Stoichiometry Chapter 3 Avogadros Number n n NA

Stoichiometry Chapter 3

Avogadro’s Number n n NA 6. 022 X 1023 H atoms = 1. 008 g H 6. 022 X 1023 O atoms = 16. 00 g O

The mole n n 1 mole = 6. 022 X 1023 atoms 1 mole H = 6. 022 X 1023 H atoms = 1. 008 g H 1 mole Cl 2 = 6. 022 X 1023 Cl 2 molecules = 70. 09 g Cl 2 1 mole e- = 6. 022 X 1023 e-

Molar mass n n n MM, GMM, Is numerically equal to the sum of the masses of the atoms in the formula Ca. Cl 2 =110. 98 g/mol

Mass-mole conversions Calculate the mass in grams of 13. 2 mol Ca. Cl 2

Mass-mole conversions Calculate the mass in grams of 13. 2 mol Ca. Cl 2

Calculate the number of moles in 16. 4 g C 6 H 12 O 6

Calculate the number of moles in 16. 4 g C 6 H 12 O 6
![Molarity n Concentration of a solute in a solution [] n M= n Molarity n Concentration of a solute in a solution [] n M= n](http://slidetodoc.com/presentation_image/d50d0b7a4a57ccf2b373797c719a142b/image-9.jpg)
Molarity n Concentration of a solute in a solution [] n M= n

What volume of 12 M HCl must be taken to obtain 0. 10 mol HCl?
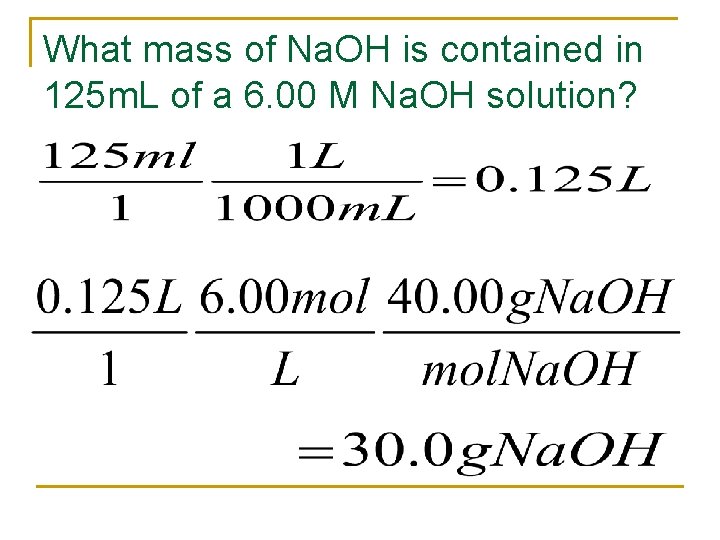
What mass of Na. OH is contained in 125 m. L of a 6. 00 M Na. OH solution?

What are the molarities of Al 3+ and SO 42 - in 0. 100 M Al 2(SO 4)3 ?
What is the percent composition of K 2 Cr. O 4 ?

What is the percent composition of K 2 Cr. O 4 ? MM = 2(39. 10) + 52. 00 + 4(16. 00) = 194. 20 g/mol
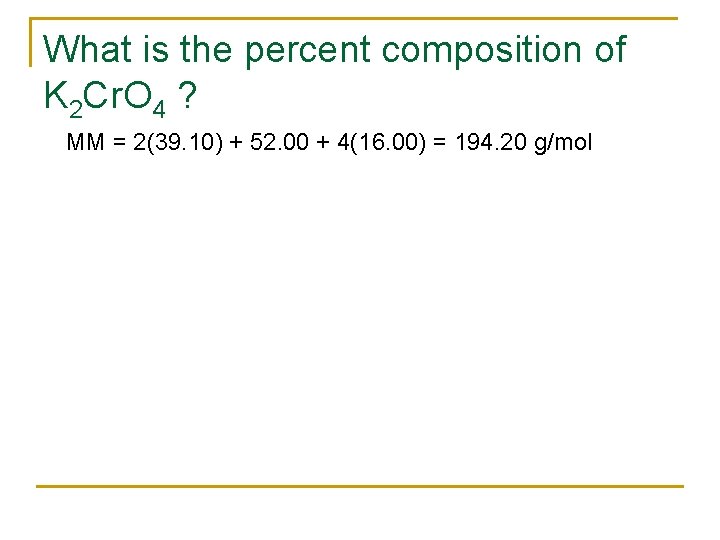
What is the percent composition of K 2 Cr. O 4 ? MM = 2(39. 10) + 52. 00 + 4(16. 00) = 194. 20 g/mol

Empirical formula-simplest n n n Assume a 100 g sample unless otherwise stated Convert grams to moles Find the mole ratio

Find the empirical formula of a compound containing 26. 6 % K, 35. 4 % Cr, and 38. 0 % O



A sample of acetic acid weighing 1. 000 g burns to give 1. 466 g CO 2 and 0. 6001 g H 2 O. Find the empirical formula.

