STERILIZATION DISINFECTION STERILIZATION Any process that eliminates removes



















































































- Slides: 88

STERILIZATION & DISINFECTION

STERILIZATION Any process that eliminates (removes) or kills all forms of life, including transmissible agents (such as fungi, bacteria, viruses, spore forms, etc. ) present on a surface, contained in a fluid, in medication or in biological culture media. Sterilization can be achieved by applying the proper combinations of heat, chemicals, irradiation, high pressure, and filtration.

DISINFECTION DISINFECTANTS ARE SUBSTANCES THAT ARE APPLIED TO NON-LIVING OBJECTS TO DESTROY MICROORGANISMS THAT ARE LIVING ON THE OBJECTS. DISINFECTION DOES NOT NECESSARILY KILL ALL MICROORGANISMS, ESPECIALLY NONRESISTANT BACTERIAL SPORES; IT IS LESS EFFECTIVE THAN STERILISATION, WHICH IS AN EXTREME PHYSICAL AND/OR CHEMICAL PROCESS THAT KILLS ALL TYPES OF LIFE. . UNLIKE STERILIZATION, DISINFECTION IS NOT SPORICIDAL

A few disinfectants will kill spores with prolonged exposure times (3– 12 hours); these are called chemical sterilants. At similar concentrations but with shorter exposure periods (e. g. , 20 minutes for 2% glutaraldehyde), these same disinfectants will kill all microorganisms except large numbers of bacterial spores; they are called high-level disinfectants.

Chemical Sterilants >2. 4% glutaraldehyde-based formulations 0. 95% glutaraldehyde with 1. 64% phenol 7. 5% stabilized hydrogen peroxide 7. 35% hydrogen peroxide with 0. 23% peracetic acid 0. 2% peracetic acid 0. 08% peracetic acid with 1. 0% hydrogen peroxide. Liquid chemical sterilants reliably produce sterility only if cleaning precedes treatment and if proper guidelines are followed regarding concentration, contact time, temperature, and p. H.

Cleaning is the removal of visible soil (e. g. , organic and inorganic material) from objects and surfaces and normally is accomplished manually or mechanically using water with detergents or enzymatic products. Thorough cleaning is essential before high-level disinfection and sterilization because inorganic and organic materials that remain on the surfaces of instruments interfere with the effectiveness of these processes.

Antisepsis-when a disinfectant is applied to a living tissue. Decontamination removes pathogenic microorganisms from objects so they are safe to handle, use, or discard.

METHODS OF STERILIZATION 1. HEAT STERLIZATION 2. RADIATION STERLIZATION 3. CHEMICAL STERLIZATION

1. HEAT STERLIZATION DRY HEAT MOIST HEAT INCINERATION AUTOCLAVING(steam above 100 deg. C) RED HEAT FLAMING HOT AIR OVEN BOILING WATER(steam at atmospheric pressure)

2. RADIATION STERILIZATION 3. CHEMICAL STERILIZATION UV RAYS GAMMA RAYS FORMALDEHYDE GLUTARALDEHYDE HYDROGEN PEROXIDE ALCOHOLS BLEACHING AGENTS PER ACETIC ACID CHLORINE AND ITS COMPOUNDS IODOPHORES PERACETIC ACID AND HYDROGEN PEROXIDE

METHODS OF DISINFECTION

Factors Affecting The Efficacy Of Disinfection And Sterilization

1. Number and Location of Microorganisms the larger the number of microbes, the more time a germicide needs to destroy all of them. scrupulous cleaning of medical instruments before disinfection and sterilization.

