Stoichiometry Molar relationships in chemical reactions 3 1





























- Slides: 36

Stoichiometry Molar relationships in chemical reactions

3. 1 Chemical Equations: A Review • Law of conservation of mass • Relationship between reactant and products produces a balanced chemical equation • Reactants Products A+B C+D • Balancing chemical eqns

Balancing Chemical Equations • Must have correct chemical formulas • Change coefficients as needed to balance equation • Do not change subscripts of chemical formulas • Indicate physical states by writing in parentheses (s), (l), (g), (aq) • Begin with an element that appears in only one reactant and product • If possible, do not begin with O or H

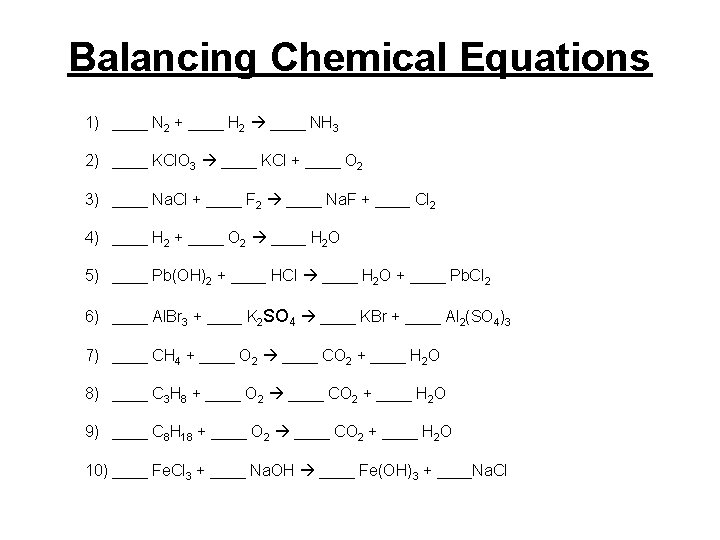
Balancing Chemical Equations 1) ____ N 2 + ____ H 2 ____ NH 3 2) ____ KCl. O 3 ____ KCl + ____ O 2 3) ____ Na. Cl + ____ F 2 ____ Na. F + ____ Cl 2 4) ____ H 2 + ____ O 2 ____ H 2 O 5) ____ Pb(OH)2 + ____ HCl ____ H 2 O + ____ Pb. Cl 2 6) ____ Al. Br 3 + ____ K 2 SO 4 ____ KBr + ____ Al 2(SO 4)3 7) ____ CH 4 + ____ O 2 ____ CO 2 + ____ H 2 O 8) ____ C 3 H 8 + ____ O 2 ____ CO 2 + ____ H 2 O 9) ____ C 8 H 18 + ____ O 2 ____ CO 2 + ____ H 2 O 10) ____ Fe. Cl 3 + ____ Na. OH ____ Fe(OH)3 + ____Na. Cl

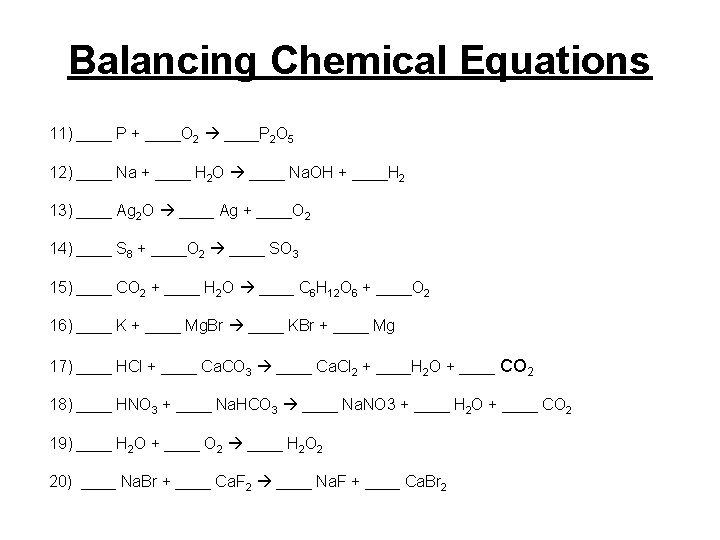
Balancing Chemical Equations 11) ____ P + ____O 2 ____P 2 O 5 12) ____ Na + ____ H 2 O ____ Na. OH + ____H 2 13) ____ Ag 2 O ____ Ag + ____O 2 14) ____ S 8 + ____O 2 ____ SO 3 15) ____ CO 2 + ____ H 2 O ____ C 6 H 12 O 6 + ____O 2 16) ____ K + ____ Mg. Br ____ KBr + ____ Mg 17) ____ HCl + ____ Ca. CO 3 ____ Ca. Cl 2 + ____H 2 O + ____ CO 2 18) ____ HNO 3 + ____ Na. HCO 3 ____ Na. NO 3 + ____ H 2 O + ____ CO 2 19) ____ H 2 O + ____ O 2 ____ H 2 O 2 20) ____ Na. Br + ____ Ca. F 2 ____ Na. F + ____ Ca. Br 2

