Stoichiometry of Combustion and Boiler Efficiency Calculations By

Stoichiometry of Combustion and Boiler Efficiency Calculations By Dr. S. A. Channiwala Professor, Mechanical Engineering Department S. V. National Institute of Technology Ichchhanath, Surat, Gujarat

CONTENTS INTRODUCTION STOICHIOMETRY OF COMBUSTION ESTIMATION OF BOILER EFFICIENCY BY DIRECT METHOD ESTIMATION OF BOILER EFFICIENCY BY INDIRECT METHOD OR LOSS METHOD AS PER BS-2885 BASIS OF THE METHOD ESTIMATION OF VARIOUS LOSSES ESTIMATION OF BOILER EFFICIENCY NUMERICAL EXAMPLES PACKAGE BOILER UKAI THERMAL POWER STATION PULVERISED FUEL FIRED BOILER GIPCL THERMAL POWER STATION CIRCULATING FLUIDISED BOILER WITH LIMESTONE ADDITION CONCLUSIONS

INTRODUCTION FUEL - A combustion liberates heat with minimum emissions. CALORIFIC VALUE : Energy content of fuel per unit mass or unit volume of fuel. UNITS : k. J/kg or kcals/kg for Solid & Liquids : k. J/nm 3 or kcals/nm 3 for Gaseous Fuels Gross calorific value or Higher Heating Value : Amount of heat energy liberated per unit mass or unit volume with H 2 O in its liquid state under standard conditions [25 C, 1 atm. pressure] Lower Calorific Value/Lower heating value : Amount of heat energy liberated per unit mass or unit volume with H 2 O in its vapour form under standard condition [25 C, temp. 1 atm. pressure]

FOR CONSTANT VOLUME COMBUSTION : Qvh = GCVv=c = LCVv=c+ufg x mw ufg = hfg –p. Vfg at 25 C = 2305 k. J/kg mw = MC+9 H, kg/kg of fuel MC = Moisture, kg/kg of fuel H = Hydrogen, kg/kg of fuel FOR CONSTANT PRESSURE COMBUSTION : Qph = Qpl + hfg. mw GCVp=c = LCVp=c +hfg. mw hfg = 2442. 3 k. J/kg at 25 C mw = MC+9 H RELATION BETWEEN CONSTANT PRESSURE & CONSTANT VOLUME COMBUSTION GCVp=c n = GCVv=c + n. Ro. T = np - nr = No. of Moles of Gaseous Product - No. of moles of Gaseous Reactants Ro = 8. 314 k. J/kg mole- K T = Temp. in K=298 K

STOICHIOMETRY OF COMBUSTION DATA : Coal : Ultimate analysis : C = 66. 0 % H = 4. 1 % S = 1. 7 % O =7. 2 % N= 1. 3 % MC = 12. 0 % Ash = 7. 7 % Gross CV=27 MJ/kg. Proximate : Analysis FC = 50. 19 % VM=30. 11 % Ash = 7. 7 % MC = 12. 0 % Excess Air= 40 % DETERMINE : (a) Theoretical Air Required (b) Actual Air (c) Composition of Flue Gas (d) Density of Flue Gas at 0 C & 25 C & at 160 C (e) Adiabatic Flame Temperature SOLUTION : 1. Combustion of Carbon :

2. Combustion Hydrogen : 3. Combustion of Sulphur : (a) Theoritical O 2 Required : Theoretical air required for complete combustion is : 8. 839 kg/kg of fuel (b) Actual Air [40% excess] : O 2 N 2 2. 8462 kg 9. 5284 kg Air/Fuel = 12. 3746 : 1 Air 12. 3746 kg
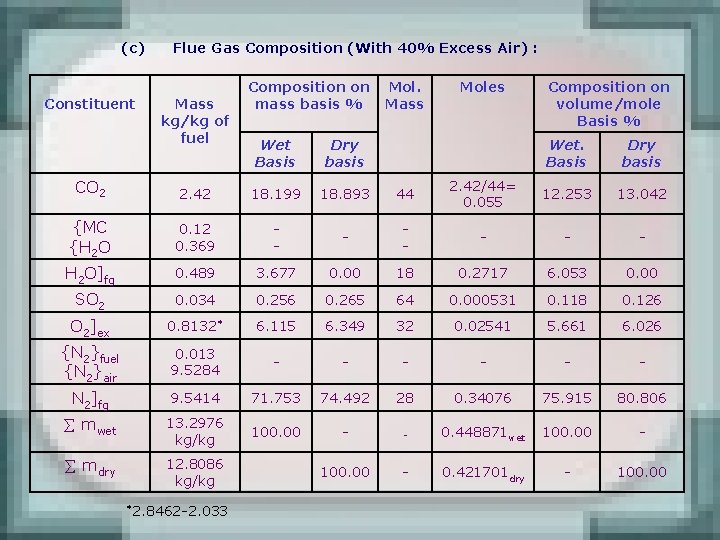
(c) Constituent Flue Gas Composition (With 40% Excess Air) : Mass kg/kg of fuel Composition on mass basis % Wet Basis Dry basis Mol. Mass Moles Composition on volume/mole Basis % Wet. Basis Dry basis CO 2 2. 42 18. 199 18. 893 44 2. 42/44= 0. 055 12. 253 13. 042 {MC {H 2 O 0. 12 0. 369 - - - H 2 O]fg 0. 489 3. 677 0. 00 18 0. 2717 6. 053 0. 00 SO 2 0. 034 0. 256 0. 265 64 0. 000531 0. 118 0. 126 O 2]ex 0. 8132* 6. 115 6. 349 32 0. 02541 5. 661 6. 026 {N 2}fuel {N 2}air 0. 013 9. 5284 - - - N 2]fg 9. 5414 71. 753 74. 492 28 0. 34076 75. 915 80. 806 mwet 13. 2976 kg/kg 100. 00 - - 0. 448871 wet 100. 00 - mdry 12. 8086 kg/kg 100. 00 - 0. 421701 dry - 100. 00 *2. 8462 -2. 033
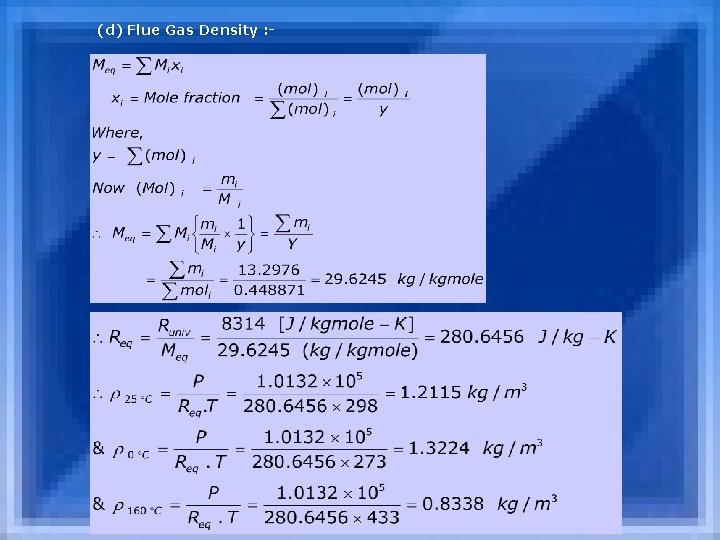
(d) Flue Gas Density : -
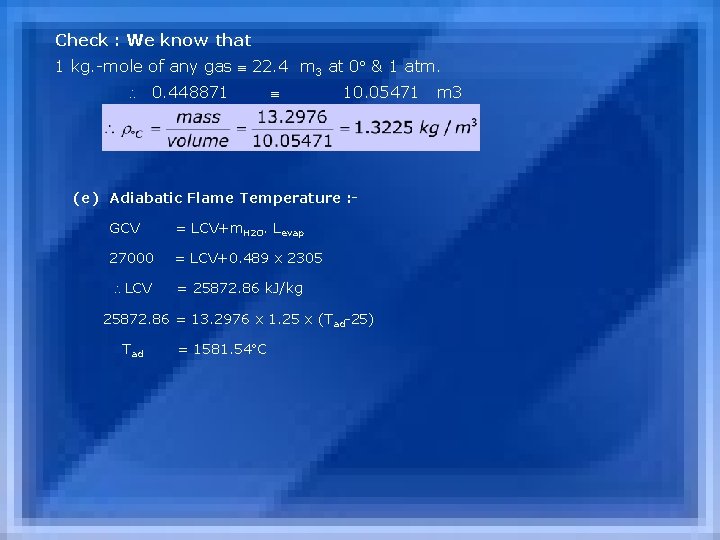
Check : We know that 1 kg. -mole of any gas 22. 4 m 3 at 0 & 1 atm. 0. 448871 10. 05471 m 3 (e) Adiabatic Flame Temperature : GCV = LCV+m. H 2 O. Levap 27000 = LCV+0. 489 x 2305 LCV = 25872. 86 k. J/kg 25872. 86 = 13. 2976 x 1. 25 x (Tad-25) Tad = 1581. 54 C