2. Innate Resistance of Microorganisms Intrinsic resistance mechanisms in microorganisms to disinfectants vary. For example, spores are resistant to disinfectants because the spore coat and cortex act as a barrier & mycobacteria have a waxy cell wall that prevents disinfectant. Implicit in all disinfection strategies is the consideration that the most resistant microbial subpopulation controls the sterilization or disinfection time. That is, to destroy the most resistant types of microorganisms (i. e. , bacterial spores), the user needs to employ exposure times and a concentration of germicide needed to achieve complete destruction

3. Concentration and Potency of Disinfectants The more concentrated the disinfectant, the greater its efficacy and the shorter the time necessary to achieve microbial kill. 4. Physical & chemical factors The activity of most disinfectants increases as the TEMPERATURE increases, but too great an increase in temperature causes the disinfectant to degrade and weakens its germicidal activity and thus might produce a potential health hazard.

increase in p. H improves the antimicrobial activity of some disinfectants but decreases the antimicrobial activity of others. RELATIVE HUMIDITY is the single most important factor influencing the activity of gaseous disinfectants/sterilants, such as Et. O, chlorine dioxide, and formaldehyde. WATER HARDNESS (i. e. , high concentration of divalent cations) reduces the rate of kill of certain disinfectants because divalent cations (e. g. , magnesium, calcium) in the hard water interact with the disinfectant to form insoluble precipitates.

5. ORGANIC MATTER serum, blood, pus, or fecal or lubricant material can interfere with the antimicrobial activity by: : As interference occurs by a chemical reaction between the germicide and the organic matter resulting in a complex that is less germicidal or nongermicidal, leaving less of the active germicide available for attacking microorganisms. Chlorine and iodine disinfectants, in particular, are prone to such interaction

6. DURATION OF EXPOSURE Items must be exposed to the germicide for the appropriate minimum contact time. All lumens and channels of endoscopic instruments must contact the disinfectant. Air pockets interfere with the disinfection process, and items that float on the disinfectant will not be disinfected. The disinfectant must be introduced reliably into the internal channels of the device. MCT varies. . but longer better than shorter.

7. BIOFILMS Microorganisms may be protected from disinfectants by production of thick masses of cells and extracellular materials, or biofilms. Biofilms are microbial communities that are tightly attached to surfaces and cannot be easily removed. Once these masses form, microbes within them can be resistant to disinfectants by multiple mechanisms, including physical characteristics of older biofilms, genotypic variation of the bacteria, microbial production of neutralizing enzymes, and physiologic gradients within the biofilm (e. g. , p. H). Bacteria within biofilms are up to 1, 000 times more resistant to antimicrobials than are the same bacteria in suspension.

Hypochlorites can effectively inactivate biofilm bacteria. Their presence can have serious implications for immunocompromised patients and patients who have indwelling medical devices. No products are EPA-registered or FDAcleared for this purpose.

High Level Disinfection Complete elimination of all microorganisms in or on an instrument, except for small numbers of bacterial spores. The FDA definition of high-level disinfection is a sterilant used for a shorter contact time to achieve a 6 -log 10 kill of an appropriate Mycobacterium species. Cleaning followed by high-level disinfection should eliminate enough pathogens to prevent transmission of infection.

Chemical Sterilants. . >2. 4% glutaraldehyde-based formulations 0. 95% glutaraldehyde with 1. 64% phenol 7. 5% stabilized hydrogen peroxide 7. 35% hydrogen peroxide with 0. 23% peracetic acid, 0. 2% peracetic acid, and 0. 08% peracetic acid with 1. 0% hydrogen peroxide. Liquid chemical sterilants reliably produce sterility only if cleaning precedes treatment and if proper guidelines are followed regarding concentration, contact time, temperature, and p. H.

MOST COMMONLY USED: : AUTOCLAVING CHEMICAL METHODS

DISINFECTION OF OPERATION THEATRE

2 steps: : 1. Cleaning with carbolic acid 2. Fumigation-with HCHO vapours -with aldehyde based germicides -Silver & hydrogen peroxide

1. FUMIGATION WITH HCHO VAPOURS Floor is cleaned with warm water, detergent and 5% sodium hypochlorite or 3% phenol followed by fumigation. Seal the room with adhesive tape applied round the edges of doors, windows and ventilator apertures, etc . For each 1000 cubic feet of space place 5000 ml of HCHO solution and 1000 ml of water in an electric boiler with a safety cut out when boiling dry and a time switch. Set the latter to open just before the evaporation is completed. Switch on the boiler, leave the room filled with HCHO and seal the door for 24 hours Then open the doors and windows to allow vapours to disperse and neutralize any residual HCHO with ammonia by exposing 250 ml of SG ammonia per litre of formalin used. CONVENTIONAL METHOD


