Stoichiometry Calculating Reactants and Products Stoichiometry Defined The







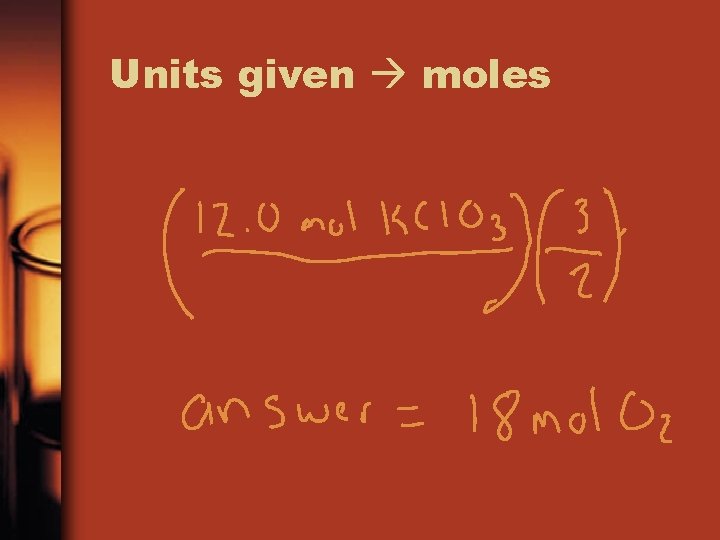


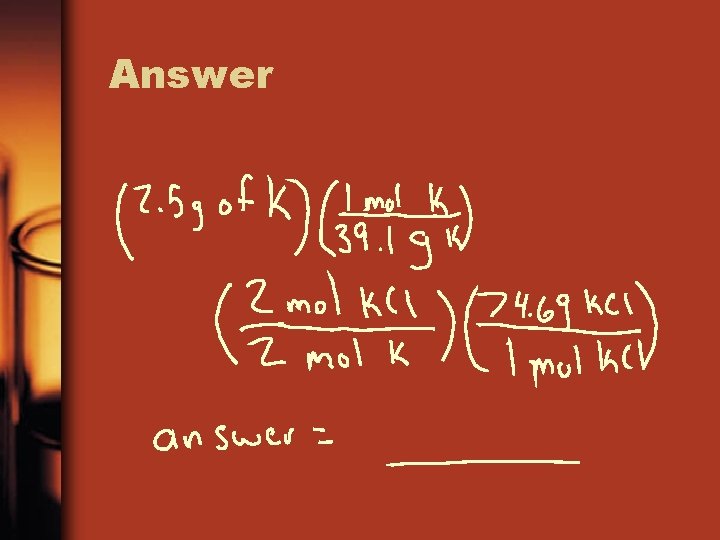
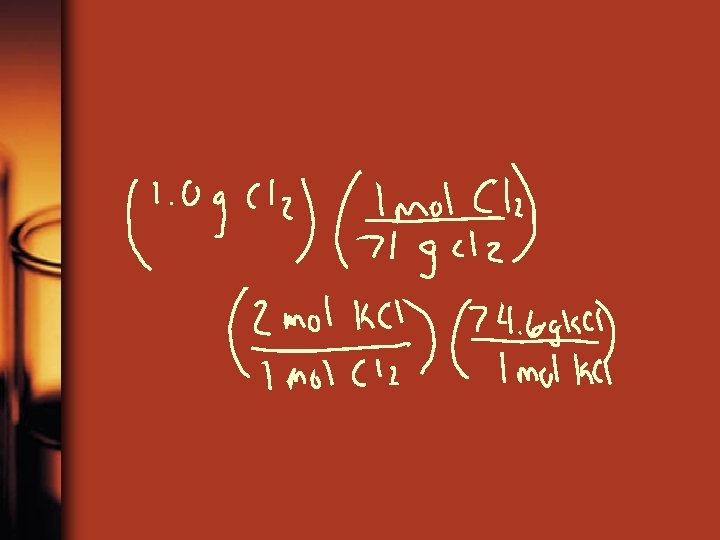










- Slides: 22

Stoichiometry Calculating Reactants and Products

Stoichiometry Defined • The calculation of quantities in chemical reactions • The use of balanced equations to determine numbers of reactants and products used and needed

Another Way To Look at It • Just like a recipe is needed to bake cookies, these balanced equations act as the recipe • What if you need to triple or quadruple the recipe? You need to recalculate your ingredients

Why is this important? • Nearly everything we use is made of chemicals. Chemicals that need to be mixed in exact quantities • Soap, medicine, make-up, clothes, shampoos, fertilizers, and so on

How it Works • You’ll be given a balanced equation • Next you will be asked to determine the amount of reactants needed to yield so much product Or • You may be asked to determine how much product will be yielded if so much of a reactant is added

Example • Given the following equation: 2 KCl. O 3 2 KCl + 3 O 2 1. How many moles of O 2 can be produced by letting 12. 00 moles of KCl. O 3 react?

