STOICHIOMETRY Reaction Stoichiometry the numerical relationships between chemical

STOICHIOMETRY
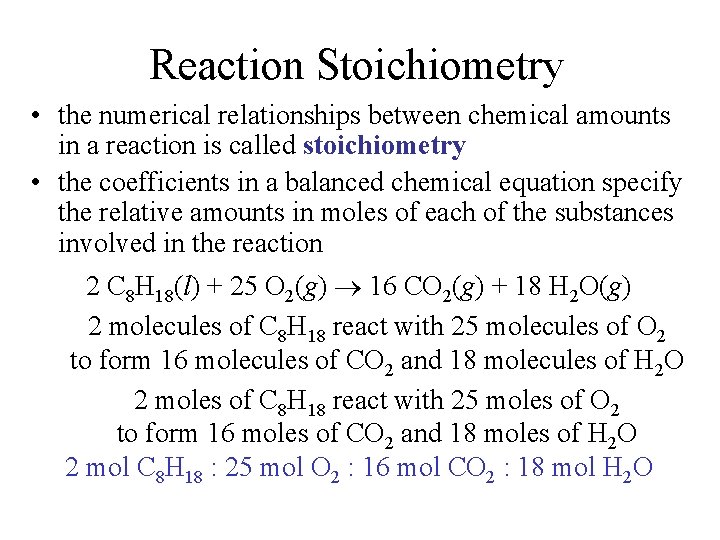
Reaction Stoichiometry • the numerical relationships between chemical amounts in a reaction is called stoichiometry • the coefficients in a balanced chemical equation specify the relative amounts in moles of each of the substances involved in the reaction 2 C 8 H 18(l) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(g) 2 molecules of C 8 H 18 react with 25 molecules of O 2 to form 16 molecules of CO 2 and 18 molecules of H 2 O 2 moles of C 8 H 18 react with 25 moles of O 2 to form 16 moles of CO 2 and 18 moles of H 2 O 2 mol C 8 H 18 : 25 mol O 2 : 16 mol CO 2 : 18 mol H 2 O

Predicting Amounts from Stoichiometry • the amounts of any other substance in a chemical reaction can be determined from the amount of just one substance • How much CO 2 can be made from 22. 0 moles of C 8 H 18 in the combustion of C 8 H 18? 2 C 8 H 18(l) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(g) 2 moles C 8 H 18 : 16 moles CO 2

Stoichiometry – Lecture Questions 1. Finely divided sulfur ignites spontaneously in fluorine to produce sulfur hexafluoride according to the following unbalanced equation: S(s) + F 2(g) SF 6(g) A. How many grams of SF 6(g) can be produced from 5. 00 g of sulfur? B. How many grams of fluorine are required to react with the 5. 00 g of sulfur? 2. Deuterated ammonia, ND 3(g), can be prepared by reacting lithium nitride with heavy water, D 2 O(l), according to the following equation: Li 3 N(s) + D 2 O(l) Li. OD(s) + ND 3(g) A. How many milligrams of heavy water are required to produce 7. 15 mg of ND 3(g)? Take the atomic mass of deuterium to be 2. 014 amu. B. Given that the density of heavy water is 1. 106 g/m. L at room temperature, how many milliliters of heavy water are required?

Workshop ST#1 on Balancing Chemical Equations & Stoichiometry 1. Balance the following chemical equations shown below: A. B. C. D. KCl. O 3(s) KCl(s) + O 2(g) NH 3(g) + O 2(g) N 2(g) + H 2 O(g) Fe(s) + H 2 O(g) Fe 3 O 4(s) + H 2(g) H 2 S(g) + SO 2(g) H 2 O(l) + S(s) 2. A frequently used method for preparing oxygen in the laboratory is by thermal decomposition of potassium chlorate according to the following unbalanced chemical equation: KCl. O 3(s) KCl(s) + O 2(g) How many grams of O 2(g) can be prepared from 30. 6 g of KCl. O 3(s)? 3. Consider the combustion of propane, C 3 H 8. A. How many grams of O 2 are required to burn 75. 0 g of propane? B. How many grams of CO 2 are produced?

Limiting Reagent When chemicals are mixed together to undergo a reaction, they are often mixed in stoichiometric quantities, that is, in exactly the correct amounts so that all reactants “run out” at the same time. However, if one or more reactant(s) is used in excess, then the scarce reagent is called the limiting reagent (or reactant). In any stoichiometric problem, it is ESSENTIAL to determine which reactant is limiting in order to calculate correctly the amounts of products that will be formed. A mixture is prepared from 25. 0 g of aluminum and 85. 0 g of Fe 2 O 3. The reaction that occurs is described by the following equation: Fe 2 O 3(s) + Al(s) Al 2 O 3(s) + Fe(l) How much iron is produced in the reaction?

Theoretical Yield • The theoretical yield is the maximum amount of product that can be made. – In other words, it is the amount of product possible as calculated through the stoichiometry problem. • This is different from the actual yield, which is the amount one actually produces and measures.