Home Work Example : The following data refers to a heavy oil fired in a boiler C=85. 4 % N=0. 10 % H=11. 4 % MC=0. 10 % S=2. 8 % Ash=0. 10 % O=0. 1 % GCV=42. 900 k. J/kg Cpfg = 1. 36 k. J/kg-K It is operated at 25% excess air levels. Determine : (i) Amount of air required per kg of fuel (ii) Flue gas analysis on dry basis (iii) Density of wet flue gas (iv) Adiabatic flame temperature

ESTIMATION OF BOILER EFFICIENCY DIRECT METHOD : 1. 2 Efficiency of Boiler : -

NUMERICAL EXAMPLE : A two hour boiler trial was conducted on a coal fired, smoke-tube boiler in a process house & the following data was collected. Rating : Equivalent Evaporation = 5 T/h Max. Pressure = 10. 5 kg/cm 2 Av. Steam Pressure during trial = 7. 5 kg/cm 2 (g) Barometric Pressure = 1. 0132 bar Av. Steam temperature = 179 C O 2 in flue gas (dry basis) = 6. 2 % Ambient temperature = 35 C Unburnt in Ash = 7. 2 % Size of feed tank = 2. 5 m x 3. 0 m Depression of water level during trial = 1. 87 m Temperature of feed water = 70 C GCV of fuel = 27000 k. J/kg Coal consumption during trial = 1025 kg Determine : (i) Boiler output in k. W (ii) Equivalent evaporation in T/h (iii) Boiler efficiency, in %
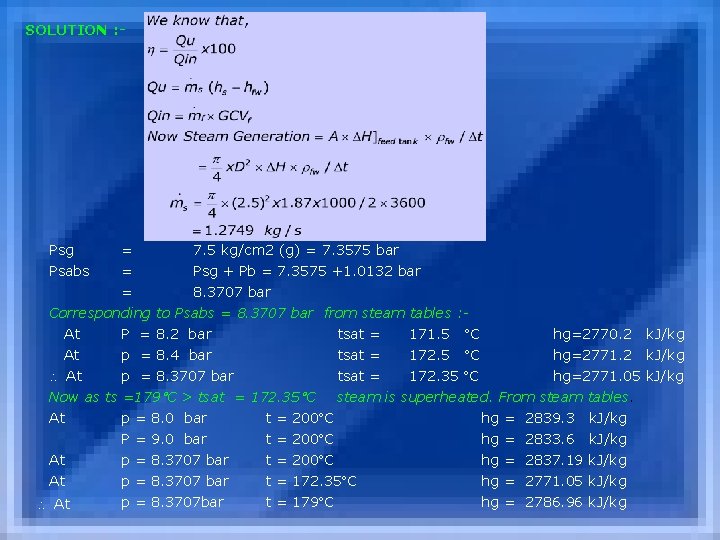
SOLUTION : - Psg = 7. 5 kg/cm 2 (g) = 7. 3575 bar Psabs = Psg + Pb = 7. 3575 +1. 0132 bar = 8. 3707 bar Corresponding to Psabs = 8. 3707 bar from steam tables : At P = 8. 2 bar tsat = 171. 5 C At p = 8. 4 bar tsat = 172. 5 C hg=2770. 2 k. J/kg hg=2771. 2 k. J/kg At p = 8. 3707 bar tsat = 172. 35 C hg=2771. 05 k. J/kg Now as ts =179 C > tsat = 172. 35 C steam is superheated. From steam tables. At p = 8. 0 bar t = 200 C hg = 2839. 3 k. J/kg P = 9. 0 bar t = 200 C hg = 2833. 6 k. J/kg At p = 8. 3707 bar t = 200 C hg = 2837. 19 k. J/kg At p = 8. 3707 bar t = 172. 35 C hg = 2771. 05 k. J/kg p = 8. 3707 bar t = 179 C hg = 2786. 96 k. J/kg At

