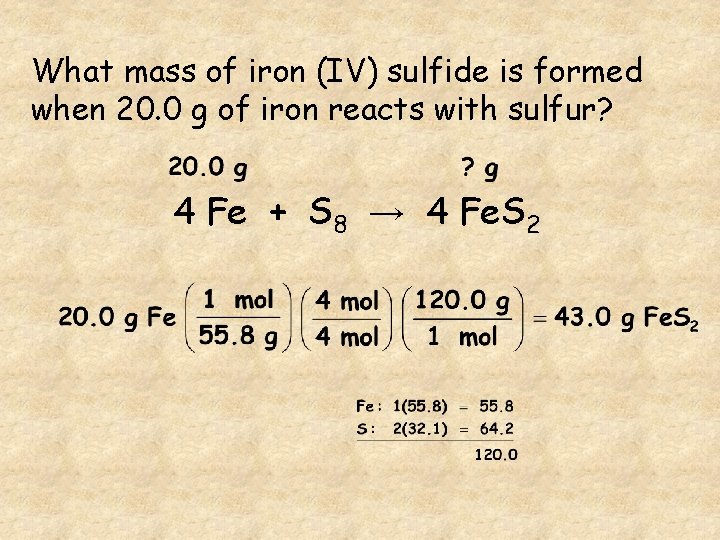
What mass of iron (IV) sulfide is formed when 20. 0 g of iron reacts with sulfur? 4 Fe + S 8 → 4 Fe. S 2
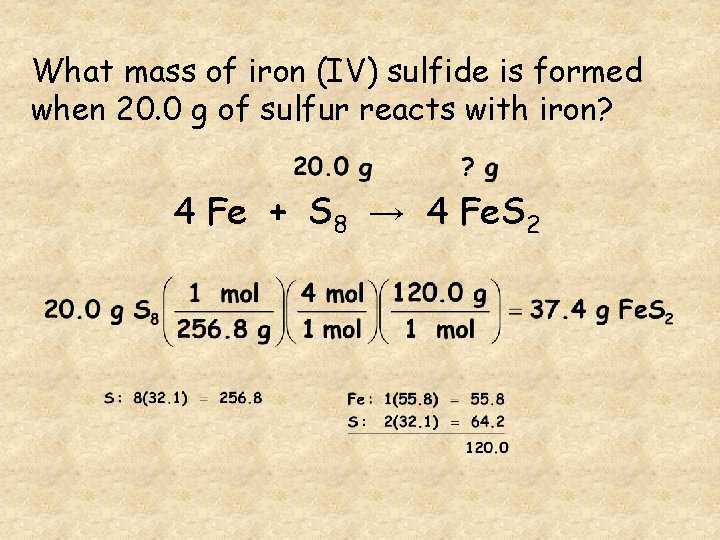
What mass of iron (IV) sulfide is formed when 20. 0 g of sulfur reacts with iron? 4 Fe + S 8 → 4 Fe. S 2

Summary 4 Fe + S 8 → 4 Fe. S 2

What happens at this point? 4 Fe + S 8 → 4 Fe. S 2 The reaction STOPS!

