Stoichiometry Chapter 11 REVIEW Chemical ReactionsEquations Reactant Reactant










































- Slides: 51

Stoichiometry Chapter 11

REVIEW Chemical Reactions/Equations Reactant + Reactant → Product + Product Balanced chemical equations are written to show chemical changes taking place Balanced chemical equations reflect the Law of Conservation of Mass Coefficients balance chemical equations and indicate the relative amounts of substances

REVIEW Conversion Factors 1. Avogadro’s number and moles 1 mol = 6. 02 x 1023 representative particles Representative particles: Atoms – element from periodic table Ions – charged atom (due to loss/gain of e–) Formula units – ionic compound Molecules – covalent compound 2. Molar mass 3. 1 mol = g of substance where = (atomic mass × subscript) Mole ratio of element to compound mol element = 1 mol compound where = subscript of element in compound

PREVIEW Unit Objectives Interpret chemical equations in terms of particles, moles, and mass Write mole ratios from balanced equations Calculate number of moles and mass of a reactant or product when given number of moles or mass of another reactant or product Identify limiting reactants in chemical reactions Determine percent yield of chemical reactions

U 8 -5 Stoichiometry Defined: The study of quantitative relationships between amounts of reactants used and products formed by a chemical reaction Involves calculating quantities of reactants or products in a reaction using relationships found in balanced chemical equation Based on the Law of Conservation of Mass: Mass is neither created nor destroyed in any process; it is conserved

U 8 -5 Interpreting Equations Write the balanced chemical equation for the synthesis of ammonia from nitrogen and hydrogen gases. N 2 Interpret + 3 H 2 → 2 NH 3 the equation in terms of particles, moles, and mass. Then, show the Law of Conservation of Mass is obeyed.

U 8 -5 Interpreting Equations Interpret the equation in terms of particles. N 2 + 3 H 2 → 2 NH 3 Number of particles for each substance is indicated by the coefficients Four particle types: atoms, ions, formula units, or molecules 1 molecule N 2 + 3 molecules H 2 → 2 molecules NH 3

U 8 -5 Interpreting Equations Interpret the equation in terms of moles. N 2 + 3 H 2 → 2 NH 3 Number of moles for each substance is indicated by the coefficients 1 mole N 2 + 3 moles H 2 → 2 moles NH 3

U 8 -5 Interpreting Equations Interpret the equation in terms of mass. N 2 + 3 H 2 → 2 NH 3 Calculate the molar mass for each reactant and product N 2: 2 mol x 14. 0 g/mol 28. 0 g/mol N 2 H 2: 2 mol x 1. 0 g/mol 6. 0 g/mol H 2 NH 3: 1 mol x 14. 0 g/mol = 14. 0 g 3 mol x 1. 0 g/mol = 3. 0 g 17. 0 g/mol NH 3

U 8 -5 Interpreting Equations Interpret the equation in terms of mass. N 2 + 3 H 2 → 2 NH 3 28. 0 g/mol 2. 0 g/mol 17. 0 g/mol Multiply number of moles of each reactant and product by molar mass g N 2 = 1 mol N 2 28. 0 g N 2 1 mol N 2 = 28. 0 g N 2

U 8 -5 Interpreting Equations Interpret the equation in terms of mass. N 2 + 3 H 2 → 2 NH 3 28. 0 g/mol 2. 0 g/mol 17. 0 g/mol Multiply number of moles of each reactant and product by molar mass g H 2 = 3 mol H 2 2. 0 g H 2 1 mol H 2 = 6. 0 g H 2

U 8 -5 Interpreting Equations Interpret the equation in terms of mass. N 2 + 3 H 2 → 2 NH 3 28. 0 g/mol 2. 0 g/mol 17. 0 g/mol Multiply number of moles of each reactant and product by molar mass g NH 3 = 2 mol NH 3 17. 0 g NH 3 1 mol NH 3 = 34. 0 g NH 3

U 8 -5 Interpreting Equations Interpret the equation in terms of mass. N 2 + 3 H 2 → 2 NH 3 28. 0 g 6. 0 g 34. 0 g 28. 0 g N 2 + 6. 0 g H 2 → 34. 0 g NH 3

U 8 -5 Interpreting Equations Show that the Law of Conservation of Mass is observed. N 2 + 3 H 2 → 2 NH 3 28. 0 g Add 6. 0 g 34. 0 g the masses of the reactants. 28. 0 g N 2 + 6. 0 g H 2 = 34. 0 g reactants Add the masses of the products. 34. 0 g NH 3 = 34. 0 g products When the mass of the reactants equals the mass of the products, the Law of Conservation of Mass is observed. 34. 0 g reactants = 34. 0 g products