How to Solve • 1 st and always 1 st use the following conversion guide Units given moles of needed units needed
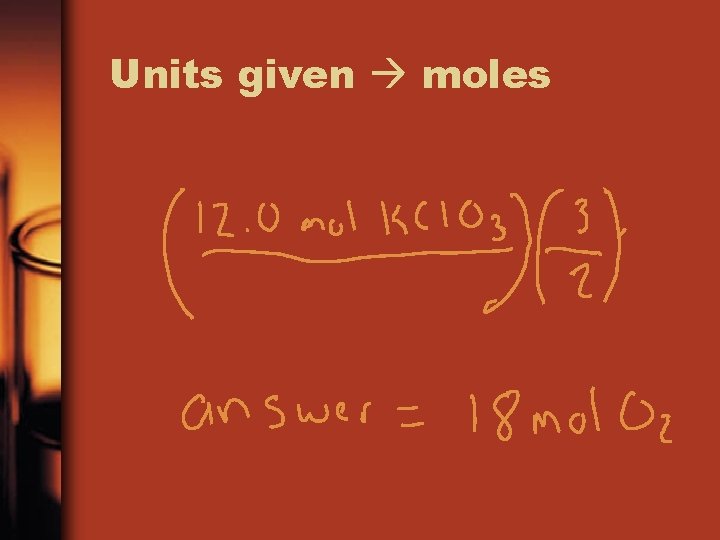
Units given moles

Example 2 • 2 K + Cl 2 2 KCl How many grams of KCl are produced from 2. 50 g of K and excess Cl 2 Also from 1. 00 g of Cl 2 and excess K

Solve • Grams to moles to grams
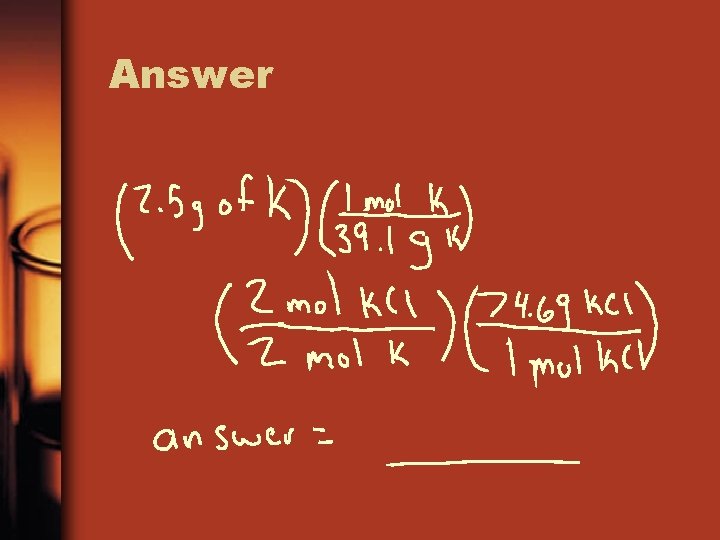
Answer
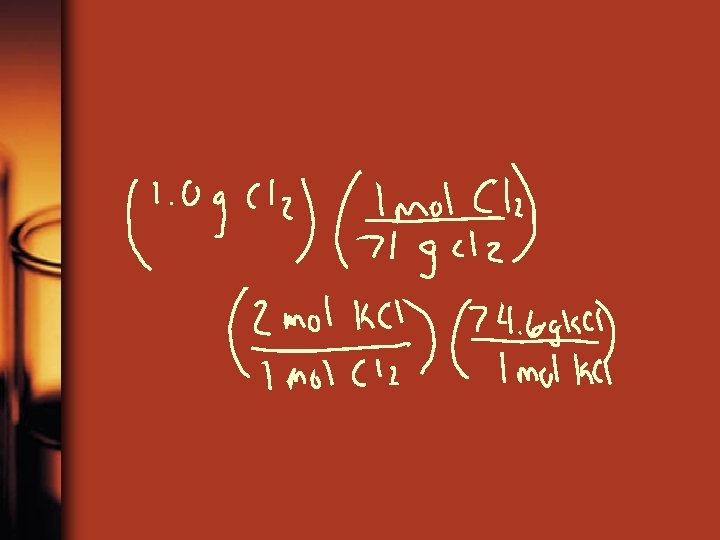


Practice problem • 1 st balance the following Na 2 O + H 2 O Na. OH • Next answer the following 1. How many grams of water are needed to produce 20 grams of product 2. How many molecules of product will be produced from 50 grams of Na 2 O

The Many Possible Conversions • • Grams to grams Grams to moles Moles to grams Liters to moles or grams or liters Grams to molecules Molecules to all the above

Homework • You will have 1 example problem to work and specific instructions on what to do with that answer on ecompanion tonight • Remember mhsfirst. last first

Gases and the Mole • At equal temperatures and equal pressures, equal volumes of gases contain the same number of molecules

STP • Standard temperature and pressure • The values for gasses at STP are as follows – Temp. is 0 o C – Pressure is 1 atm (atmosphere)

Idea. L Gas Law • PV = n. RT – P is pressure (101. 3 k. Pa) – V is volume (? ) – n is number of moles (1) – R is called the ideal gas constant (8. 31) – T is temperature (273 K)

Find V V = n. RT/P V=(1)(8. 31)(273)/101. 3 V= 22. 4

What’s this tell us? • 1 mole of ANY gas at STP is equal to a volume of 22. 4 liters

Example • 2 SO 2 (g) + O 2 (g) 2 SO 3 (g) Assuming STP, how many liters of oxygen are needed to produce 19. 8 L SO 3