(iii) hffw = ? From steam table at tfw=70 C, hfw=293 k. J/kg

(vi) Equivalent Evaporation : (vii) Steam to Fuel Ratio (viii) Specific Equivalent Evaporation :
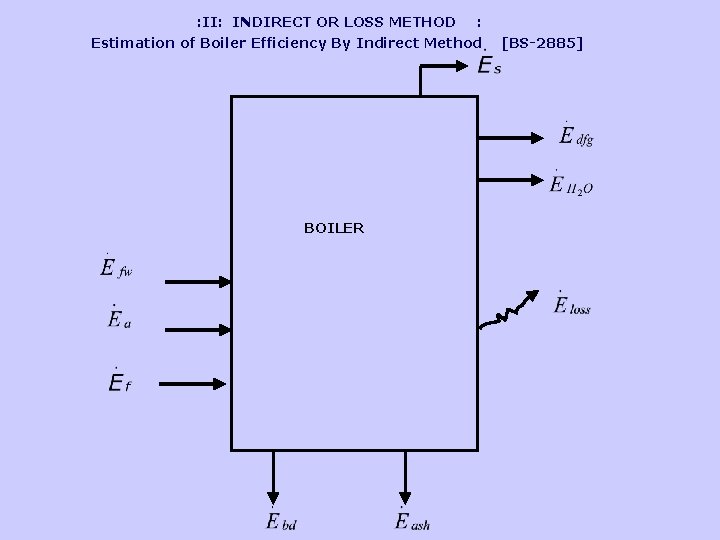
: II: INDIRECT OR LOSS METHOD : Estimation of Boiler Efficiency By Indirect Method BOILER [BS-2885]
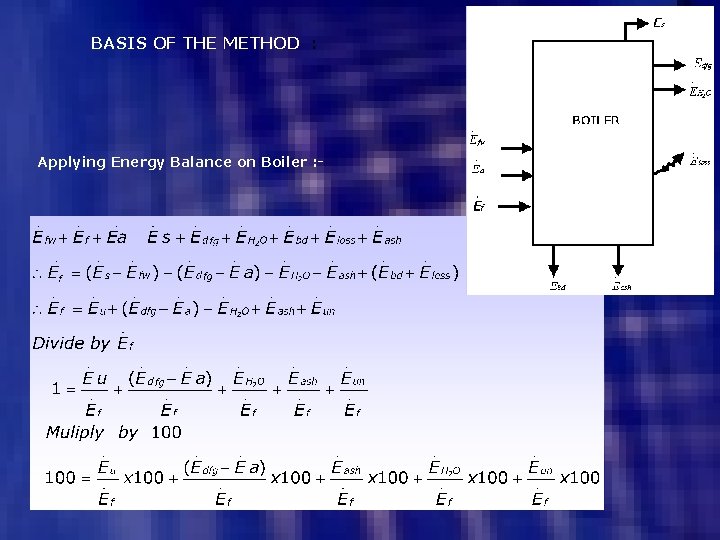
BASIS OF THE METHOD : Applying Energy Balance on Boiler : -

This is the Basis of Loss or Indirect Method
![DETERMINATION OF VARIOUS LOSSES 1. Dry Gas Loss : [Stack Loss] 2. Losses in DETERMINATION OF VARIOUS LOSSES 1. Dry Gas Loss : [Stack Loss] 2. Losses in](http://slidetodoc.com/presentation_image/0591a9ce72e52eed4d9f503a95669627/image-19.jpg)
DETERMINATION OF VARIOUS LOSSES 1. Dry Gas Loss : [Stack Loss] 2. Losses in Ash (b) Sensible heat loss in Ash :

(3) Loss due to hydrogen in fuel : (4) Loss due to moisture in fuel : (5) Unaccounted Losses : (i) Radiation loss = 0. 3 to 1. 0 % (ii) Blow down loss=0. 1 to 0. 5 % (iii) Loss due to unburnt gas 0. 1 to 1. 0 % Total unaccounted losses =1. 5 % THUS BOILER EFFICIENCY =100 -SUM OF VARIOUS LOSSES

NUMERICAL EXAMPLE : The following data refers to a boiler trial conducted on a 5 T/h, 10 kg/cm 2, coal fired smoke tube type boiler : Duration of trial : 1 hour Coal consumption : 510 kg/h Stack Temperature : 210 C O 2 in flue gas : 6. 2 % Ambient Temperature : 35 C Unburnt in Ash : 7. 2 % Steam pressure : 7. 5 kg/cm 2 (g) Steam temperature : 179 C Feed water Temperature : 70 C [By orsat apparatus ] Fuel Analysis : C=66. 0 %, H=4. 1 %, S=1. 7 %, O=7. 2 %, N=1. 3%, MC=12. 0%, Ash=7. 7% GCVf Determine : - : 27000 k. J/kg (i) Various Losses, (ii) Boiler Efficiency (iii) Boiler Output (iv) Steam generation in T/h (v) Equivalent Evaporation in T/h
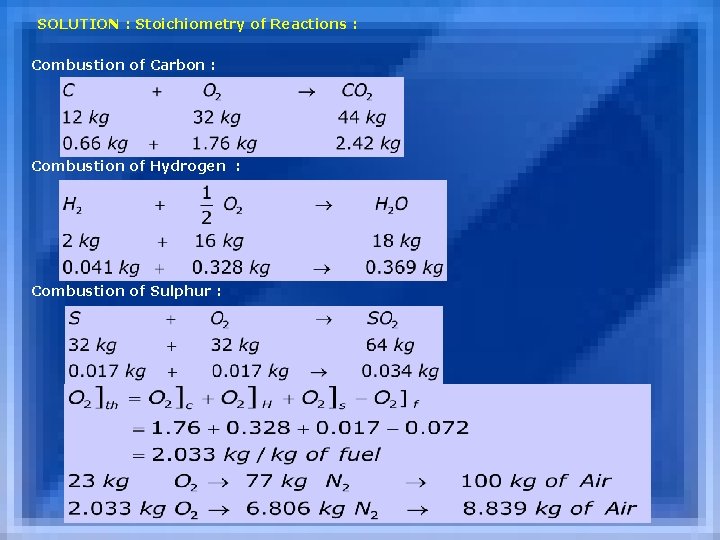
SOLUTION : Stoichiometry of Reactions : Combustion of Carbon : Combustion of Hydrogen : Combustion of Sulphur :
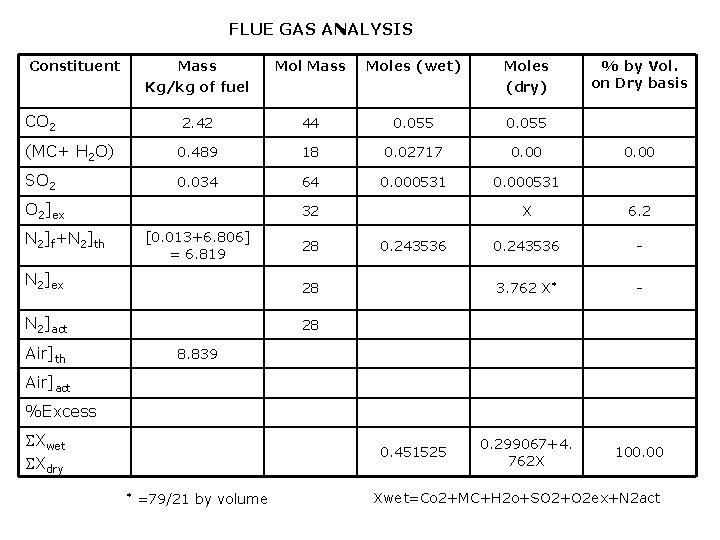
FLUE GAS ANALYSIS Constituent Mass Kg/kg of fuel Mol Mass Moles (wet) Moles (dry) 2. 42 44 0. 055 (MC+ H 2 O) 0. 489 18 0. 02717 0. 00 SO 2 0. 034 64 0. 000531 CO 2 O 2]ex N 2]f+N 2]th 32 [0. 013+6. 806] = 6. 819 28 N 2]ex 28 N 2]act 28 Air]th 0. 243536 % by Vol. on Dry basis 0. 00 X 6. 2 0. 243536 - 3. 762 X* - 0. 299067+4. 762 X 100. 00 8. 839 Air]act %Excess Xwet Xdry 0. 451525 * =79/21 by volume Xwet=Co 2+MC+H 2 o+SO 2+O 2 ex+N 2 act