Workshop ST#2 on Limiting Reagent 1. Calcium sulfide can be made by heating calcium sulfate with charcoal at high temperature according to the following unbalanced chemical equation: Ca. SO 4(s) + C(s) Ca. S(s) + CO(g) How many grams of Ca. S(s) can be prepared from 100. 0 g each of Ca. SO 4(s) and C(s)? How many grams of unreacted reactant remain at the end of this reaction? 2. If 21. 4 g of solid zinc are treated with 3. 13 L 0. 200 M HCl, how many grams of hydrogen gas will theoretically be formed? How much of which reactant will be left unreacted? The products of this reaction are hydrogen gas and zinc chloride.

Percentage Yield - lecture questions Percent yield = 1. Liquid tin(IV) chloride can be made by heating tin in an atmosphere of dry chlorine. If the percentage yield of this process is 64. 3%, then how many grams of tin are required to produce 0. 106 g of the product? 2. Aluminum burns in bromine, producing aluminum bromide. When 6. 0 g of aluminum was reacted with an excess of bromine, 50. 3 g of aluminum bromide was isolated. Calculate theoretical and percent yield of this reaction.

Workshop ST#3 on Percentage Yield 1. A 0. 473 -g sample of phosphorus is reacted with an excess of chlorine, and 2. 12 g of phosphorus pentachloride is collected. What is the percentage yield of the product? 2. A century ago, sodium bicarbonate was prepared from sodium sulfate by a three-step process: Na 2 SO 4(s) + 4 C(s) Na 2 S(s) + 4 CO(g) Na 2 S(s) + Ca. CO 3(s) Ca. S(s) + Na 2 CO 3(s) + H 2 O(l) + CO 2(g) 2 Na. HCO 3(s) How many kilograms of sodium bicarbonate could be formed from one kilogram of sodium sulfate, assuming an 82% yield in each step?

INTRODUCTION TO GRAVIMETRIC ANALYSIS (advance stoichiometry)

INTRODUCTION to GRAVIMETRIC ANALYSIS MANY TIMES IN LAB, WE DO NOT HAVE ALL THE INFORMATION AND/OR DATA NECESSARY TO COMPLETE STOICHIOMETRIC PROBLEMS WITH BALANCED CHEMICAL EQUATIONS. IN THIS CASE, WE USE A METHOD REFERED TO AS GRAVIMETRIC ANALYSIS. Step 1: Since a balanced equation is sometimes not possible; in gravimetric analysis the first step is to draw out a plan or draw a picture to represent the physical process in the lab. ORE compound solution Compound or element Step 2: Find correlations and relationships (maybe common element) Step 3: Stoichiometry

GRAVIMETRIC ANALYSIS – Lecture problems 1) Under a certain set of laboratory conditions, 2. 1 g of sodium reacts with water to form 1. 14 L of hydrogen gas. When 3. 4 g of another alkali metal is reacted with water under the same conditions, 497 m. L of hydrogen gas is evolved. Identify the alkali metal. In this problem, we will try to set up some relationships: 2 Na + 2 H 2 O → H 2 + 2 Na. OH 2. 1 g 1. 14 L 23. 0 g 2. 1 g Na 1 mol Na 1 mol H 2 Z = 1. 14 L H 2 23. 0 g Na 2 mol Na 0. 04565 Z = 1. 14 Z = 24. 97

GRAVIMETRIC ANALYSIS – Lecture problems 1) Under a certain set of laboratory conditions, 2. 1 g of sodium reacts with water to form 1. 14 L of hydrogen gas. When 3. 4 g of another alkali metal is reacted with water under the same conditions, 497 m. L of hydrogen gas is evolved. Identify the alkali metal. NOW let’s use that relationship on our unknown: 2 X + 2 H 2 O → H 2 + 2 XOH 3. 4 g ? ? 3. 4 g X 0. 497 L 1 mol X ? g. X 1 mol H 2 2 mol X Z PV=n. RT But Z = 24. 97 V=n. RT/P 42. 4514 (1/ ? ) ? = 0. 497 ? = 85. 42 g/mol V=n Z = 0. 497 L H 2 Rb
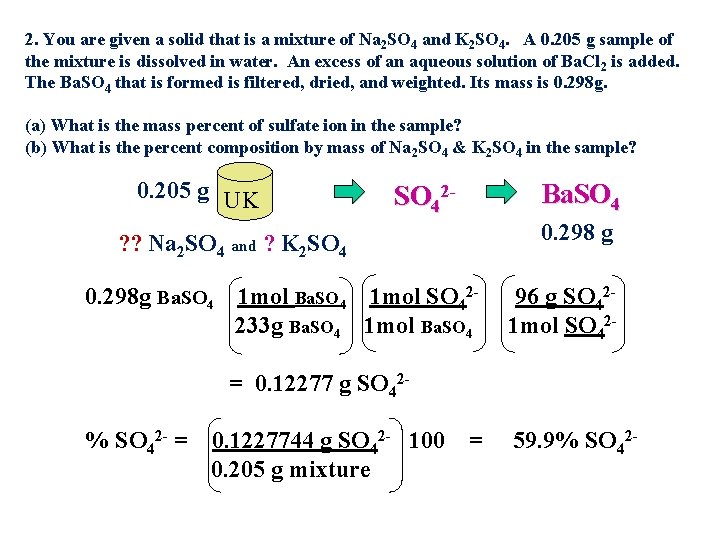
2. You are given a solid that is a mixture of Na 2 SO 4 and K 2 SO 4. A 0. 205 g sample of the mixture is dissolved in water. An excess of an aqueous solution of Ba. Cl 2 is added. The Ba. SO 4 that is formed is filtered, dried, and weighted. Its mass is 0. 298 g. (a) What is the mass percent of sulfate ion in the sample? (b) What is the percent composition by mass of Na 2 SO 4 & K 2 SO 4 in the sample? 0. 205 g UK Ba. SO 42 - 0. 298 g ? ? Na 2 SO 4 and ? K 2 SO 4 0. 298 g Ba. SO 4 1 mol SO 42233 g Ba. SO 4 1 mol Ba. SO 4 96 g SO 421 mol SO 42 - = 0. 12277 g SO 42% SO 42 - = 0. 1227744 g SO 42 - 100 0. 205 g mixture = 59. 9% SO 42 -

