Stoichiometry Interpreting Chemical Equations Chapter 12 Recipe Analogy






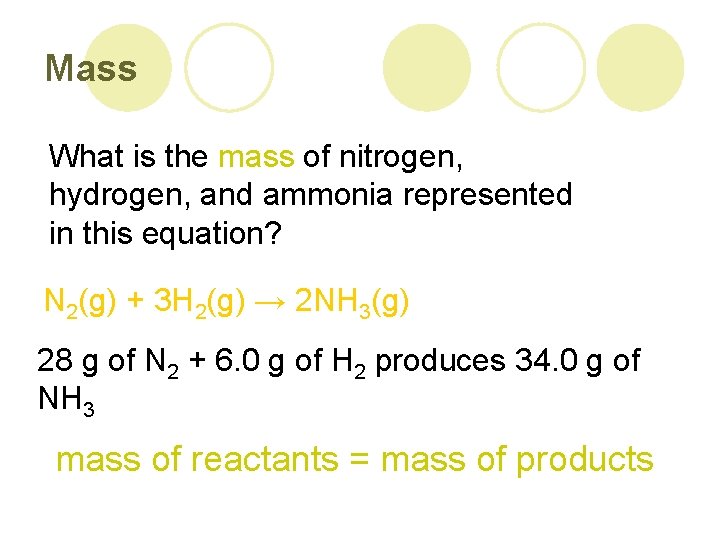


- Slides: 12

Stoichiometry Interpreting Chemical Equations Chapter 12

Recipe Analogy l l l 1 cup shortening 3/4 cup brown sugar 3/4 cup granulated sugar 1 Tsp. vanilla 1/2 Tsp. water 2 eggs 2 cup flour 1 tbs. baking soda 1 tsp. salt 1 cup chopped nuts 2 cups chocolate chips l Mix all ingredients and bake in a 350 Oven for 8 -12 minutes or until golden brown. Makes 50 chocolate chip cookies. ingredients = reactants cookies = products

Chemical Equations The production of ammonia, a fertilizer, can be represented by the following equation: N 2(g) + 3 H 2(g) → 2 NH 3(g) What does this equation tell us?

Chemical Equations l. Particles l. Moles l. Mass l. Volume - coefficients - convert moles to mass - convert moles to volume

Particles How many particles of nitrogen and hydrogen are needed to produce ammonia? N 2(g) + 3 H 2(g) → 2 NH 3(g) 1 molecule of N 2 + 3 molecules of H 2 produces 2 molecules of NH 3 1: 3: 2 ratio of particles

Moles How many moles of nitrogen and hydrogen are needed to produce ammonia? N 2(g) + 3 H 2(g) → 2 NH 3(g) 1 mole of N 2 + 3 moles of H 2 produces 2 moles of NH 3 1: 3: 2 ratio of moles
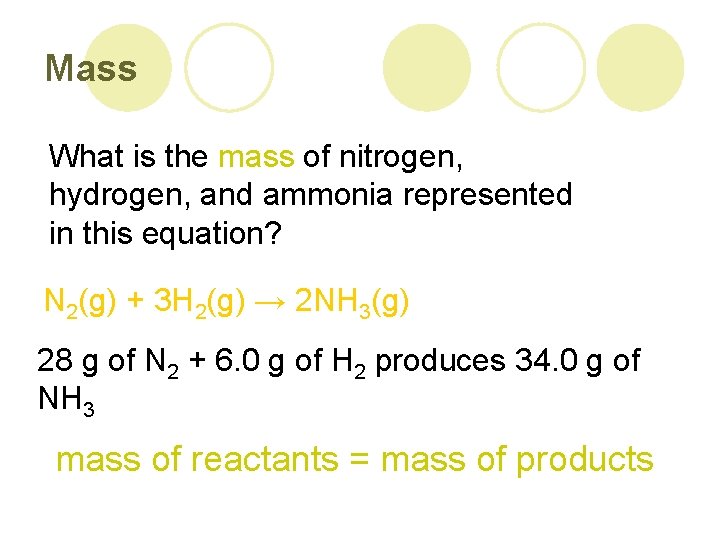
Mass What is the mass of nitrogen, hydrogen, and ammonia represented in this equation? N 2(g) + 3 H 2(g) → 2 NH 3(g) 28 g of N 2 + 6. 0 g of H 2 produces 34. 0 g of NH 3 mass of reactants = mass of products