Since sulfur causes the reaction to stop we call sulfur the limiting reactant in this reaction. Since there is some iron left over we say it is in excess.
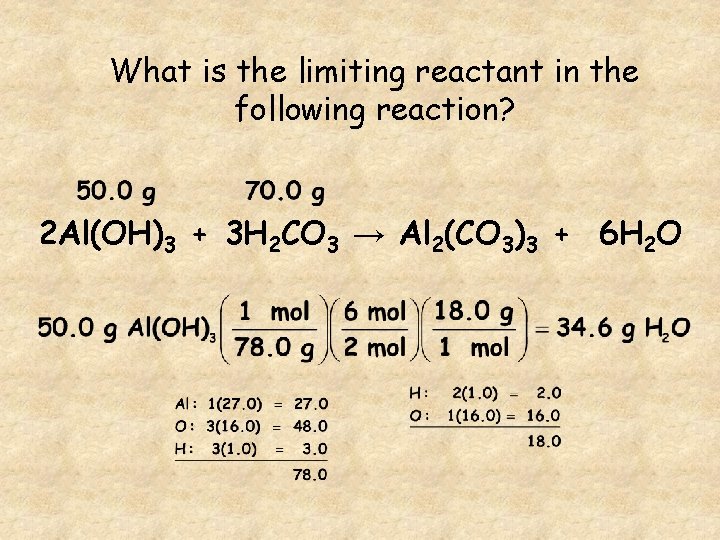
What is the limiting reactant in the following reaction? 2 Al(OH)3 + 3 H 2 CO 3 → Al 2(CO 3)3 + 6 H 2 O
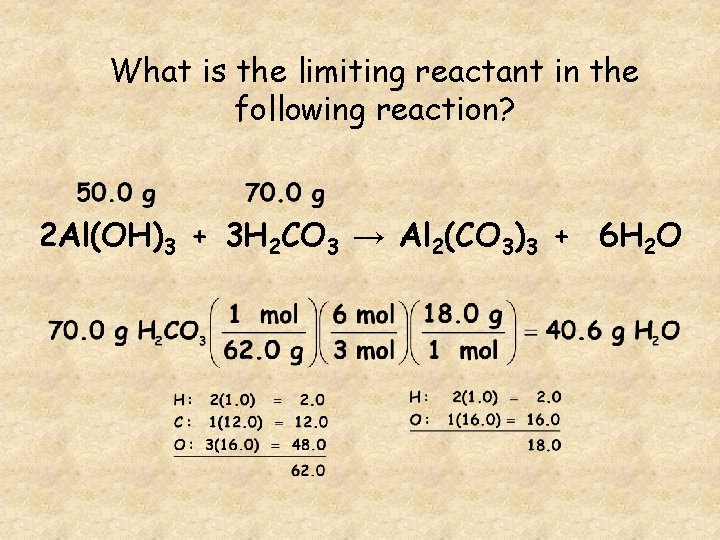
What is the limiting reactant in the following reaction? 2 Al(OH)3 + 3 H 2 CO 3 → Al 2(CO 3)3 + 6 H 2 O
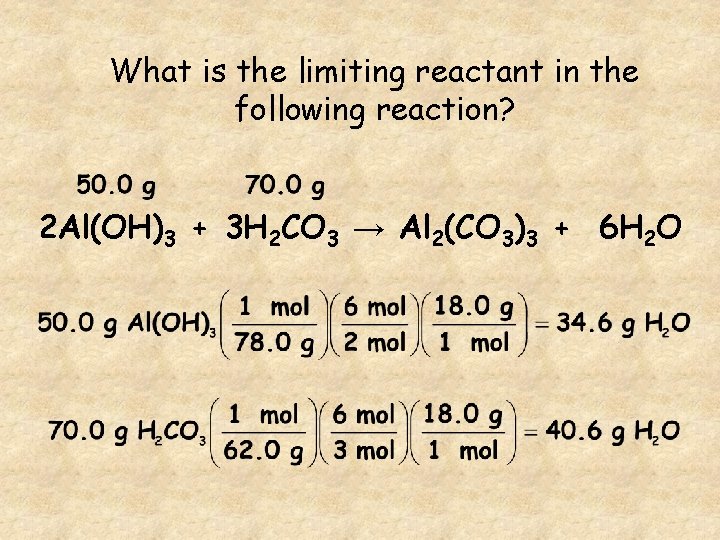
What is the limiting reactant in the following reaction? 2 Al(OH)3 + 3 H 2 CO 3 → Al 2(CO 3)3 + 6 H 2 O
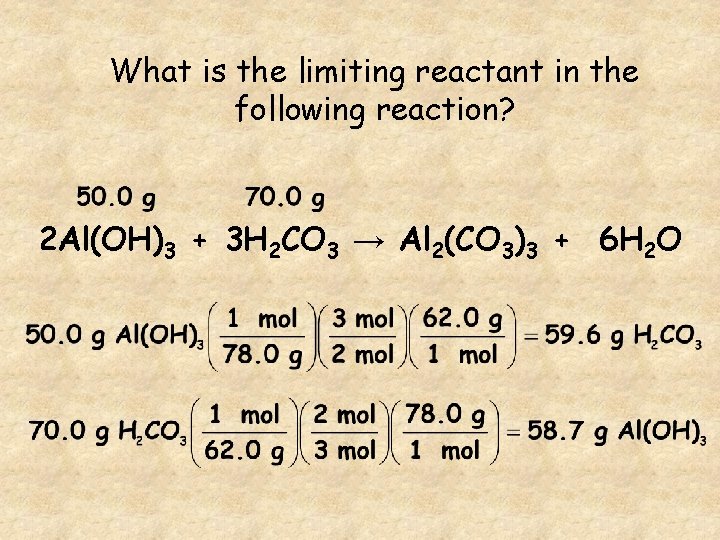
What is the limiting reactant in the following reaction? 2 Al(OH)3 + 3 H 2 CO 3 → Al 2(CO 3)3 + 6 H 2 O
- Slides: 23