stoichiometry Chemical Equations Balancing equations 1 The Eq

stoichiometry

Chemical Equations

Balancing equations 1. The Eq. must represent the known facts. 2. The Eq. must contain the correct formulas for reactants and products. 3. The law of conservation of mass must be followed. 4. Balance atoms that appear once 1 st. 5. Treat polyatomics as one unit if they appear on both sides the same way.

6. Balance other elements. 7. Balance H 8. Balance O last!! *NEVER CHANGE A SUBSCRIPT
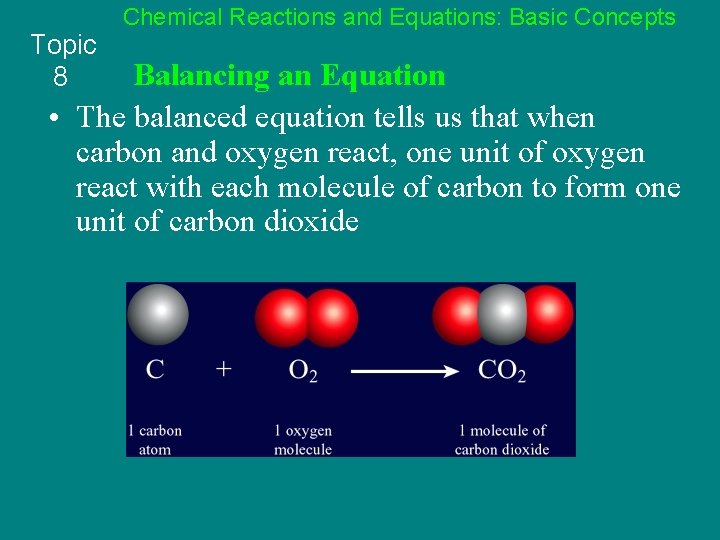
Topic 8 Chemical Reactions and Equations: Basic Concepts Balancing an Equation • The balanced equation tells us that when carbon and oxygen react, one unit of oxygen react with each molecule of carbon to form one unit of carbon dioxide

Conservation of Mass and Balancing Equations Click in this box to enter notes. Go to Slide Show View (press F 5) to play the video or animation. (To exit, press Esc. ) This media requires Power. Point® 2000 (or newer) and the Macromedia Flash Player (7 or higher). [To delete this message, click inside the box, click the border of the box, and then press delete. ] Copyright © Houghton Mifflin Company. All rights reserved.

Example • Write and balance the reaction between sodium hydroxide and calcium bromide to give calcium hydroxide and sodium bromide Na. OH + Ca. Br 2 Ca(OH)2 + Na. Br • 2 Na. OH + Ca. Br 2 Ca(OH)2 + 2 Na. Br


Here are some more to balance: a) b) c) d) e) f) g) h) i) 2 KNO 3 2 KNO 2 + O 2 2 Pb(NO 3)2 2 Pb. O + 4 NO 2 + O 2 P 4 + 6 I 2 4 PI 3 3 Mg. O + 2 H 3 PO 4 Mg 3(PO 4)2 + 3 H 2 O Br 2 + 2 KI I 2 + 2 KBr Ca(OH)2 + 2 HNO 3 Ca(NO 3)2 + 2 H 2 O Bi 2 O 3 + 3 H 2 2 Bi + 3 H 2 O 3 Fe + 2 O 2 Fe 3 O 4 2 Ca. O + 5 C 2 Ca. C 2 + CO 2

Classification of Reactions • There are 5 major classifications of reactions: – Synthesis A + B → AB – Decomposition AB → A + B – Combustion Hydrocarbon + O 2 → CO 2 + H 2 O – Single Replacement AB + C → AC + B – Double Replacement AB + CD → AD + CB


Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • In one type of reaction, two substances— either elements or compounds—combine to form a compound. • Whenever two or more substances combine to form a single product, the reaction is called a synthesis reaction.

Topic 8 Chemical Reactions and Equations: Basic Concepts A Synthesis Reaction • When iron rusts, iron metal and oxygen gas combine to form one new substance, iron(III) oxide. • The balanced equation for this synthesis reaction shows that there is more than one reactant but only one product.

Topic 8 Chemical Reactions and Equations: Basic Concepts A Synthesis Reaction

Synthesis (Combination) • Synthesis – when 2 or more substances react to produce 1 product • Of the form: A + B AB • Examples: – 2 Fe + 3 Cl 2 2 Fe. Cl 3 – 2 Na + Cl 2 2 Na. Cl – Ca. O + H 2 O Ca(OH)2 – 2 Mg + O 2 2 Mg. O

Synthesis # 1 1. Metal oxide + nonmetal oxide metal oxyanion a salt (Not Redox) • No Redox simply means that the oxidation numbers of the elements stays the same • Oxyanion = a polyatomic ion with oxygen

Synthesis # 1 Example • Sulfur dioxide gas is passed over solid calcium oxide • SO 2 + Ca. O • We know that we have to get a metal oxyanion. • So we either get Ca. SO 4 or Ca. SO 3 • We need to check the oxidation states on sulfur to see which one is the same.