HW due 01/28 Textbook Practice, p. 371 #1, #2

U 8 -5 Mole Ratio between the numbers of moles of any two substances in a balanced chemical equation

U 8 -5 Mole Ratios Determine all possible mole ratios for the balanced equation showing the synthesis of ammonia. N 2 + 3 H 2 → 2 NH 3 How many mole ratios are possible? This reaction has three participating species. Multiply the number of species present by the next lower number. 3 x 2 = 6 mole ratios For the synthesis of ammonia, six mole ratios are possible.

U 8 -5 Mole Ratios Determine all possible mole ratios for the balanced equation showing the synthesis of ammonia. N 2 + 3 H 2 → 2 NH 3 1 mol N 2 3 mol H 2 2 mol NH 3 1 mol N 2 2 mol NH 3 3 mol H 2

U 8 -5 Mole Ratios Which mole ratio should be used? The needed mole ratio is the one involving the UNKNOWN and the GIVEN. UNKNOWN GIVEN

HW due 01/29 Textbook Practice, p. 372 #3

U 8 -5 Stoichiometric Calculations Write the balanced chemical equation for the reaction. Identify the UNKNOWN and the GIVEN; draw the bridge/grid. The GIVEN must be in moles or converted to moles. Identify the conversion factor that will cancel the unit of the GIVEN. Set up conversion factors and cancel units until the only unit left standing matches the UNKNOWN. Do the math* and express the answer to the correct number of significant figures.

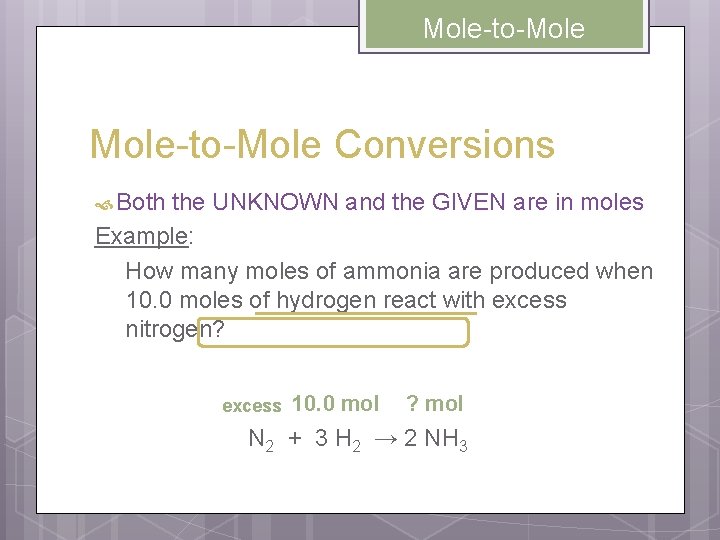
Mole-to-Mole Conversions Both the UNKNOWN and the GIVEN are in moles Example: How many moles of ammonia are produced when 10. 0 moles of hydrogen react with excess nitrogen? excess 10. 0 mol ? mol N 2 + 3 H 2 → 2 NH 3

Mole-to-Mole 10. 0 mol ? mol N 2 + 3 H 2 → 2 NH 3 UNKNOWN mol NH 3 = = GIVEN 10. 0 mol H 2 g→mol (G) Mole Ratio 2 mol NH 3 3 mol H 2 6. 67 mol NH 3 mol→g (U)

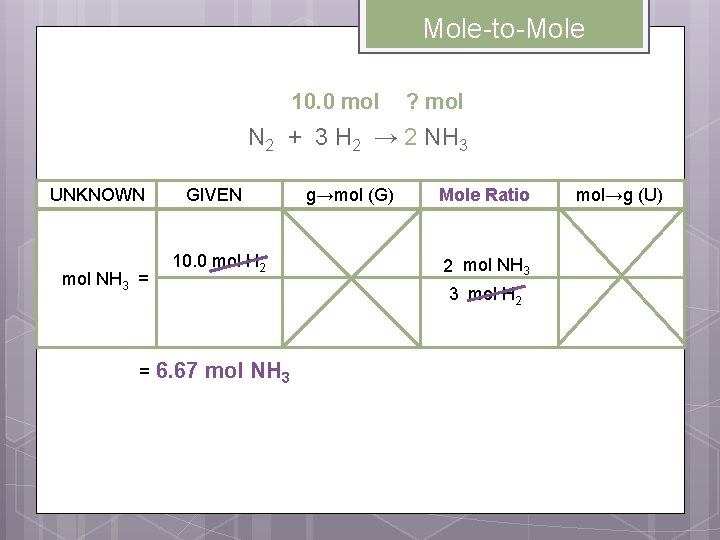
Mole-to-Mole Conversions Practice 1 How many moles of zinc chloride will be formed when 17. 0 moles of hydrochloric acid react with excess zinc metal? 17. 0 mol 2 HCl + ? mol excess Zn → Zn. Cl 2 + H 2 How many mole ratios are possible for this equation? 12