CARBOLIZATION(Carboxyl acid) & FUMIGATION with Formaldehyde Advantages – Established age old technique Cost effective Disadvantages – a. Carboxylic acid has efficacy as scouring and disinfecting agent b. Time consuming, min 24 hrs turn around time c. Self defeating – OT fumigated with Formaldehyde needs to be force de-aired with unclean air d. Unsafe Formaldehyde is carcinogenic

HAZARDS OF HCHO: : -mutagenic & carcinogenic Irritable to eyes, nose & mucous membranes Asthma attacks can be precipitated. Breathing difficulties.

2. Fumigation with aldehyde based germicides: : Glutaraldehyde & formaldehyde (200 ml in 10 liters of water i. e 2%) through fogging machine is the commonly used procedure. Advantages- Effective Disadvantages – a. Leaves sticky residue because of surfactant base b. Self defeating – OT fumigated with Formaldehyde needs to be force de-aired with unclean air c. Unsafe- Formaldehyde has been identified as a carcinogen

3. Silver (Ag) and Hydrogen Peroxide (H 2 O 2) Advantages – a. Has deep penetrating capability b. Has no known resistant strains c. Effective against Bacteria, Viruses, Mycobacteria, Amoeba, Fungi and spore forming organisms NEW METHOD

A fogging system Droplets~7 microns thrust by a motor running at 20, 000 RPM to a distance of 15 m Solution Ecoshield – 11% Hydrogen Peroxide and 0. 01% dilute sliver nitrate

NEW METHOD: : FUMIGATION WITH ECOSHIELD: : Brand name. Marketed by Johnson and Johnson. A solution based on stabilised hydrogen peroxide with silver nitrate. It has been seen to be effective against bacteria, virus, fungi, protozoa, mycobacteria, bacteriophages and biofilms. Works best with an ultra low volume fogger that releases droplets of seven to 20 microns. It traps suspended particulate matters and kills the microorganisms. cost-effective option that could be used by hospitals that do not have sophisticated filtering systems like the high efficiency particulate air filter or laminar filter.


Ecoshield helps disinfect operations theatres within two hours. This can ensure a quicker turnaround time. HCHO takes upto 24 hours and is a known irritant, mutagenic and carcinogenic.

AT HIHT: : Fumigation with ecoshield Routinely, at the end of the day. Normally, 3 -4 noninfected cases can be performed in an o. t. sterlized once by fumigation.

BUT in infected cases (HIV +ve, Hbs. Ag +ve. . ) Immediate fumigation of the OT is done after the operation All disposables are discarded.

A Rational Approach To Disinfection And Sterilization EH Spaulding believed that how an object will be disinfected depended on the object’s intended use. CRITICAL - objects which enter normally sterile tissue or the vascular system or through which blood flows should be sterile. SEMICRITICAL - objects that touch mucous membranes or skin that is not intact require a disinfection process (high-level disinfection [HLD]) that kills all microorganisms but high numbers of bacterial spores. NONCRITICAL -objects that touch only intact skin require low-level disinfection (or non-germicidal detergent).

Critical Objects Classification: Critical objects enter normally sterile tissue or vascular system, or through which blood flows. Object: Sterility. Level germicidal action: Kill all microorganisms, including bacterial spores. Examples: surgical instruments, cardiac and urinary catheters, implants, and ultrasound probes used in sterile body cavities; etc. Method: Steam, gas, hydrogen peroxide plasma, ozone or chemical sterilization.