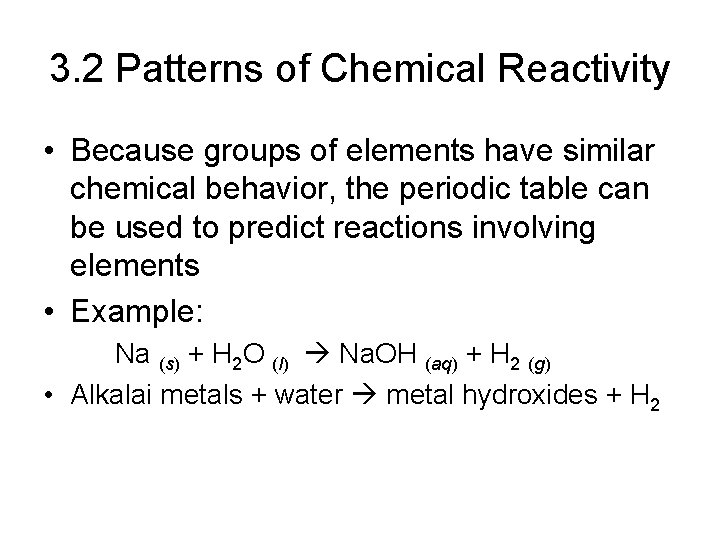
3. 2 Patterns of Chemical Reactivity • Because groups of elements have similar chemical behavior, the periodic table can be used to predict reactions involving elements • Example: Na (s) + H 2 O (l) Na. OH (aq) + H 2 (g) • Alkalai metals + water metal hydroxides + H 2

Types of Chemical Reactions • Synthesis (Combination) reactions • Two or more substances combine to form a new substance • Often involve elements combining to form a compound • A + B C 4 Na (s) + O 2 (g) 2 Na 2 O (s) sodium oxide

Types of Chemical Reactions • Decomposition reactions • A single substance breaks down into two or more smaller substances • A B + C H 2 CO 3 (aq) CO 2 (g) + H 2 O (l) carbonic acid

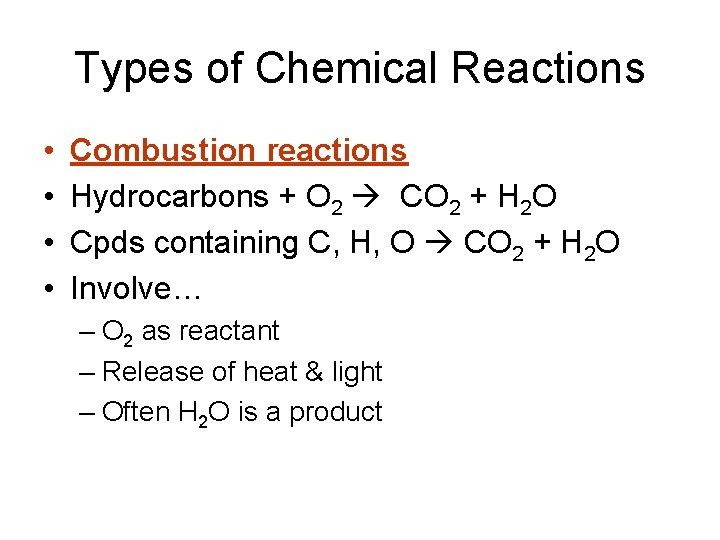
Types of Chemical Reactions • • Combustion reactions Hydrocarbons + O 2 CO 2 + H 2 O Cpds containing C, H, O CO 2 + H 2 O Involve… – O 2 as reactant – Release of heat & light – Often H 2 O is a product

3. 3 Formula Weights • Atomic mass unit (amu) • A unit invented to describe extremely small masses • Defined as 1/12 the mass of an atom of 12 C • 1 amu = 1. 66054 x 10 -24 g

Average Atomic Masses • Atomic masses given on the PT are weighted averages of all the isotopes of that element • Equals the sum of the products of the (mass) x (frequency) of the isotopes • mavg = Σ (m 1 f 1) + (m 2 f 2) + (m 3 f 3) + …. .