Mole-to-Mole Conversions Practice 1 2 HCl + Zn → Zn. Cl 2 + H 2 Write all possible mole ratios for the balanced equation. 1 mol Zn. Cl 2 1 mol H 2 2 mol HCl 1 mol Zn. Cl 2 1 mol Zn 1 mol H 2 1 mol Zn. Cl 2 2 mol HCl 1 mol H 2 1 mol Zn 2 mol HCl 1 mol Zn. Cl 2 1 mol Zn 1 mol H 2 1 mol Zn. Cl 2 2 mol HCl 1 mol Zn

Mole-to-Mole ? mol 17. 0 mol 2 HCl + Zn → Zn. Cl 2 + H 2 UNKNOWN mol Zn. Cl 2 = = GIVEN g→mol (G) 17. 0 mol HCl Mole Ratio 1 mol Zn. Cl 2 2 mol HCl 8. 50 mol Zn. Cl 2 mol→g (U)

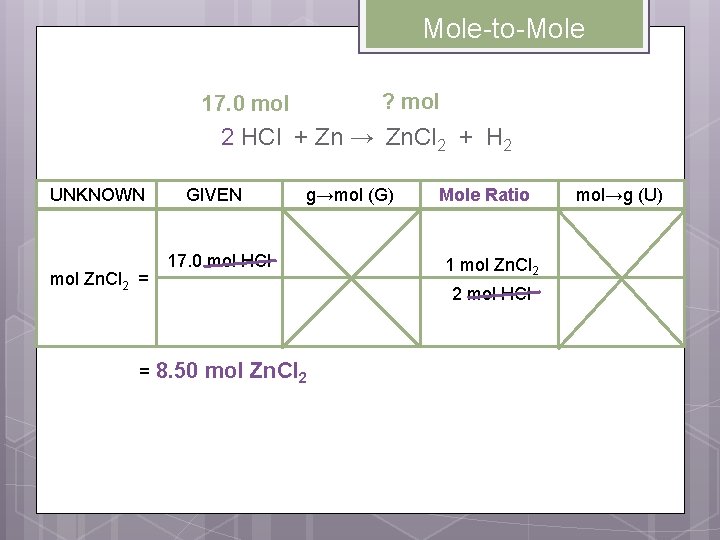
Mole-to-Mole Conversions Practice 2 Potassium chlorate decomposes into potassium chloride and oxygen. How many moles of oxygen are formed when 3. 20 moles KCl. O 3 decompose? 3. 20 mol 2 KCl. O 3 → ? mol 2 KCl + 3 O 2 Write the mole ratios involving the UNKNOWN and the GIVEN in the problem. 3 mol O 2 2 mol KCl. O 3 3 mol O 2

Mole-to-Mole ? mol 3. 20 mol 2 KCl. O 3 → 2 KCl + 3 O 2 UNKNOWN mol O 2 = = GIVEN 3. 20 mol KCl. O 3 g→mol (G) Mole Ratio 3 mol O 2 2 mol KCl. O 3 4. 80 mol O 2 mol→g (U)

HW due 02/04 Textbook Practice, p. 375 #11

Mole-to-Mass Conversions p. 4 of Stoichiometry for Students Notes Packet

Mole-to-Mass Conversions GIVEN in moles and UNKNOWN in grams Example: Balance the following equation for the combustion of propane. ____ C 3 H 8 + 5____ O 2 → 3____ CO 2 + 4____ H 2 O 44. 0 g/mol 32. 0 g/mol 18. 0 g/mol Calculate the molar mass for each substance.