Sterilization of “Critical Objects” Steam sterilization Hydrogen peroxide gas plasma Ethylene oxide Peracetic acid-chemical sterilization Ozone Vaporized hydrogen peroxide Steam formaldehyde

Chemical Sterilization of “Critical Objects” Glutaraldehyde (> 2. 0%) Hydrogen peroxide-HP (7. 5%) Peracetic acid-PA (0. 2%) HP (1. 0%) and PA (0. 08%) HP (7. 5%) and PA (0. 23%) Glut (1. 12%) and Phenol/phenate (1. 93%) ______________________ ___ Exposure time per manufacturers’ recommendations

“Semicritical” Patient Care Objects Classification: Object: Germicidal action: Examples: Method: Semicritical objects come in contact with mucous membranes or skin that is not intact. Free of all microorganisms except high numbers of bacterial spores. kills allmicroorganisms except high numbers of bacterial spores. Respiratory therapy and anesthesia equipment, GI endoscopes, endocavitary probes, etc. High-level disinfection

Semicritical Items Endoscopes Respiratory therapy equipment Anesthesia equipment Endocavitary probes Tonometers Diaphragm fitting rings

High Level Disinfection of “Semicritical Objects” Exposure Time > 12 m-30 m, 20 o. C Germicide Concentration_____ Glutaraldehyde > 2. 0% Ortho-phthalaldehyde (12 m) 0. 55% Hydrogen peroxide* 7. 5% Hydrogen peroxide and peracetic acid* 1. 0%/0. 08% Hydrogen peroxide and peracetic acid* 7. 5%/0. 23% Hypochlorite (free chlorine)* 650 -675 ppm Glut and phenol/phenate** *May cause cosmetic and functional damage; **efficacy not verified

“Noncritical” Patient Care Objects Classification: Noncritical objects will not come in contact with mucous membranes or skin that is not intact. Object: Can be expected to be contaminated with some microorganisms. Level germicidal action: Kill vegetative bacteria, fungi and lipid viruses. Examples: Bedpans; crutches; bed rails; EKG leads; bedside tables; walls, floors and furniture. Method: Low-level disinfection (or detergent for housekeeping surfaces)

Low-Level Disinfection for “Noncritical” Objects Exposure time > 1 min Ethyl or isopropyl alcohol Chlorine Phenols Iodophores Accelerated hydrogen peroxide _________________________

Items must be cleaned using water with detergents or enzymatic cleaners before processing. Cleaning reduces the bioburden and removes foreign material (organic residue and inorganic salts) that interferes with the sterilization process. Cleaning and decontamination should be done as soon as possible after the items have been used as soiled materials become dried onto the instruments.

Rinsing endoscopes and flushing channels with sterile water, filtered water, or tap water will prevent adverse effects associated with disinfectant retained in the endoscope (e. g. , disinfectant-induced colitis) Items can be rinsed and flushed using sterile water after high-level disinfection to prevent contamination with organisms in tap water, such as nontuberculous mycobacteria, Legionella or gramnegative bacilli such as Pseudomonas. Alternatively, a tapwater or filtered water (0. 2 m filter) rinse should be followed by an alcohol rinse. After rinsing, items should be dried and stored (e. g. , packaged) in a manner that protects them from recontamination.

Noncritical items Intact skin acts as an effective barrier to most microorganisms; therefore, the sterility of items coming in contact with intact skin is "not critical. Most noncritical reusable items may be decontaminated where they are used and do not need to be transported to a central processing area.

Noncritcal environmental surfaces include bed rails, food utensils, bedside tables, patient furniture and floors. Noncritical environmental surfaces frequently touched by hand (e. g. , bedside tables, bed rails) potentially could contribute to secondary transmission by contaminating hands of health-care workers or by contacting medical equipment that subsequently contacts patients. Mops and reusable cleaning cloths are regularly used to achieve low-level disinfection. The water-disinfectant mixture should be changed regularly (e. g. , after every three to four rooms, at no longer than 60 -minute intervals), the mopping procedure actually can spread heavy microbial contamination throughout the health-care facility.