Synthesis # 1 Example • • In SO 2, the oxidation number of O is -2 So the oxidation number of S must be +4 Our product choices are Ca. SO 3 or Ca. SO 4 In Ca. SO 3…S has an oxidation # of +4 In Ca. SO 4…S has an oxidation # of +6 Therefore the product must be Ca. SO 3 SO 2 + Ca. O Ca. SO 3

Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • In a decomposition reaction, a compound breaks down into two or more simpler substances. • The compound may break down into individual elements, such as when mercury(II) oxide decomposes into mercury and oxygen.

Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • The products may be an element and a compound, such as when hydrogen peroxide decomposes into water and oxygen. • The compound may break down into simpler compounds.

Topic 8 Chemical Reactions and Equations: Basic Concepts A Decomposition Reaction • When ammonium nitrate is heated to a high temperature, it explosively breaks down into dinitrogen monoxide and water. • The decomposition reaction taking place is represented by a balanced equation that shows one reactant and more than one product.

Topic 8 Chemical Reactions and Equations: Basic Concepts A Decomposition Reaction

Decomposition • Decomposition – when a single compound breaks down into 2 or more compounds • Of the form: AB A + B • Note that this is the exact opposite of synthesis • Examples: – 2 Na 3 N 6 Na + N 2 – NH 4 NO 3 N 2 O + 2 H 2 O – 2 H 2 O 2 H 2 + O 2

Decomposition Synthesis Decomposition Redox Metal + Nonmetal salt Salt Metal + Nonmetal Non Redox Metal oxide + H 2 O base Metal oxide + H 2 O Nonmetal oxide + H 2 O acid Nonmetal oxide + H 2 O Metal oxide + Nonmetal oxide salt Metal oxide + Nonmetal oxide

More Decomposition • • Metal chlorates metal chloride + O 2 (NH 4)2 CO 3 2 NH 3 + H 2 O + CO 2 2 H 2 O 2 2 H 2 O + O 2 If you get any of these products…they decompose… – – NH 4 OH NH 3 + H 2 O H 2 CO 3 CO 2 + H 2 O H 2 SO 3 SO 2 + H 2 O HNO 2 NO + NO 2 + H 2 O

Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • A combustion reaction is one in which a substance rapidly combines with oxygen to form one or more oxides. Click box to view movie clip.

Topic 8 Chemical Reactions and Equations: Basic Concepts Combustion • When welding is done with an acetylene torch, acetylene combines with oxygen to form carbon dioxide and water. • This combustion reaction is exothermic, and enough energy is released to melt metal.

Topic 8 Chemical Reactions and Equations: Basic Concepts Combustion

Combustion • Combustion – when O 2 combines with a hydrocarbon to form CO 2 and H 2 O • Of the form: hydrocarbon + O 2 CO 2 + H 2 O • For example: – CH 4 + 2 O 2 CO 2 + 2 H 2 O – 2 CH 3 OH + 3 O 2 2 CO 2 + 4 H 2 O

Combustion 1. • • • Hydrocarbon + O 2 CO 2+ H 2 O (No ions) Combustion of methane CH 4 + O 2 CO 2+ H 2 O CH 4 + 2 O 2 CO 2+ 2 H 2 O


When Balancing Combustion Reactions • Put a two in front of the hydrocarbon to start. • Balance them in the order C, H, O • Make sure your answer is in the lowest whole number ratio

Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • In a single-displacement reaction, one element takes the place of another in a compound. • The element can replace the first part of a compound, or it can replace the last part of a compound.

Topic 8 Chemical Reactions and Equations: Basic Concepts Single Displacement • If an iron nail is placed into an aqueous solution of copper(II) sulfate, the iron displaces the copper ions in solution, and copper metal forms on the nail.