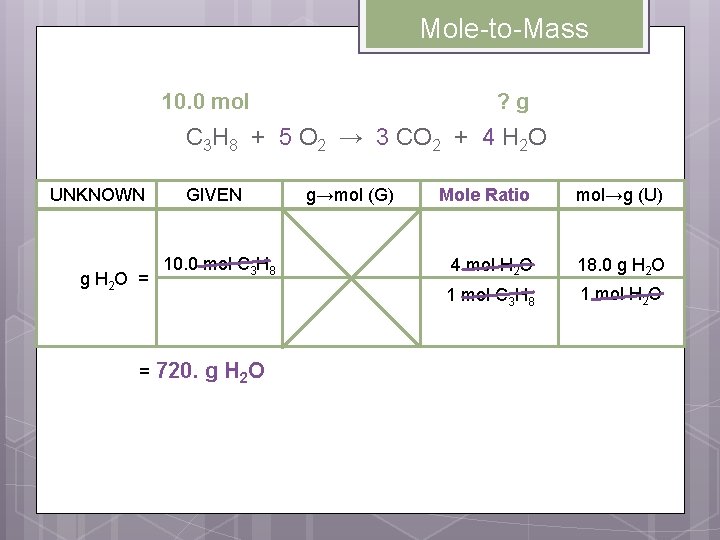
Mole-to-Mass Conversions Example: If 10. 0 moles of propane are used, how many grams of water are formed? 10. 0 mol ? g C 3 H 8 + 5 O 2 → 3 CO 2 + 4 H 2 O Write the mole ratios involving the UNKNOWN and the GIVEN. 1 mol C 3 H 8 4 mol H 2 O 1 mol C 3 H 8

Mole-to-Mass 10. 0 mol ? g C 3 H 8 + 5 O 2 → 3 CO 2 + 4 H 2 O UNKNOWN g H 2 O = = GIVEN 10. 0 mol C 3 H 8 720. g H 2 O g→mol (G) Mole Ratio mol→g (U) 4 mol H 2 O 18. 0 g H 2 O 1 mol C 3 H 8 1 mol H 2 O

Mole-to-Mass Conversions Practice 1: Sulfuric acid is produced when sulfur dioxide reacts with oxygen and water. Balance the equation for the synthesis reaction. 2____ SO 2 + ____ O 2 +2 ____ H 2 O → 2 ____ H 2 SO 18. 0 64. 1 g/mol 32. 0 g/mol 4 98. 1 g/mol Calculate the molar mass for each substance.

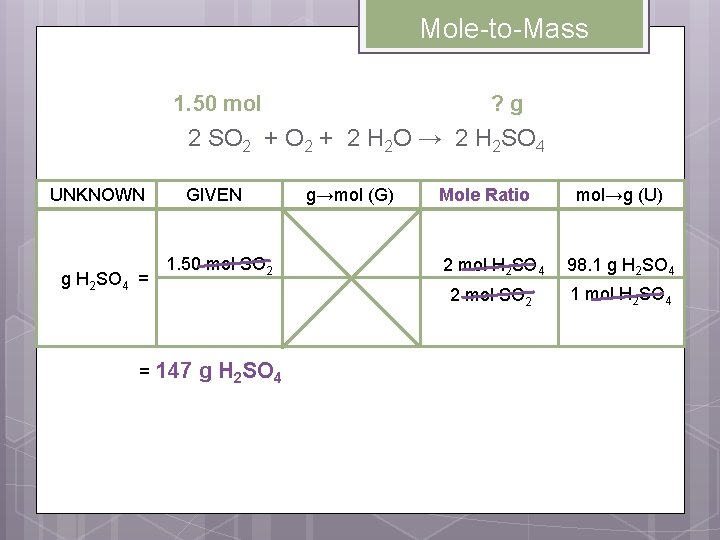
Mole-to-Mass Conversions Practice 1: How many grams of sulfuric acid are produced when 1. 50 moles of sulfur dioxide completely reacts with oxygen and water ? 1. 50 mol ? g 2 SO 2 + 2 H 2 O → 2 H 2 SO 4 Write the mole ratios involving the UNKNOWN and the GIVEN. 2 mol H 2 SO 4 2 mol SO 2 2 mol H 2 SO 4

Mole-to-Mass 1. 50 mol ? g 2 SO 2 + 2 H 2 O → 2 H 2 SO 4 UNKNOWN g H 2 SO 4 = = GIVEN 1. 50 mol SO 2 147 g H 2 SO 4 g→mol (G) Mole Ratio mol→g (U) 2 mol H 2 SO 4 98. 1 g H 2 SO 4 2 mol SO 2 1 mol H 2 SO 4

Mass-to-Mole Conversions GIVEN in grams and UNKNOWN in moles Example: Methane and sulfur produce carbon disulfide and hydrogen sulfide gas, as indicated by the following chemical equation. 2 CS 2 + ____ 4 H 2 S 2 CH 4 + ____ S 8 → ____ 16. 0 g/mol 256. 8 g/mol 76. 2 g/mol 34. 1 g/mol Calculate the molar mass for each substance.

