Stoichiometry Easy as 1 2 3 Stoichiometry The








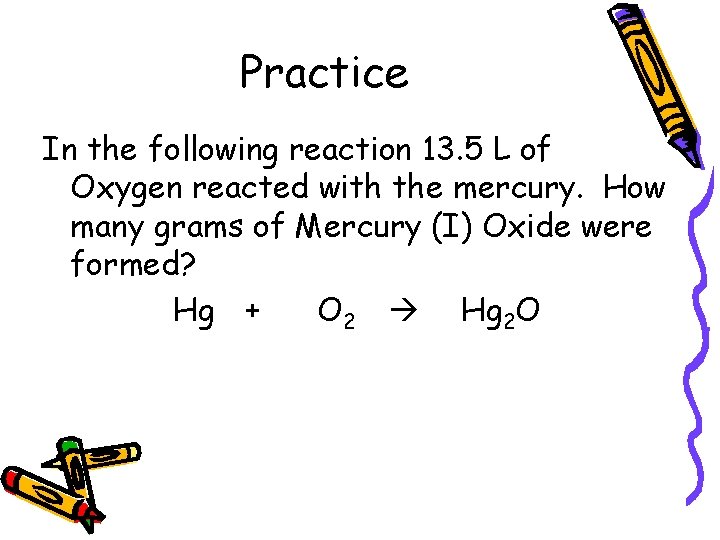
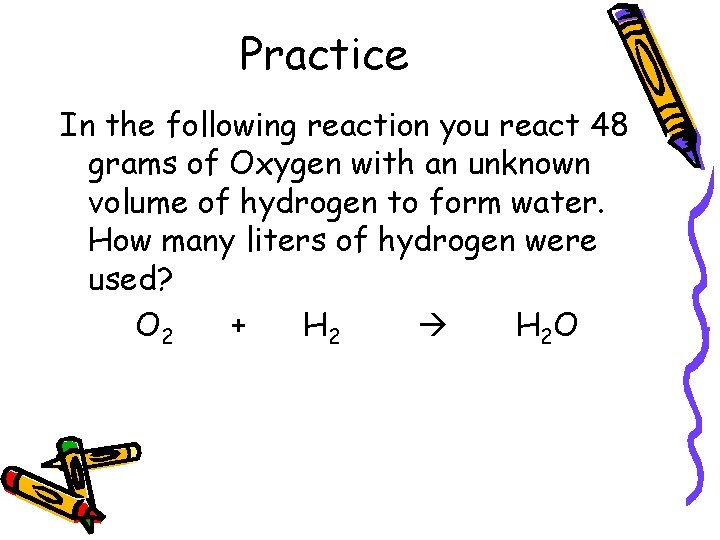









- Slides: 23

Stoichiometry Easy as 1, 2, 3!!!

Stoichiometry • The study of the relationships or ratios between two or more substances undergoing chemical reactions ***Must balance reaction 1 st before solving***

Mole Ratio • In a balanced equation you can create ratios from one compound to another creating a bridge conversion factor __ Mg + __ O 2 __ Mg. O
MASS TO MASS

Diving In (Practice) If you have 6. 85 g of phosphorus how much oxygen gas (in grams) do you need to combine with it? __ P + __ O 2 __ P 4 O 10

Practice If you begin with 200 g of sodium hydroxide how many grams of sodium sulfate will be produced? Na. OH + H 2 SO 4 H 2 O + Na 2 SO 4

Practice If you are give 8 g of oxygen gas how many grams of product can be produced? __ P + __ O 2 __ P 4 O 10

Practice If you produce 250 g of Li 2 SO 4 how many grams of Li. NO 3 did you begin with? Pb(SO 4)2 + Li. NO 3 Pb(NO 3) 4 + Li 2 SO 4

Practice If you create 7. 46 g of P 4 O 10 how many grams of phosphorus would you need to start with? __ P + __ O 2 __ P 4 O 10
MASS TO VOLUME
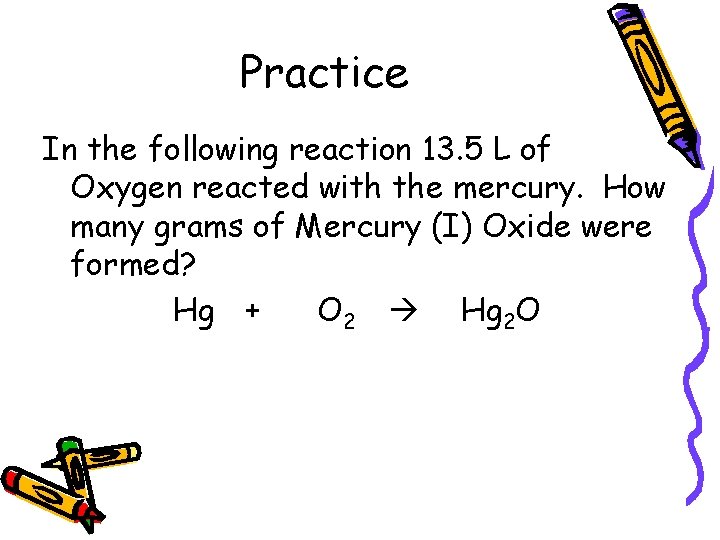
Practice In the following reaction 13. 5 L of Oxygen reacted with the mercury. How many grams of Mercury (I) Oxide were formed? Hg + O 2 Hg 2 O
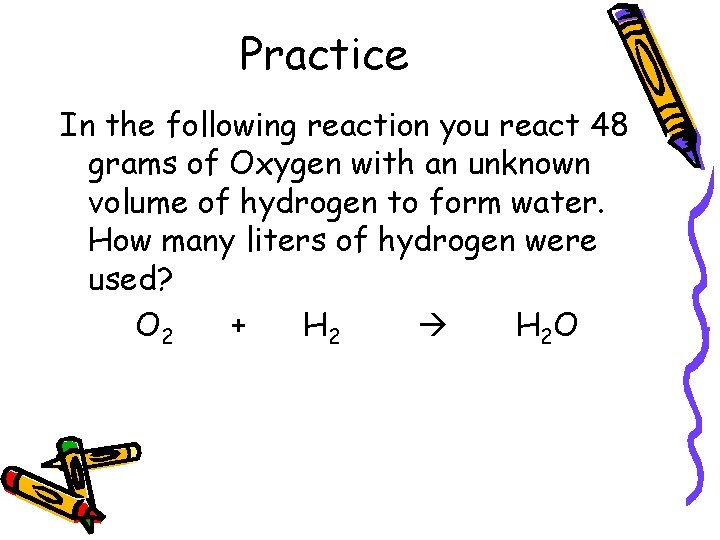
Practice In the following reaction you react 48 grams of Oxygen with an unknown volume of hydrogen to form water. How many liters of hydrogen were used? O 2 + H 2 O