X=0. 02631 moles of excess O 2 3. 762 x =0. 098978 moles of excess N 2 Total moles of dry flue gas = 0. 424355 moles Total mass of dry flue gas = 12. 8863 kg/ kg of fuel
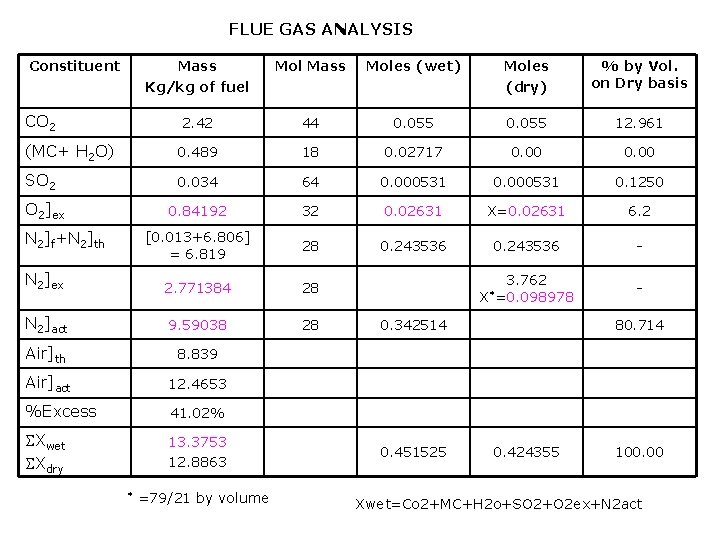
FLUE GAS ANALYSIS Constituent Mass Kg/kg of fuel Mol Mass Moles (wet) Moles (dry) % by Vol. on Dry basis 2. 42 44 0. 055 12. 961 (MC+ H 2 O) 0. 489 18 0. 02717 0. 00 SO 2 0. 034 64 0. 000531 0. 1250 0. 84192 32 0. 02631 X=0. 02631 6. 2 [0. 013+6. 806] = 6. 819 28 0. 243536 - N 2]ex 2. 771384 28 N 2]act 9. 59038 28 Air]th 8. 839 Air]act 12. 4653 %Excess 41. 02% Xwet Xdry 13. 3753 12. 8863 CO 2 O 2]ex N 2]f+N 2]th * =79/21 by volume 3. 762 X*=0. 098978 0. 342514 0. 451525 80. 714 0. 424355 100. 00 Xwet=Co 2+MC+H 2 o+SO 2+O 2 ex+N 2 act
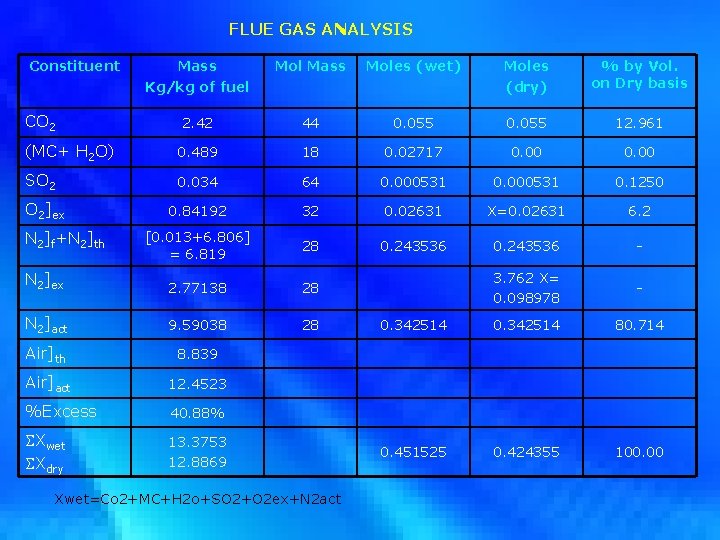
FLUE GAS ANALYSIS Constituent Mass Kg/kg of fuel Mol Mass Moles (wet) Moles (dry) % by Vol. on Dry basis 2. 42 44 0. 055 12. 961 (MC+ H 2 O) 0. 489 18 0. 02717 0. 00 SO 2 0. 034 64 0. 000531 0. 1250 0. 84192 32 0. 02631 X=0. 02631 6. 2 [0. 013+6. 806] = 6. 819 28 0. 243536 - N 2]ex 2. 77138 28 3. 762 X= 0. 098978 - N 2]act 9. 59038 28 0. 342514 80. 714 Air]th 8. 839 Air]act 12. 4523 %Excess 40. 88% Xwet Xdry 13. 3753 12. 8869 0. 451525 0. 424355 100. 00 CO 2 O 2]ex N 2]f+N 2]th Xwet=Co 2+MC+H 2 o+SO 2+O 2 ex+N 2 act