A sample of acetic acid weighing 1. 000 g burns to give 1. 466 g CO 2 and 0. 6001 g H 2 O. Find the empirical formula. CHO + O CO + HO x y z 1. 000 g 2 2 1. 466 g 2 0. 6001 g Find the mass of C from CO 2 Find the mass of H from H 2 O Find the mass of O by difference




Molecular formulas n n Must be told the molar mass MM/FM

Find the molecular formula of acetic acid. MM = 60 g/mol

Chemical Equations n n Balancing The coefficients of a balanced equation represent numbers of moles of reactant and products
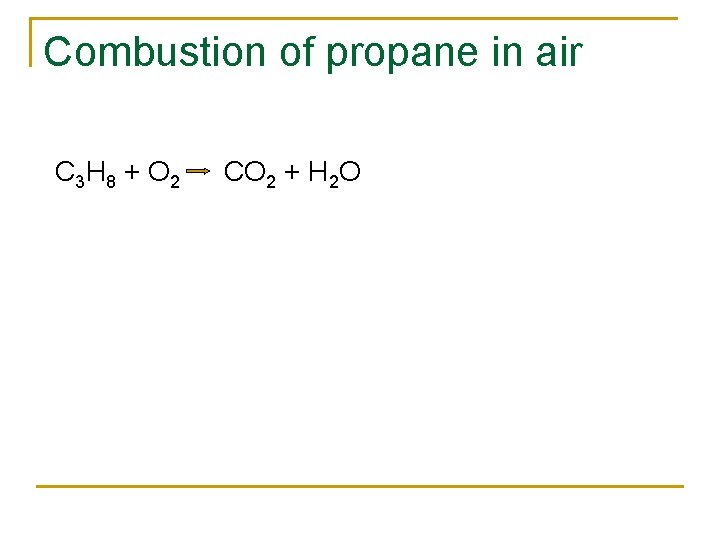
Combustion of propane in air C 3 H 8 + O 2 CO 2 + H 2 O
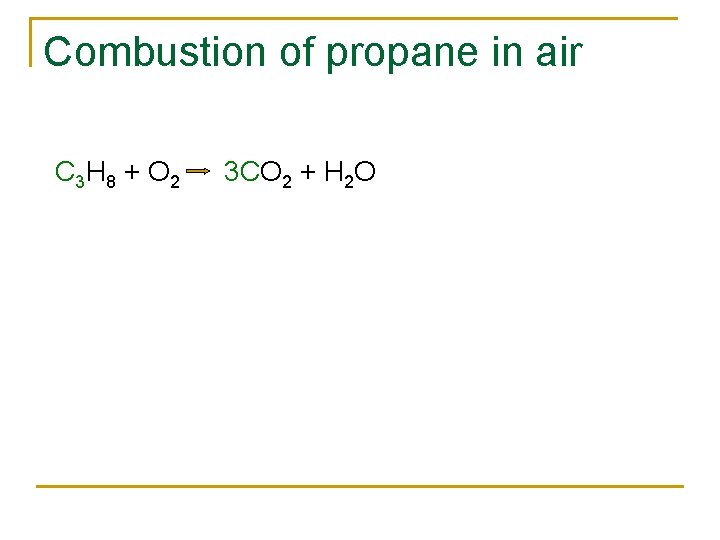
Combustion of propane in air C 3 H 8 + O 2 3 CO 2 + H 2 O

Combustion of propane in air C 3 H 8 + O 2 3 CO 2 + 4 H 2 O

Combustion of propane in air C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O

Combustion of propane in air C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (l)

How many moles of CO 2 are produced when 1. 65 mol C 3 H 8 burns?

How many moles of CO 2 are produced when 1. 65 mol C 3 H 8 burns?
What mass of O 2 is required to react with 12. 0 g C 3 H 8
What mass of O 2 is required to react with 12. 0 g C 3 H 8

Limiting reactant n Determines the amount of product formed q q It is completely used up Is used to determine theoretical yield n The other reactant is in excess q There will be some left over

Calculate theoretical yield of Ag. I and determine the limiting reactant of a reaction between 1. 00 g Ag and 1. 00 g I 2

Calculate theoretical yield of Ag. I and determine the limiting reactant of a reaction between 1. 00 g Ag and 1. 00 g I 2 2 Ag(s) + I 2(s) 2 Ag. I(s)

Calculate theoretical yield of Ag. I and determine the limiting reactant of a reaction between 1. 00 g Ag and 1. 00 g I 2 2 Ag(s) + I 2(s) 2 Ag. I(s)

n n n 1. 85 g Ag. I is theoretical yield I 2 is the limiting reactant Ag is in excess
How much Ag is in excess?
What is the percent yield if the actual yield was 1. 50 g Ag. I?
- Slides: 43