Disinfection Of Healthcare Equipment

AT HIHT: : All the metallic instruments (non-infected) Washed with Running water Autoclaved (121 deg. C for 60 mins)

Disinfection of HBV-, HCV-, HIV- or TB-Contaminated Devices Instruments and gowns-dipped in sodium hypochlorite (4%) soln for 90 mins. -washed with running water -Autoclave(121 deg. C for 60 mins. )

CIDEX- Activated Dialdehyde- a 2. 4% alkaline glutaraldehyde solution Fast and effective way to disinfect and sterilize medical devices and instruments. 14 day use destroys 99. 8% of Mycobacterium tuberculosis in 45 minutes at 25 degrees Celsius Cidex Plus (a high-level disinfectant, 20 minutes at 25 degrees Celsius) is a 3. 4% glutaraldehyde solution. Cidex OPA (ortho-phthalaldehyde) is compatible with a wide range of medical devices. Requiring no activation or mixing, Cidex OPA destroys Mycobacteruim tuberculosis in 12 minutes at 20 degrees Celsius.


CIDEX (contd. ) STERILIZATION-12 -14 hrs DISINFECTION-1520 MINS Heat sensitive & disposable materials -corrugated drains -suction tubes -Catheters -Laparoscopes etc.

Disinfection of laparoscopes/endoscopes endoscope disinfection or sterilization with a liquid chemical sterilant involves steps after leak testing: five

1. Clean: mechanically clean internal and external surfaces, including brushing internal channels and flushing each internal channel with water and a detergent or enzymatic cleaners (leak testing is recommended for endoscopes before immersion). 2. Disinfect: immerse endoscope in high-level disinfectant (or chemical sterilant) and perfuse (eliminates air pockets and ensures contact of the germicide with the internal channels) disinfectant into all accessible channels, such as the suction/biopsy channel and air/water channel and expose for a time recommended for specific products. 3. Rinse: rinse the endoscope and all channels with sterile water, filtered water or tap water

4. Dry: rinse the insertion tube and inner channels with alcohol, and dry with air after disinfection and before storage. 5. Store: store the endoscope in a way that prevents recontamination and promotes drying (e. g. , hung vertically).

AT HIHT Laparoscopes/endoscopes Washed with running water Air dried Dipped in CIDEX and kept Remove after 12 -14 hours(sterilization) Dried in air Stored in their respective boxes (after oiling) with FORMALIN TABLETS.

FORMALIN CHAMBERS

10 -15 tabs of HCHO are kept rolled up in a gauze piece in the lowermost part of the chambers. Plastics, ventouse cups, suction tubes, laparoscopic instruments, sharps etc. Change the tablets after 7 -10 days.

Cervical Diaphragm Fitting Rings a soap-and-water wash followed by a 15 minute immersion in 70% alcohol. This disinfection method is adequate to inactivate HIV, HBV, and HSV even though alcohols are not classified as high-level disinfectants because their activity against picornaviruses is somewhat limited

Endocavitary probes Vaginal probes A vaginal probe and all endocavitary probes without a probe cover are semicritical devices because they have direct contact with mucous membranes. Use of a new condom/probe cover for the probe for each patient, and because condoms/probe covers can fail , the probe also should be high-level disinfected. Condoms have been found superior to commercially available probe covers for covering the ultrasound probe. 2% glutaraldehyde for high-level disinfection of contaminated transvaginal transducers.

Inactivation of Clostridium difficile Source-not determined possible sources of infection –environment & hands of health workers spore-production increases on exposure to nonchlorine-based cleaning agents and the spores are more resistant than vegetative cells to commonly used surface disinfectants. use of dilute solutions of hypochlorite for routine environmental disinfection of rooms of patients with C. difficile-associated diarrhea or colitis. regular bleach can inactivate 106 C. difficile spores in <10 minutes. Data demonstrate that 2% glutaraldehyde and peracetic acid reliably kill C. difficile spores using exposure times of 5– 20 minutes.

Disinfection in Ambulatory Care, Home Care, and the Home The healthcare provider is responsible for providing the responsible family member information about infection-control procedures to follow in the home, including hand hygiene, proper cleaning and disinfection of equipment, and safe storage of cleaned and disinfected devices. APIC recommends that reusable objects (e. g. , tracheostomy tubes) that touch mucous membranes be disinfected by immersion in 3% hydrogen peroxide for 30 minutes. Additionally, a 1: 50 dilution of 5. 25%– 6. 15% sodium hypochlorite (household bleach) for 5 minutes should be effective. Noncritical items (e. g. , blood pressure cuffs, crutches) can be cleaned with a detergent.