Average atomic mass • mavg = Σ (m 1 f 1) + (m 2 f 2) + (m 3 f 3) + …. . • Example • Chlorine occurs as two isotopes: • Isotope amu freq • 35 Cl 34. 964 75. 53% • 37 Cl 36. 966 24. 47% • Atomic mass of chlorine = (m x f) 26. 41 +9. 05 35. 46

Formula & Molecular Weights • Formula mass refers to ionic cpds • Molecular mass refers to covalent cpds • Is the sum of atomic masses of the molecule Calculate mol mass of Ca (NO 3)2

Percent Composition of Formulas • Percentage by mass contributed by each element in the formula

Percent Composition • Determine the % composition of all elements in sucrose, C 12 H 22 O 11 • %C = (12 x 12) / 342 = 42. 0% • %H = (1 x 22) / 342 = 6. 4% • %O = (16 x 11) / 342 = 51. 6%

3. 4 The Mole • • Is a counting unit Avagadro’s number = 6. 022 x 1023 Interconvert between moles & particles Molar mass = the mass of one mole of a substance • Molar mass of elements = atomic mass in grams

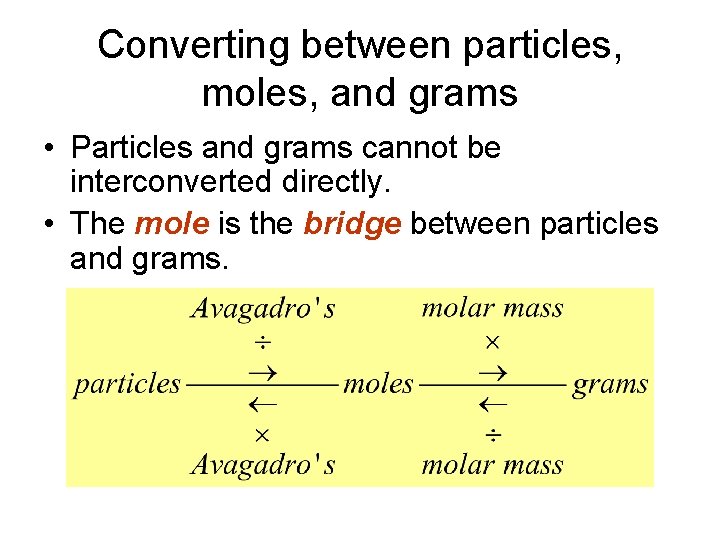
Converting between particles, moles, and grams • Particles and grams cannot be interconverted directly. • The mole is the bridge between particles and grams.

3. 5 Empirical Formula Analysis • To determine empirical formula from percent composition data • Empirical formula gives simplest ratio of atoms in a compound • Therefore, always convert to moles when calculating empirical formulas • Sample exercise 3. 13

Combustion Analysis • Determines empirical formula based upon analysis of products of combustion • Example: 18. 4 g of a compound of CHO produced 41. 7 g CO 2 and 19. 65 g H 2 O • Cx. Hy. Oz + O 2 CO 2 + H 2 O 18. 4 g 41. 7 g 19. 65 g • All carbon in CO 2 came from the CHO • All hydrogen in H 2 O came from CHO

Combustion Analysis 1. Determine amt of C in CHO by determining amt of C in the CO 2 produced. %C in CO 2 x g CO 2 = g C in CHO. 2729 x 41. 17 = 11. 23 g C = 0. 935 mol C

Combustion Analysis 2. Determine amt of H in CHO by determining amt of H in the H 2 O produced. %H in H 2 O x g H 2 O = g H in CHO. 1119 x 19. 65 = 2. 20 g H = 2. 18 mol H

Combustion Analysis • Determine amt of O in CHO by determining amt of H in the H 2 O produced • By law of conservation of mass, grams of CHO = g. C + g. H + g. O • So grams O = grams CHO – g. C – g. H • 18. 40 – 11. 23 – 2. 20 = 4. 97 g. O =. 311 mol O • Mole ratio C 0. 935 H 2. 18 O 0. 311 • Empirical formula C 3 H 7 O

3. 6 Quantitative Information from Balanced Equations • Balanced chemical equations are like specific recipes • Balanced equations tell us …. – the relative amounts of reactants (ingredients) – the relative amounts of products – other relevant conditions needed (e. g. heat) • The unit of measure in a balanced equation is the mole – Indicated by coefficients

Apple Snapple Oatmeal (www. Mr. Breakfast. com) Apple Snapple Oatmeal (4 servings) 1 medium apple or pear 3 cups apple juice or cider 1 and 1/3 cups regular rolled oats 1/4 cup raisins or chopped pitted dates 1/4 teaspoon ground cinnamon milk (if you like) brown sugar (if you like)

Stoichiometric Calculations The coefficients in the balanced equation give the ratio of moles of reactants and products.