VOLUME TO VOLUME

Practice Nitric Oxide (NO) reacts instantly with oxygen gas to give Nitrogen Dioxide, a dark brown gas. If you finished with 65. 2 liters of Nitrogen Dioxide, how many liters of Oxygen were used? NO + O 2 NO 2

PERCENT YIELD

Formula Actual x 100 Theoretical

Percent Yield In the following reaction, you started off with 35 grams of Hydrogen. When BT did the experiment, they produced 302. 25 grams of Dihydrogen Monoxide. What is their percent yield? H 2 + O 2 H 2 O

Percent Yield In the following reaction, you started off with 2000 grams of Rubidium. When BT did the experiment, they produced 1500 grams of Rubidium Sulfide. What is their percent yield? Rb + S 8 Rb 2 S

LIMITING AND EXCESS REACTANTS

Practice If 5. 00 grams of copper metal react with a solution containing 20. 0 grams of Ag. NO 3, which reactant is limiting? What is in excess and how much of it is left over? Cu + Ag. NO 3 Cu(NO 3)2 + Ag

Practice What reactant is limiting if 3 liters of Cl 2 react with a solution containing 25. 0 grams of Na. Br? What is in excess and how much of it is left over? Na. Br + Cl 2 Na. Cl + Br 2
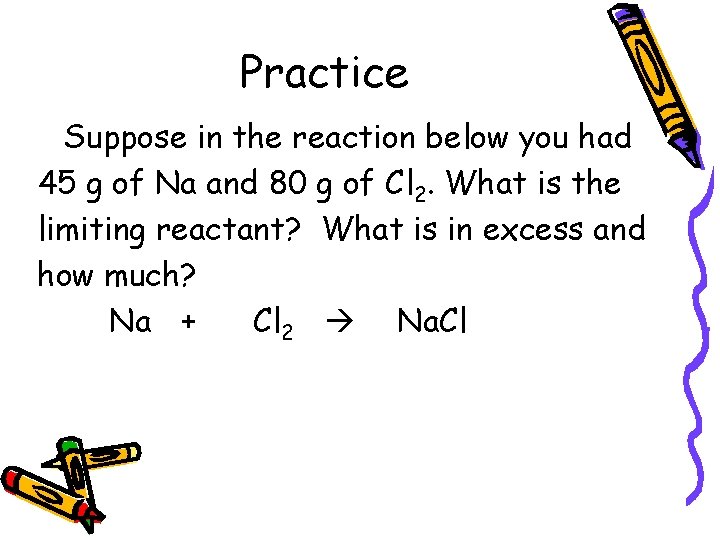
Practice Suppose in the reaction below you had 45 g of Na and 80 g of Cl 2. What is the limiting reactant? What is in excess and how much? Na + Cl 2 Na. Cl
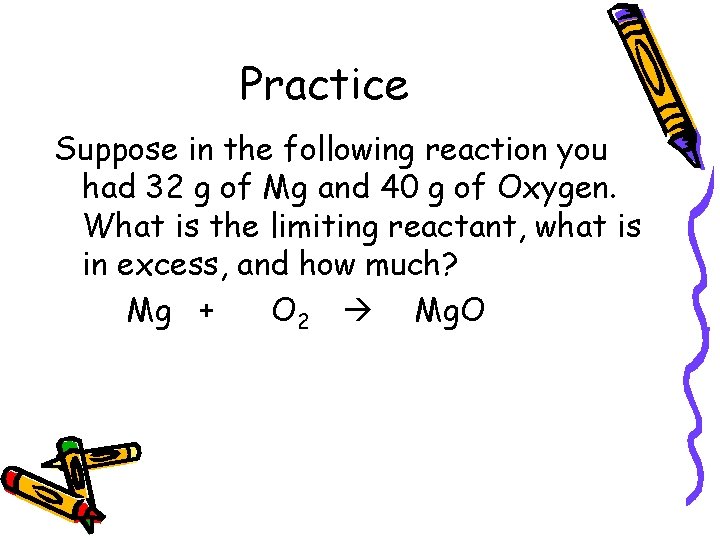
Practice Suppose in the following reaction you had 32 g of Mg and 40 g of Oxygen. What is the limiting reactant, what is in excess, and how much? Mg + O 2 Mg. O