Stoichiometry involves the mass relationships between the reactants

Stoichiometry involves the mass relationships between the reactants and products in a chemical reaction. RULE # 1 All ratios are MOLE ratios. So you must always find the moles before you can find the mass.

Calculations molar mass Grams Avogadro’s number Moles Particles Everything must go through Moles!!!
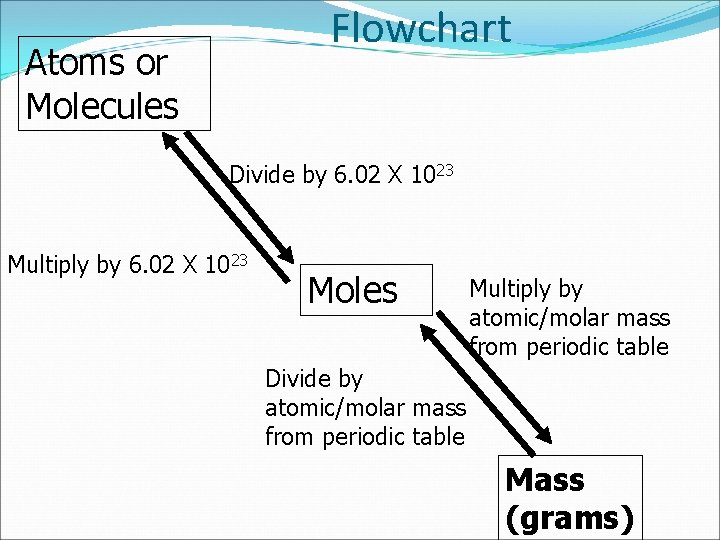
Flowchart Atoms or Molecules Divide by 6. 02 X 1023 Multiply by 6. 02 X 1023 Moles Multiply by atomic/molar mass from periodic table Divide by atomic/molar mass from periodic table Mass (grams)

Write all the mole ratios for the following example 4 NH 3 + 6 NO 5 N 2 + 6 H 2 O

Example 2 2 Al + 3 H 2 SO 4 Al 2(SO 4)3 + 3 H 2

In a spacecraft the carbon dioxide exhaled by astronauts can be removed by its reaction with lithium hydroxide according to the following chemical equation: CO 2 + 2 Li. OH Li 2 CO 3 + H 2 O How many moles of lithium hydroxide are required to react with 20 moles of CO 2, the average amount exhaled by a person each day?

In photosynthesis, plants use energy from the sun to produce glucose, C 6 H 12 O 6, and oxygen, from the reaction of carbon dioxide and water. What mass, in grams, of glucose is produced when 3. 00 moles of water react with carbon dioxide?

What mass of carbon dioxide, in grams, is needed to react with 3. 00 moles of H 2 O in the photosynthetic reaction described in the last problem?

The first step in the industrial manufacture of nitric acid is the catalytic oxidation of ammonia: NH 3 + O 2 NO + H 2 O Balance the equation. The reaction is run using 824 g of NH 3 and excess oxygen. A. How many moles of H 2 O are formed? B. How many moles of NO are formed?

Milk of magnesia, a suspension of Mg(OH)2 in water, reacts with stomach acid, HCl, in a neutralization reaction. Mg(OH)2 + 2 HCl 2 H 2 O + Mg. Cl 2 a. What mass in grams of Mg. Cl 2 will be produced if 3. 00 g of Mg(OH)2 reacts? b. What mass in grams of HCl is required to completely react with 3. 00 grams of Mg. Cl 2

problems: Mole to mole Mole to mass Mass to mass Most problems are mass to mass problems, because we want to know what mass of a substance we would need to produce a certain mass of another substance
- Slides: 11