Topic 8 Chemical Reactions and Equations: Basic Concepts Single Displacement

Single Replacement • Single Replacement – when the atoms of one element replace the atoms of another element in a compound • Of the form : A + BX B + AX • For example: – 2 Li + 2 H 2 O 2 Li. OH + H 2 – Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2

Activity Series Page 286 TABLE 3 in your text book

Single Replacement Reactions • In order for a single replacement reaction to occur, the element that is by itself must be strong enough to push the other element out of the way. • The activity series show you the relative pushing power of the elements. • In order for the reaction to occur, the lone element must be above the element in the compound • Otherwise…NO REACTION NR

Single Replacement Reactions • You need to know which chart you are to look at…metals or halogens. • A metal can replace another metal • A halogen can replace another halogen

Will these reactions occur? • Will the following reaction occur? If so, complete and balance the reaction. • Ag + Cu(NO 3)2 NR • No reaction because Ag is below Cu in the Activity series of metals

Will these reactions occur? • Will the following reaction occur? If so, complete and balance the reaction. • Mg + Al. Cl 3 • 3 Mg + 2 Al. Cl 3 3 Mg. Cl 2 + 2 Al • The reaction WILL occur because Mg is above Al in the activity series

Will these reactions occur? • Will the following reaction occur? If so, complete and balance the reaction. • Br 2 + Mg. Cl 2 NR • This reaction will NOT occur because Br is not above Cl in the activity series of halogens

Double Replacement • Double Replacement – a reaction involving the exchange of ions between 2 compounds • Of the form: AX + BY BX + AY

Topic 8 Chemical Reactions and Equations: Basic Concepts Major Classes of Reactions • In double-displacement reactions, the positive portions of two ionic compounds are interchanged. • For a double-displacement reaction to take place, at least one of the products must be a precipitate or water.

Topic 8 Chemical Reactions and Equations: Basic Concepts Double Displacement • When clear aqueous solutions of lead(II) nitrate and potassium iodine are mixed, a double-displacement reaction takes place and a yellow solid appears in the mixture. • This solid is lead(II) iodine, and it precipitates out because it is insoluble in water, unlike the two reactants and the other product.

Topic 8 Chemical Reactions and Equations: Basic Concepts Double Displacement

Double Replacement (Metathesis) • Double Replacement – a reaction involving the exchange of ions between 2 compounds • Of the form: AX + BY BX + AY • Examples: – Pb(NO 3)2 + 2 KI Pb. I 2 + 2 KNO 3 – Na 2 CO 3 + 2 Ag. NO 3 2 Na. NO 3 + Ag 2 CO 3

Double Replacement (Metathesis) • In order for a double replacement reaction to take place, one of 3 things must be formed: • Precipitate (solid) • Gas • Weak electrolyte – usually water
Precipitation Reactions Click in this box to enter notes. Go to Slide Show View (press F 5) to play the video or animation. (To exit, press Esc. ) This media requires Power. Point® 2000 (or newer) and the Macromedia Flash Player (7 or higher). [To delete this message, click inside the box, click the border of the box, and then press delete. ] Copyright © Houghton Mifflin Company. All rights reserved.

Solubility Rules!!!

Double Replacement # 2 (Formation of a gas) 2. • • • Formation of a gas (acid + sulfide, sulfite, carbonate, or bicarbonate…or ammonium salt + a strong base NH 3(g), H 2 O, and a salt) Hydrobromic acid is added to a solution of potassium bicarbonate HBr + KHCO 3 H 2 CO 3 + KBr H 2 CO 3 ALWAYS breaks down into CO 2 + H 2 O HBr + KHCO 3 CO 2 + H 2 O + KBr H+ + HCO 3 - CO 2 + H 2 O

Double Replacement #4 (Acid Base neutralization) • Acid + base salt + water • Hydrogen sulfide gas is bubbled through excess potassium hydroxide solution • H 2 S + 2 KOH 2 HOH + K 2 S • H 2 S + 2 K+ + 2 OH- 2 H 2 O + 2 K+ + S-2 • H 2 S + 2 OH- 2 H 2 O + S-2
Proton Transfer Click in this box to enter notes. Go to Slide Show View (press F 5) to play the video or animation. (To exit, press Esc. ) This media requires Power. Point® 2000 (or newer) and the Macromedia Flash Player (7 or higher). [To delete this message, click inside the box, click the border of the box, and then press delete. ] Copyright © Houghton Mifflin Company. All rights reserved.

Ionic Equations

Equations • Molecular equations – show the complete chemical formulas. Does not indicate ionic character • Complete ionic equation – shows all ions. Actually how the particles exist in the solution

Steps for Writing Ionic Equations 1. Write the balances molecular equation (balanced chemical equation) 2. Break every thing down into its ions EXCEPT the solid, gas, or water (complete ionic equation) 3. Cross out everything that is the same on both sides (spectator ions) 4. Write what is left (net ionic equation)

Rules • When writing ionic equations, you must keep together the solid, gas, or water • Spectator ions – ions that appear on both sides of the equation. They have very little to do with the chemical reaction

Example • Write the balanced chemical equation, the complete ionic equation, and the net ionic equation for the reaction between lead (II) nitrate and potassium iodide. Solid potassium iodide will be formed.