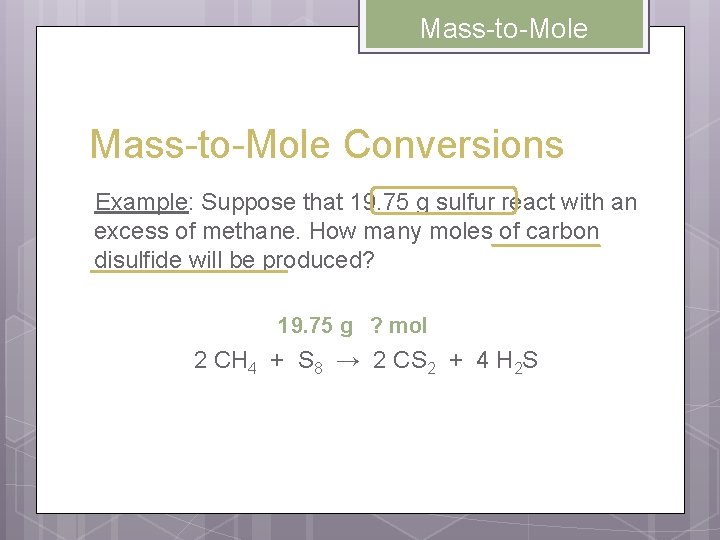
Mass-to-Mole Conversions Example: Suppose that 19. 75 g sulfur react with an excess of methane. How many moles of carbon disulfide will be produced? 19. 75 g ? mol 2 CH 4 + S 8 → 2 CS 2 + 4 H 2 S

Mass-to-Mole 19. 75 g ? mol 2 CH 4 + S 8 → 2 CS 2 + 4 H 2 S UNKNOWN mol CS 2 = = GIVEN g→mol (G) 19. 75 g S 8 1 mol S 8 2 mol CS 2 256. 8 g S 8 1 mol S 8 0. 1538 mol CS 2 Mole Ratio mol→g (U)

Mass-to-Mole Conversions Practice 1: Sodium fluoride is formed when sodium metal reacts with fluorine gas. Calculate the molar mass for each substance. 2 Na. F 2 Na + ____ F 2 → ____ 23. 0 g/mol 38. 0 g/mol Balance the equation. 42. 0 g/mol

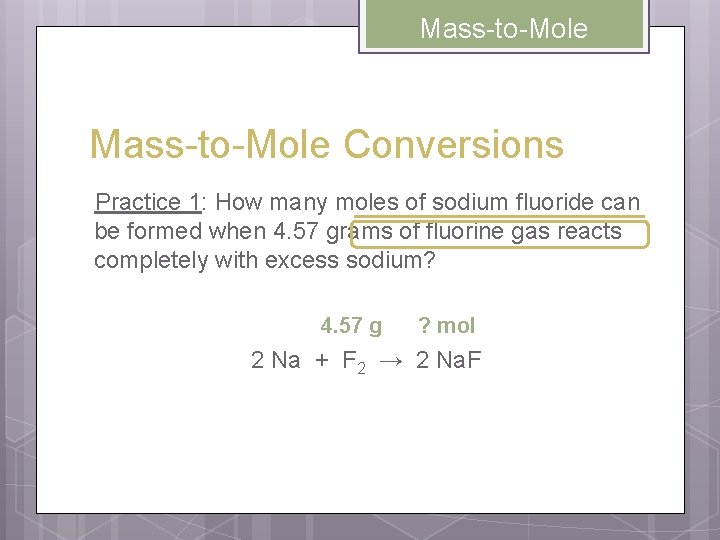
Mass-to-Mole Conversions Practice 1: How many moles of sodium fluoride can be formed when 4. 57 grams of fluorine gas reacts completely with excess sodium? 4. 57 g ? mol 2 Na + F 2 → 2 Na. F