X=0. 02631 moles of excess O 2 3. 762 x =0. 098978 moles of excess N 2 Total moles of dry flue gas = 0. 424355 moles Total mass of dry flue gas = 12. 8863 kg/ kg of fuel 2. Calculation of Losses : (i) Dry Gas Loss : [Stack Loss] (ii) Losses in Ash (a) Loss Due to Combustibles in Ash
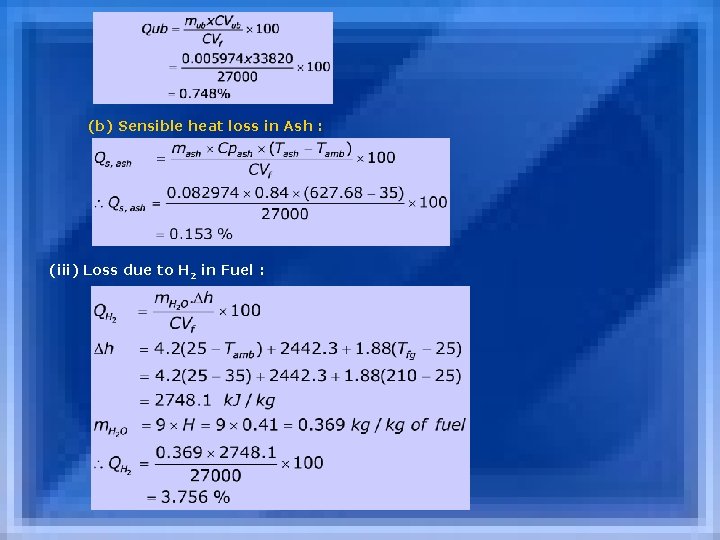
(b) Sensible heat loss in Ash : (iii) Loss due to H 2 in Fuel :

(iv) Loss due to moisture in fuel : (v) Unaccounted losses : (a) Radiation loss : 1. 0% (b)Blow down loss : 0. 5 (c) Loss due to unburnt gas : 0. 2% --------- Qun = 1. 5% From Steam table, at 70 C Water Temperature hfw=293 k. J/kg

For hs=? Ps =7. 5 kg/cm 2 (g) +1. 033 kg/cm 2 (atm) Pabs = 7. 3575 x 105 N/m 2 +1. 0132 x 105 = 8. 3707 x 105 N/m 2 = 8. 3707 bar At this pressure Tsat= ? hg= ? At At p=8. 2 bar p=8. 4 bar p=8. 3707 bar tsat= 171. 5 C tsat= 172. 5 C tsat=172. 35 C hg=2770. 2 k. J/kg hg=2771. 05 k. J/kg Now tsteam = 179 C > Tsat steam is super heated from superheated steam table : At p=8. 0 At p=9. 0 At p=8. 3707 & t=200 C tsat=172. 35, tsup = 179 C, hg=2839. 3 k. J/kg hg=2833. 6 k. J/kg hg=2837. 19 k. J/kg hg=2771. 05 k. J/kg hg=2786. 96 k. J/kg
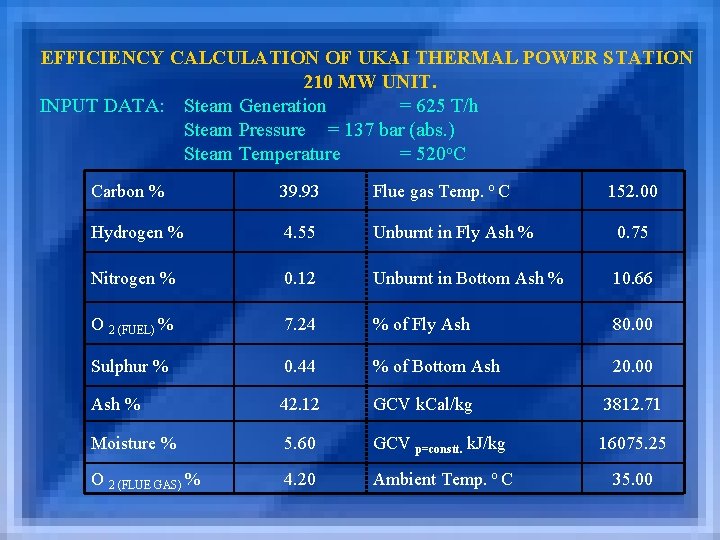
EFFICIENCY CALCULATION OF UKAI THERMAL POWER STATION 210 MW UNIT. INPUT DATA: Steam Generation = 625 T/h Steam Pressure = 137 bar (abs. ) Steam Temperature = 520 o. C Carbon % 39. 93 Flue gas Temp. o C 152. 00 Hydrogen % 4. 55 Unburnt in Fly Ash % 0. 75 Nitrogen % 0. 12 Unburnt in Bottom Ash % 10. 66 O 2 (FUEL) % 7. 24 % of Fly Ash 80. 00 Sulphur % 0. 44 % of Bottom Ash 20. 00 Ash % 42. 12 GCV k. Cal/kg 3812. 71 Moisture % 5. 60 GCV p=constt. k. J/kg 16075. 25 O 2 (FLUE GAS) % 4. 20 Ambient Temp. o C 35. 00