3. 6 Quantitative Information from Balanced Equations • Mole ratio used to calculate actual molar amounts in a balanced eqn • Sample Problem 3. 16 • Determine mass of water produced from combustion of 1. 00 g glucose • C 6 H 12 O 6 + __ O 2 __ CO 2 +__ H 2 O • C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O

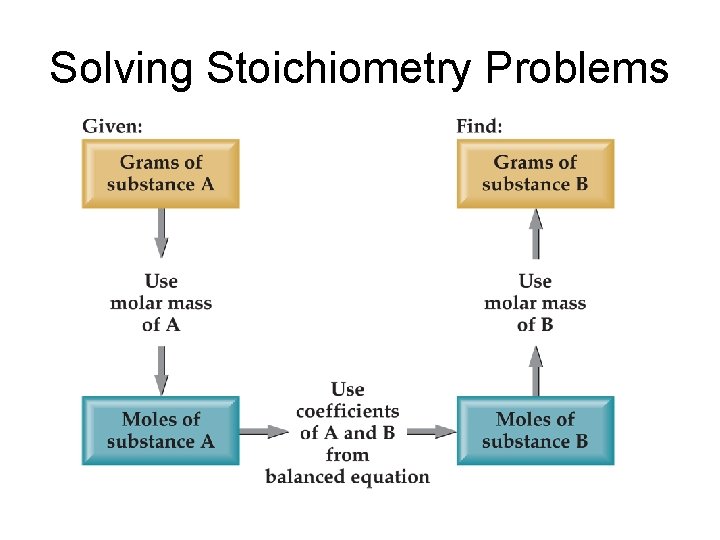
Solving Stoichiometry Problems

Sample problem • Determine mass of water produced from combustion of 1. 00 g glucose • C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O • Determine moles glucose • Determine moles water • Determine grams water

3. 7 Limiting Reactants • Stoichiometrically least abundant reactant • Completely consumed in the reaction • Determines theoretical yield of the reaction • When all reactants are present in stoichiometric amounts, all are limiting reagents

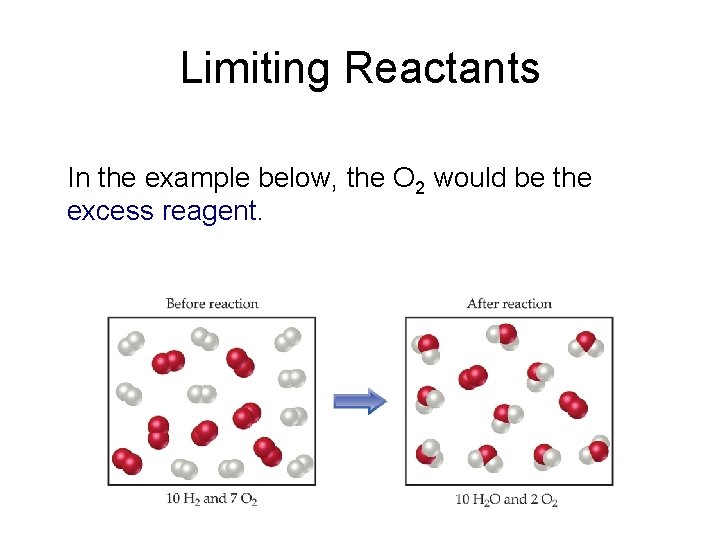
Limiting Reactants • The limiting reactant is the reactant present in the smallest stoichiometric amount. – In other words, it’s the reactant you’ll run out of first (in this case, the H 2).

Limiting Reactants In the example below, the O 2 would be the excess reagent.

Limiting Reagents • How many moles of ammonia can be produced when 3. 0 mol nitrogen and 6. 0 mol of hydrogen are permitted to react? • Write & balance the equation • Determine the LR to determine yield – Reactant least yield is the LR • What is the excess reagent? How much is left over?

Theoretical & Actual Yields • Theoretical Yield • Quantity of product produced when 100% of limiting reagent reacts • Determined from balanced equation • Actual Yield • Quantity of product actually produced • No reaction is 100% efficient, therefore…. • Theoretical Yield does not equal Actual Yield

Sample Problem 3. 20 • 2 C 6 H 12 + 5 O 2 2 H 2 C 6 H 8 O 4 + 2 H 2 O • Given 25. 0 g cyclohexane and excess oxygen, determine theoretical yield of adipic acid

Percent Yield • Expresses the efficiency of a reaction • Is ratio of actual yield to theoretical yield • Percent Yield = Actual Y/ Theroretical Y %Y = AY / TY

Sample Problem 3. 20 (cont) • 2 C 6 H 12 + 5 O 2 2 H 2 C 6 H 8 O 4 + 2 H 2 O • Theoretical yield = 43. 5 g adipic acid (from first part) • If actual yield of adipic acid was 33. 5 g, determine percent yield of reaction • %Y = Actual yield / Theoretical yield • %Y = 33. 5 g/43. 5 g =. 77 = 77%