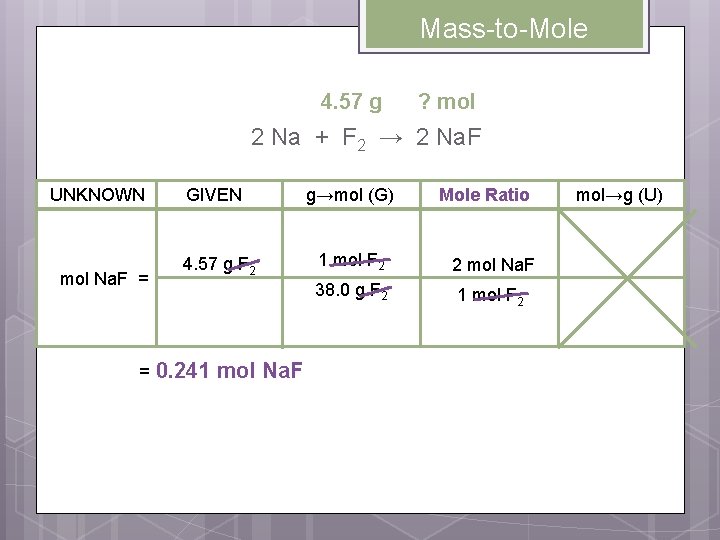
Mass-to-Mole 4. 57 g ? mol 2 Na + F 2 → 2 Na. F UNKNOWN mol Na. F = = GIVEN g→mol (G) 4. 57 g F 2 1 mol F 2 2 mol Na. F 38. 0 g F 2 1 mol F 2 0. 241 mol Na. F Mole Ratio mol→g (U)

HW due 02/05 Textbook Practice, p. 376 #14

Mass-to-Mass Conversions GIVEN in grams and UNKNOWN in grams Example: The following equation shows the combustion of butane. Calculate the molar mass for each substance. Balance the equation. 2____ C 4 H 10 +13 ____ O 2 → 8____ CO 2 +10____ H 2 O 44. 0 g/mol 58. 0 g/mol 32. 0 g/mol 18. 0 g/mol

Mass-to-Mass Conversions Example: If 75. 5 grams of carbon dioxide are produced, how many grams of butane were used? ? g 75. 5 g 2 C 4 H 10 + 13 O 2 → 8 CO 2 + 10 H 2 O

Mass-to-Mass 75. 5 g ? g 2 C 4 H 10 + 13 O 2 → 8 CO 2 + 10 H 2 O UNKNOWN g C 4 H 10 = = GIVEN g→mol (G) Mole Ratio mol→g (U) 75. 5 g CO 2 1 mol CO 2 2 mol C 4 H 10 58. 0 g C 4 H 10 44. 0 g CO 2 8 mol CO 2 1 mol C 4 H 10 24. 9 g C 4 H 10

Mass-to-Mass Conversions Practice 1: Use the balanced equation for the combustion of butane. How many grams of oxygen are necessary to react completely with 217 grams of butane? 217 g ? g 2 C 4 H 10 + 13 O 2 → 8 CO 2 + 10 H 2 O

Mass-to-Mass 217 g ? g 2 C 4 H 10 + 13 O 2 → 8 CO 2 + 10 H 2 O UNKNOWN g O 2 = = GIVEN g→mol (G) Mole Ratio mol→g (U) 217 g C 4 H 10 1 mol C 4 H 10 13 mol O 2 32. 0 g O 2 58. 0 g C 4 H 10 2 mol C 4 H 10 1 mol O 2 778 g O 2

Mass-to-Mass Conversions Practice 2: Balance the following single replacement reaction. ? g 75. 9 g 2 Fe + ____ 3 H 2 O → ____ Fe 2 O 3 + ____ 3 H 2 ____ How many grams of iron must react in order to produce 75. 9 grams of iron (III) oxide? Because grams are involved for both UNKNOWN and GIVEN, the molar mass for both must be calculated. Molar mass: 55. 8 g/mol UNKNOWN Molar mass: 159. 6 g/mol GIVEN

Mass-to-Mass ? g 75. 9 g 2 Fe + 3 H 2 O → Fe 2 O 3 + 3 H 2 UNKNOWN g Fe = = GIVEN g→mol (G) 75. 9 g Fe 2 O 3 53. 1 g Fe Mole Ratio mol→g (U) 1 mol Fe 2 O 3 2 mol Fe 55. 8 g Fe 159. 6 g Fe 2 O 3 1 mol Fe

HW due 02/06 Textbook Practice, p. 378 #21