STOICHIOMETRY OF REACTION (1) Combustion of Carbon : C + O 2 12 kg 32 kg 0. 3993 kg 1. 0648 kg (2) Combustion of Hydrogen : H + 1/2 O 2 2 kg 16 kg 0. 0455 kg 0. 364 kg CO 2 44 kg 1. 4641 kg H 2 O 18 kg 0. 4095 kg (3) Combustion of Sulphur : S + O 2 32 kg SO 2 64 kg 0. 0044 kg 0. 0088 kg 0. 0044 kg
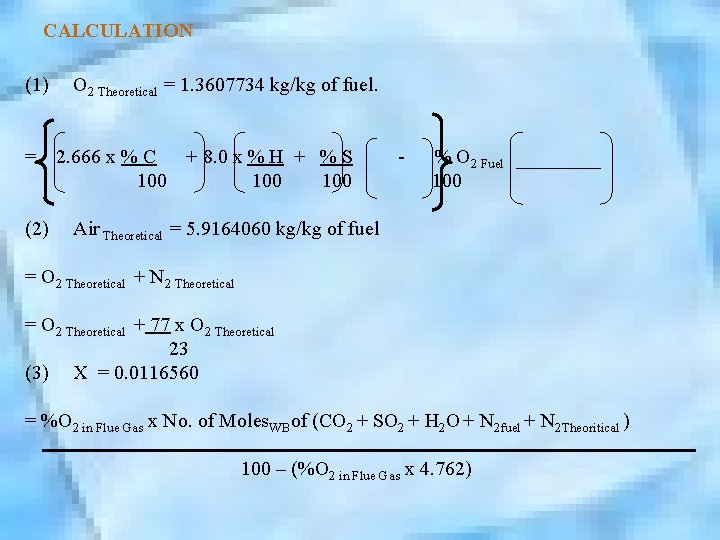
CALCULATION (1) O 2 Theoretical = 1. 3607734 kg/kg of fuel. = 2. 666 x % C + 8. 0 x % H + % S - % O 2 Fuel 100 100 (2) Air Theoretical = 5. 9164060 kg/kg of fuel = O 2 Theoretical + N 2 Theoretical = O 2 Theoretical + 77 x O 2 Theoretical 23 (3) X = 0. 0116560 = %O 2 in Flue Gas x No. of Moles. WBof (CO 2 + SO 2 + H 2 O + N 2 fuel + N 2 Theoritical ) 100 – (%O 2 in Flue Gas x 4. 762)
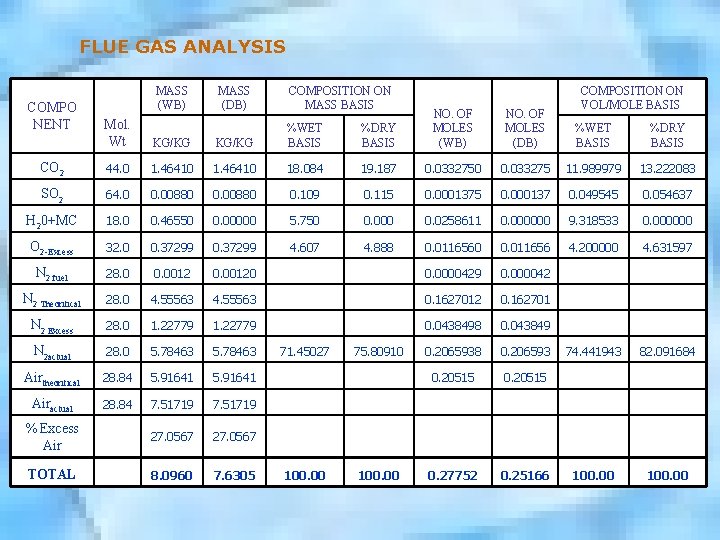
FLUE GAS ANALYSIS MASS (WB) MASS (DB) COMPOSITION ON MASS BASIS Mol. Wt KG/KG %WET BASIS CO 2 44. 0 1. 46410 SO 2 64. 0 0. 00880 H 20+MC 18. 0 O 2 -Excess COMPO NENT COMPOSITION ON VOL/MOLE BASIS %DRY BASIS NO. OF MOLES (WB) NO. OF MOLES (DB) %WET BASIS %DRY BASIS 18. 084 19. 187 0. 0332750 0. 033275 11. 989979 13. 222083 0. 00880 0. 109 0. 115 0. 000137 0. 049545 0. 054637 0. 46550 0. 00000 5. 750 0. 000 0. 0258611 0. 000000 9. 318533 0. 000000 32. 0 0. 37299 4. 607 4. 888 0. 0116560 0. 011656 4. 200000 4. 631597 N 2 fuel 28. 0 0. 00120 0. 0000429 0. 000042 N 2 Theoritical 28. 0 4. 55563 0. 1627012 0. 162701 N 2 Excess 28. 0 1. 22779 0. 0438498 0. 043849 N 2 actual 28. 0 5. 78463 71. 45027 75. 80910 0. 2065938 0. 206593 74. 441943 82. 091684 Airtheoritical 28. 84 5. 91641 0. 20515 Airactual 28. 84 7. 51719 % Excess Air 27. 0567 TOTAL 8. 0960 7. 6305 100. 00 0. 27752 0. 25166 100. 00

Calculated Results: (1) Theoretical Nitrogen: (Kg/ Kg of fuel) = 4. 5556326 = 77 x Theoretical Oxygen 23 (2) Theoretical Air: (Kg/ Kg of fuel) = 5. 9164060 = Theoretical Oxygen + Theoretical Nitrogen (3) Actual Air: (Kg/ Kg of fuel) (4) % Excess Air = 7. 5171922 = 27. 05673
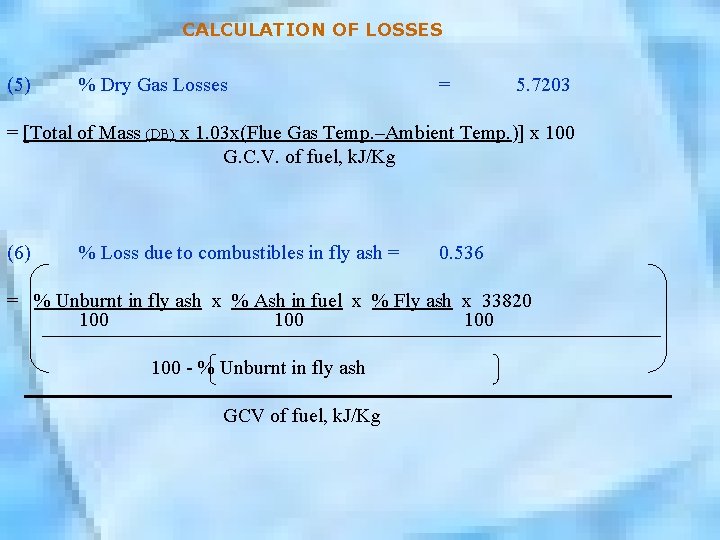
CALCULATION OF LOSSES (5) % Dry Gas Losses = 5. 7203 = [Total of Mass (DB) x 1. 03 x(Flue Gas Temp. –Ambient Temp. )] x 100 G. C. V. of fuel, k. J/Kg (6) % Loss due to combustibles in fly ash = 0. 536 = % Unburnt in fly ash x % Ash in fuel x % Fly ash x 33820 100 100 - % Unburnt in fly ash GCV of fuel, k. J/Kg

(7) % Sensible Heat Loss in Fly Ash = = % Unburnt in fly ash x % Fly ash x % Ash in Fuel 100 (100 - % Unburnt in fly ash) 0. 2076 + 100 % Fly ash x % Ash in fuel x 0. 84 x(Flue gas Temp. – Ambient Temp. ) x 100 ( GCV of Fuel, k. J/Kg )