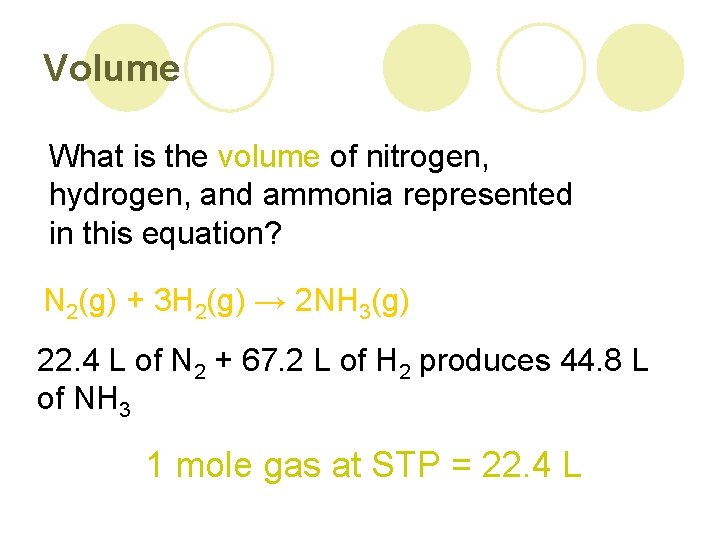
Volume What is the volume of nitrogen, hydrogen, and ammonia represented in this equation? N 2(g) + 3 H 2(g) → 2 NH 3(g) 22. 4 L of N 2 + 67. 2 L of H 2 produces 44. 8 L of NH 3 1 mole gas at STP = 22. 4 L

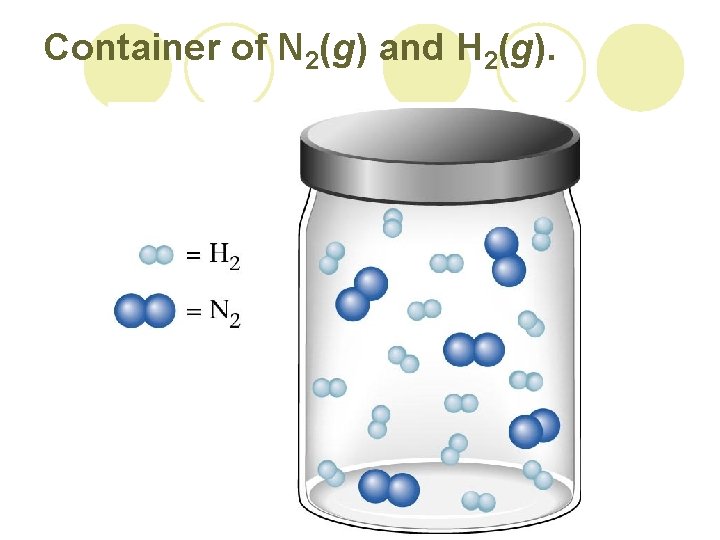
Container of N 2(g) and H 2(g).

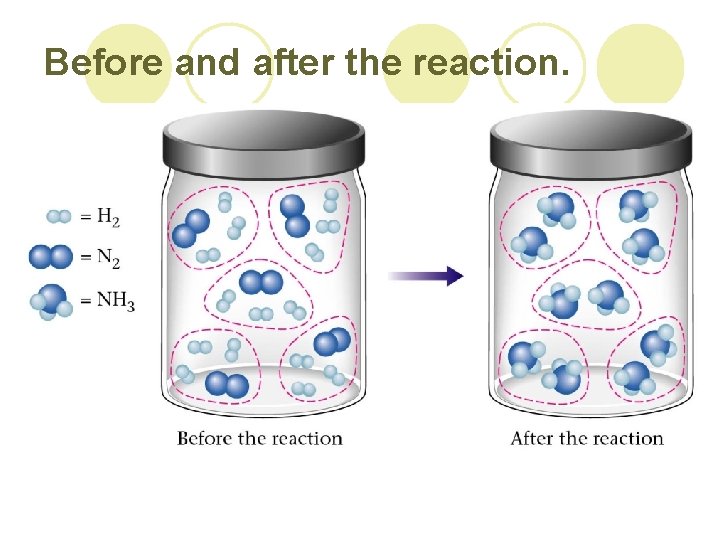
Before and after the reaction.

Practice l Hydrogen sulfide, a foul-smelling gas, is found in nature in volcanic areas. Interpret the equation for the production of hydrogen sulfide in terms of number of representative particles, number of moles, mass and volume of reactants and products. 2 H 2 S(g) + 3 O 2(g) → 2 SO 2(g) + 2 H 2 O(g)

Practice l Interpret this equation in terms of the following quantities: number of particles, number of moles, mass of reactants and products. 2 K(s) + 2 H 20(l) → 2 KOH(aq) + H 2(g)