2. You are given a solid that is a mixture of Na 2 SO 4 and K 2 SO 4. A 0. 205 g sample of the mixture is dissolved in water. An excess of an aqueous solution of Ba. Cl 2 is added. The Ba. SO 4 that is formed is filtered, dried, and weighted. Its mass is 0. 298 g. (a) What is the mass percent of sulfate ion in the sample? (b) What is the percent composition by mass of Na 2 SO 4 & K 2 SO 4 in the sample? 0. 205 g UK ? ? Na 2 SO 4 and ? K 2 SO 42 - Ba. SO 4 0. 298 g

2. You are given a solid that is a mixture of Na 2 SO 4 and K 2 SO 4. A 0. 205 g sample of the mixture is dissolved in water. An excess of an aqueous solution of Ba. Cl 2 is added. The Ba. SO 4 that is formed is filtered, dried, and weighted. Its mass is 0. 298 g. (a) What is the mass percent of sulfate ion in the sample? (b) What is the percent composition by mass of Na 2 SO 4 & K 2 SO 4 in the sample? 0. 298 g Ba. SO 4 1 mol SO 42233 g Ba. SO 4 1 mol Ba. SO 4 = 1. 2789 x 10 -3 mol SO 42 UK ? ? Na 2 SO 4 and ? K 2 SO 4 Relationships: 0. 205 g (1) 1 mol Na 2 SO 4 = 1 mol SO 42(2) ? mol Na 2 SO 4 + ? mol K 2 SO 4 = 1. 2789 x 10 -3 mol SO 42% K 2 SO 4 = (3) n = m/MM so replace n with m/MM (0. 127 g/0. 205 g)100 (4) m. N + m. K = 1. 2789 x 10 -3 mol SO 42= 61. 95% 142 g/mol 174 g/mol m. N = 0. 205 g - m. K % Na 2 SO 4 = (0. 0778 g/0. 205 g)100 = 37. 9% = 1. 2789 x 10 -3 mol SO 2 - (5) m. N + m. K = 0. 205 g mixture so 0. 205 – m. K + m. K 4 142 174 Solving for m. K = 4. 0709/32 = 0. 127 g therefore m. N = 0. 0778 g

EXTRA STOICHIOMETRY QUESTIONS CHAT 1) A frequently used method for preparing oxygen in the laboratory is by thermal decomposition of potassium chlorate according to the following unbalanced chemical equation: KCl. O 3(s) KCl(s) + O 2(g) What mass of O 2(g) can be produced from 30. 6 g of KCl. O 3(s)? 2) Calcium sulfide can be made by heating calcium sulfate with charcoal at high temperature according to the following unbalanced chemical equation: Ca. SO 4(s) + C(s) Ca. S(s) + CO(g) a) How many grams of Ca. S(s) can be prepared from 100. 0 g each of Ca. SO 4(s) and C(s)? b) How many grams of unreacted reactant remain at the end of this reaction? 3) A 0. 473 -g sample of phosphorus is reacted with an excess of chlorine, and 2. 12 g of phosphorus pentachloride is collected. What is the percentage yield of the product? 4) A century ago, sodium bicarbonate was prepared from sodium sulfate by a three-step process: Na 2 SO 4(s) + 4 C(s) Na 2 S(s) + 4 CO(g) Na 2 S(s) + Ca. CO 3(s) Ca. S(s) + Na 2 CO 3(s) + H 2 O(l) + CO 2(g) 2 Na. HCO 3(s) How many kilograms of sodium bicarbonate could be formed from one kilogram of sodium sulfate, assuming an 82% yield in each step? 5) If 21. 4 g of solid zinc are treated with 3. 13 L 0. 200 M HCl, (a) how many grams of hydrogen gas will theoretically be formed? (b) How much of which reactant will be left unreacted? The products of this reaction are hydrogen gas and zinc chloride.
- Slides: 18