(8) % Loss due to combustibles in Bottom Ash = 2. 1147 = % Unburnt in Bottom ash x % Ash in fuel x 33820 100 (100 - % Unburnt in Bottom Ash) G. C. V. of fuel, k. J/Kg (9) % Sensible Heat Loss in Bottom Ash = 0. 29202 = % Unburnt in Bottom ash x % Bottom ash x % Ash in fuel + 100 - % Unburnt in Bottom ash % Bottom ash x % Ash in fuel x [0. 84 (627. 68 – Ambient Temp. )] x 100 GCV of fuel, k. J/Kg x 100

(10) % Loss due to Hydrogen in fuel = 6. 72273 = 9 x % Hydrogen in fuel x 4. 2 x (25 – Ambient Temp. ) + 2442. 3 100 + 1. 88 x (Flue gas Temp. – 25) x 100 G. C. V. of Fuel, k. J/Kg (11) % Loss due to Moisture in fuel = 0. 91970 = % Moisture in fuel x 4. 2 x (25 – Ambient Temp. ) + 2442. 30 + 100 1. 88 x (Flue gas Temp. - 25) x 100 G. C. V. of Fuel, k. J/Kg

(12) % Unaccounted loss = 1. 5 (13)Total Losses = (5)+(6)+(7)+………. . +(12) = 18. 0127 % (14) Efficiency = 100 - Total Losses = 100. 00 – 18. 0127 = 81. 987 %
![EFFICIENCY CALCULATION OF GIPCL, SLPP, MANGROL 2*125 MW UNIT, CFBC BOILERS WITH LIMESTONE ADDITION] EFFICIENCY CALCULATION OF GIPCL, SLPP, MANGROL 2*125 MW UNIT, CFBC BOILERS WITH LIMESTONE ADDITION]](http://slidetodoc.com/presentation_image/0591a9ce72e52eed4d9f503a95669627/image-41.jpg)
EFFICIENCY CALCULATION OF GIPCL, SLPP, MANGROL 2*125 MW UNIT, CFBC BOILERS WITH LIMESTONE ADDITION] INPUT DATA: Steam Generation = 390 T/h Steam Pressure = 130 kg/cm 2 (g) Steam Temperature = 540 o. C Carbon % 33. 50 Flue gas Temp. o C Hydrogen % 2. 50 Unburnt in Fly Ash % 1. 50 Nitrogen % 0. 50 Unburnt in Bottom Ash % 0. 05 O 2 (FUEL) % 9. 00 % of Fly Ash 70. 00 Sulphur % 0. 60 % of Bottom Ash 30. 00 Ash % 12. 50 GCV k. Cal/kg 3322. 98 Moisture % 41. 40 GCV p=c k. J/kg 13999. 60 O 2 (FLUE GAS) % 4. 0 Ambient Temp. o C 140. 00 35. 00
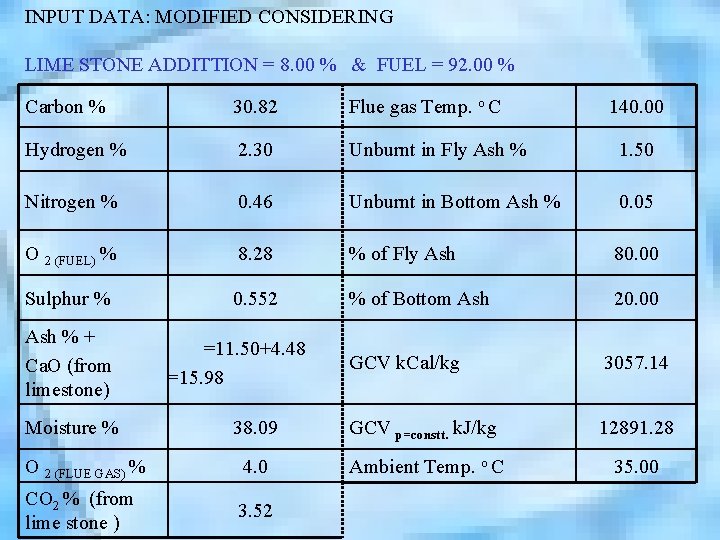
INPUT DATA: MODIFIED CONSIDERING LIME STONE ADDITTION = 8. 00 % & FUEL = 92. 00 % Carbon % 30. 82 Flue gas Temp. o C Hydrogen % 2. 30 Unburnt in Fly Ash % 1. 50 Nitrogen % 0. 46 Unburnt in Bottom Ash % 0. 05 O 2 (FUEL) % 8. 28 % of Fly Ash 80. 00 Sulphur % 0. 552 % of Bottom Ash 20. 00 Ash % + Ca. O (from limestone) =11. 50+4. 48 =15. 98 140. 00 GCV k. Cal/kg 3057. 14 38. 09 GCV p=constt. k. J/kg 12891. 28 O 2 (FLUE GAS) % 4. 0 Ambient Temp. o C 35. 00 CO 2 % (from lime stone ) 3. 52 Moisture %

STOICHIOMETRY OF REACTION (1) Combustion of Carbon : C + O 2 12 kg 32 kg 0. 3082 kg 0. 82187 kg (2) Combustion of Hydrogen : H + 1/2 O 2 2 kg 16 kg 0. 0230 kg 0. 1840 kg CO 2 44 kg 1. 13007 kg H 2 O 18 kg 0. 2070 kg (3) Combustion of Sulphur : S + O 2 32 kg SO 2 64 kg 0. 00552 kg 0. 01104 kg 0. 00552 kg