Example • • • Write the balanced chemical equation Pb(NO 3)2 + 2 KI Pb. I 2 + 2 KNO 3 You MUST identify the solid, gas, or water Pb(NO 3)2 + 2 KI Pb. I 2 (s) + 2 KNO 3 Balanced chemical equation

Example • Now break every thing except the solid, gas, or water into its ions • Remember ions are things with charges • Everything will be broken down into one positive charge and one negative charge

Example • Pb(NO 3)2 + 2 KI Pb. I 2 (s) + 2 KNO 3 • Pb+2 + 2 NO 3 -1 + 2 K+1 + 2 I -1 Pb. I 2 (s) + 2 K+1 + 2 NO 3 -1 • Complete ionic Equation

Example • Now cross out everything that is the same on both sides (spectator ions) • Pb+2 + 2 NO 3 -1 + 2 K+1 + 2 I -1 Pb. I 2 (s) + 2 K+1 + 2 NO 3 -1 • Now write what is left • Pb+2 + 2 I -1 Pb. I 2 (s) • Net ionic equation

Another Example • Write the balanced chemical equation, complete ionic equation, and net ionic equation for the reaction between calcium chloride and sodium carbonate. Solid calcium carbonate will be formed.

Another Example • Balanced chemical equation • Ca. Cl 2 + Na 2 CO 3 Ca. CO 3 (s) + 2 Na. Cl • Complete ionic equation • Ca+2 + 2 Cl -2 + 2 Na +1 + CO 3 -2 Ca. CO 3 (s) + 2 Na +1 + 2 Cl -1 • Net Ionic Equation • Ca+2 + 2 Cl -2 + 2 Na +1 + CO 3 -2 Ca. CO 3 (s) + 2 Na +1 + 2 Cl -1 • Ca+2 + CO 3 -2 Ca. CO 3 (s)

What if water is formed? • Write the balanced chemical equation, complete ionic equation, and net ionic equation for the reaction between Calcium hydroxide and nitric acid

Example with water • Balanced chemical equation • Ca(OH)2 + 2 HNO 3 Ca(NO 3)2 + 2 HOH • Complete ionic equation • Ca+2 + 2(OH) -1 + 2 H+1 + 2 NO 3 -1 Ca+2 + 2 NO 3 -1 + 2 HOH • Net Ionic Equation • Ca+2 + 2(OH) -1 + 2 H+1 + 2 NO 3 -1 Ca+2 + 2 NO 3 -1 + 2 HOH • 2(OH) -1 + 2 H+1 2 HOH

Stoichiometry • Using the methods of stoichiometry, we can measure the amounts of substances involved in chemical reactions and relate them to one another.

Stoichiometry Rules • 1. Balance the equation. • 2. convert all units to moles • 3. use a molar ratio from the balanced equation to compare what is given to what you want. • 4. convert back from moles to the unit the question asks for.

• Always no matter what unit you are given convert it to moles before you make a comparison.

Coefficients • Coefficients in a balanced chemical RXN can represent any of the following • 1. moles • 2. molecules of a compound • 3. volume of a gas at STP. (22. 7 L=1 mol) • * they never represent mass.

Mole – Mole Relationship • 4 Fe + 3 O 2 2 Fe 2 O 3 • 4 mol Fe / 3 mol O 2 • 4 mol Fe / 2 mol Fe 2 O 3 • 3 mol O 2 / 2 mol Fe 2 O 3

Mole – Mole Relationship • How many moles of Fe 2 O 3 will I form from 5. 0 mol of Fe? • 5. 0 mole Fe x 2 mol Fe 2 O 3 4 mol Fe • 2. 5 mol Fe 2 O 3

Conversion Factor # 3 # Moles A # Moles B The #’s in from if A & B MUST come from the balanced chemical equation

Mass – Mole Relationship • How many g of Na. Cl will be produced from 1. 25 mol of chlorine gas reacting with sodium? • Write the reaction • Na + Cl 2 Na. Cl • Balance the reaction • 2 Na + Cl 2 2 Na. Cl

Mass – Mole Relationship • Work the problem • 1. 25 mol Cl 2 x 2 mol Na. Cl x 58. 44 g Na. Cl • 1 mol Cl 2 1 mol Na. Cl • 146 g Na. Cl

Mass – Mass Relationships • Ammonium nitrate decomposes into dinitrogen monoxide gas and water. Determine that amount of water produced if 25. 0 g of ammonium nitrate decomposes. • NH 4 NO 3 N 2 O + H 2 O • NH 4 NO 3 N 2 O + 2 H 2 O

Mass – Mass Relationships • 25. 0 g NH 4 NO 3 x 1 mol NH 4 NO 3 x 2 mol H 2 O x 18. 02 g H 2 O 80. 04 g NH 4 NO 3 1 mol H 2 O • 11. 2 g H 2 O
- Slides: 77