CALCULATION (1) O 2 Theoretical = 0. 9285661 kg/kg of fuel = 2. 666 x % C + 8. 0 x % H + % S - % O 2 Fuel 100 (2) Air Theoretical = 4. 0372440 kg/kg of fuel = O 2 Theoretical + N 2 Theoretical = O 2 Theoretical + 77 x O 2 Theoretical 23 (3) X = 0. 0084250 = %O 2 in Flue Gas x No. of Moles. WBof (CO 2 + SO 2 + H 2 O + N 2 fuel + N 2 Theoritical ) 100 – (%O 2 in Flue Gas x 4. 762)
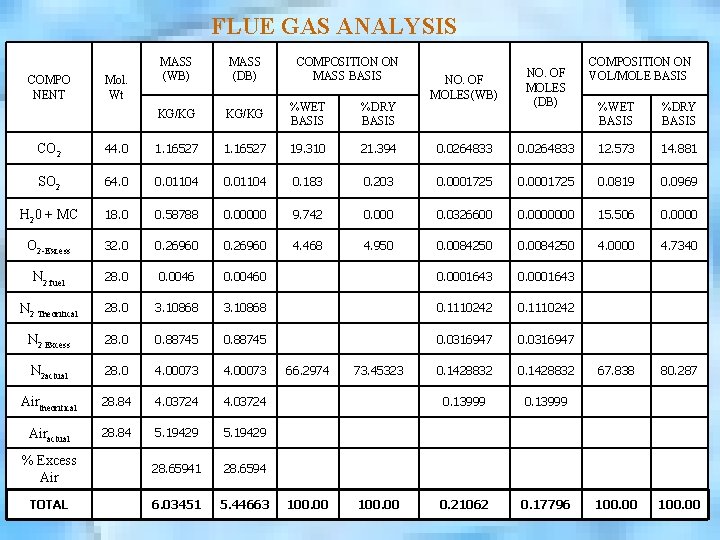
FLUE GAS ANALYSIS COMPO NENT Mol. Wt MASS (WB) MASS (DB) COMPOSITION ON MASS BASIS KG/KG %WET BASIS %DRY BASIS NO. OF MOLES(WB) NO. OF MOLES (DB) COMPOSITION ON VOL/MOLE BASIS %WET BASIS %DRY BASIS CO 2 44. 0 1. 16527 19. 310 21. 394 0. 0264833 12. 573 14. 881 SO 2 64. 0 0. 01104 0. 183 0. 203 0. 0001725 0. 0819 0. 0969 H 20 + MC 18. 0 0. 58788 0. 00000 9. 742 0. 000 0. 0326600 0. 0000000 15. 506 0. 0000 O 2 -Excess 32. 0 0. 26960 4. 468 4. 950 0. 0084250 4. 0000 4. 7340 N 2 fuel 28. 0 0. 00460 0. 0001643 N 2 Theoritical 28. 0 3. 10868 0. 1110242 N 2 Excess 28. 0 0. 88745 0. 0316947 N 2 actual 28. 0 4. 00073 66. 2974 73. 45323 0. 1428832 67. 838 80. 287 Airtheoritical 28. 84 4. 03724 0. 13999 Airactual 28. 84 5. 19429 % Excess Air 28. 65941 28. 6594 TOTAL 6. 03451 5. 44663 100. 00 0. 21062 0. 17796 100. 00

Calculated Results: (1) Theoretical Nitrogen: (Kg/ Kg of fuel) = 3. 1086779 = 77 x Theoretical Oxygen 23 (2) Theoretical Air: (Kg/ Kg of fuel) = 4. 0372440 = Theoretical Oxygen + Theoretical Nitrogen (3) Actual Air: (Kg/ Kg of fuel) (4) % Excess Air = 5. 1942942 = 28. 65941
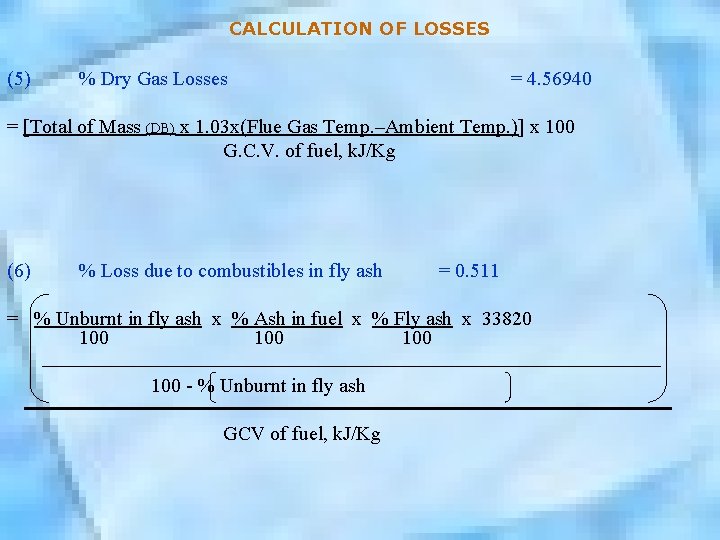
CALCULATION OF LOSSES (5) % Dry Gas Losses = 4. 56940 = [Total of Mass (DB) x 1. 03 x(Flue Gas Temp. –Ambient Temp. )] x 100 G. C. V. of fuel, k. J/Kg (6) % Loss due to combustibles in fly ash = 0. 511 = % Unburnt in fly ash x % Ash in fuel x % Fly ash x 33820 100 100 - % Unburnt in fly ash GCV of fuel, k. J/Kg

(7) % Sensible Heat Loss in Fly Ash = = % Unburnt in fly ash x % Fly ash x % Ash in Fuel 100 (100 - % Unburnt in fly ash) 0. 0888 + 100 % Fly ash x % Ash in fuel x 0. 84 x(Flue gas Temp. – Ambient Temp. ) x 100 ( GCV of Fuel, k. J/Kg )

(8) % Loss due to combustibles in Bottom Ash = 0. 0042 = % Unburnt in Bottom ash x % Ash in fuel x 33820 x 100 100 (100 - % Unburnt in Bottom Ash) G. C. V. of fuel, k. J/Kg (9) % Sensible Heat Loss in Bottom Ash = 0. 12349 = % Unburnt in Bottom ash x % Bottom ash x % Ash in fuel + 100 - % Unburnt in Bottom ash % Bottom ash x % Ash in fuel x [0. 84 (627. 68 – Ambient Temp. )] x 100 GCV of fuel, k. J/Kg

(10) % Loss due to Hydrogen in fuel = 4. 20141 = 9 x % Hydrogen in fuel x 4. 2 x (25 – Ambient Temp. ) + 2442. 3 100 + 1. 88 x (Flue gas Temp. – 25) x 100 G. C. V. of Fuel, k. J/Kg (11) % Loss due to Moisture in fuel = = % Moisture in fuel x 4. 2 x (25 – Ambient Temp. ) + 2442. 3 + 100 1. 88 x (Flue gas Temp. - 25) x 100 G. C. V. of Fuel, k. J/Kg 7. 73355

(12)% Unaccounted loss = 1. 5 (13) Total Losses = (5)+(6)+(7)+………. . +(12) = 18. 7316 % (14)Efficiency = 100 - Total Losses = 100. 00 – 18. 7316 = 81. 268 %

Conclusion • Maximize Useful Heat Energy • Minimize Losses -Improve combustion Efficiency -Improve Heat Transfer Efficiency

- Slides: 53