Environmentally safe alternatives (e. g. , ammonia, baking soda, vinegar, Borax, liquid detergent) are not registered with EPA and should not be used for disinfecting because they are ineffective against S. aureus. Borax, baking soda, and detergents also are ineffective against Salmonella Typhi and E. coli; however, undiluted vinegar and ammonia are effective against S. Typhi and E. coli. Many sites in the home kitchen and bathroom are microbially contaminated, use of hypochlorites markedly reduces bacteria and good standards of hygiene (e. g. , food hygiene, hand hygiene) can help reduce infections in the home.

Susceptibility of Antibiotic. Resistant Bacteria to Disinfectants Several studies have found antibiotic-resistant hospital strains of common healthcare-associated pathogens (i. e. , Enterococcus, P. aeruginosa, Klebsiella pneumoniae, E. coli, S. aureus, and S. epidermidis) to be equally susceptible to disinfectants as antibiotic-sensitive strains. On the basis of these data, routine disinfection and housekeeping protocols do not need to be altered because of antibiotic resistance provided the disinfection method is effective. The rotational use of disinfectants in some environments (e. g. , pharmacy production units) has been recommended and practiced in an attempt to prevent development of resistant microbes.

Surface Disinfection. Hospital floors become contaminated with microorganisms from settling airborne bacteria: by contact with shoes, wheels, and other objects; and occasionally by spills. Phenolic disinfectant (94%– 99. 9% reduction) Detergents become contaminated and result in seeding the patient's environment with bacteria. Surfaces contaminated with blood and other potentially infectious materials (e. g. , amniotic, pleural fluid) be disinfected Using a single product throughout the facility can simplify both training and appropriate practice

Heat is the most reliable treatment of cleaning cloths as a detergent washing followed by drying at 80 o. C for 2 hours produced elimination of contamination. Alternatively, immersing the cloth in hypochlorite for 2 minutes Some hospitals have begun using a new mopping technique involving microfiber materials to clean floors. The positively charged microfibers attract dust (which has a negative charge) and are more absorbent than a conventional, cotton-loop mop.

Air Disinfection Disinfectant spray-fog techniques(. , filtration, ultraviolet germicidal irradiation) for antimicrobial control in hospital rooms has been used. This technique of spraying of disinfectants is an unsatisfactory method of decontaminating air and surfaces and is not recommended for general infection control in routine patient-care areas

Microbial Contamination of Disinfectants Members of the genus Pseudomonas (e. g. , P. aeruginosa) are the Germicides used as disinfectants that were reported to have been contaminated include chlorhexidine, quaternary ammonium compounds, most frequent isolates from contaminated disinfectants. phenolics control measures The following should be instituted to reduce the frequency of bacterial growth in disinfectants: : -----disinfectants should not be diluted; those that are diluted must be prepared correctly to achieve the manufacturers' recommended use-dilution. -----infection-control professionals must learn from the literature what inappropriate activities result in extrinsic contamination (i. e. , at the point of use) of germicides and train users to prevent reinfection. -----stock solutions of germicides must be stored as indicated on the product label.

BIOMEDICAL WASTE

CLASSIFICATION

Non hazardous: approximately 75 -90% of the biomedical waste is non-hazardous and as harmless as any other municipal waste. (E. G, Plastic, Glass, Cardboared, etc Hazardous waste: 10 -25% is hazardous and can be injurious to humans or animals and deleterious to environment. It is important to realise that if both these types are mixed together then the whole waste becomes harmful.

Classification and management Category Waste Type Human Wastes Category 1 (Tissues, organs, body parts Category 2 Animal Waste Microbiology and Category 3 Biotechnology waste Category 4 Sharps Category 5 Discarded Medicines and Cytotoxic Drugs Treatment and Disposal Method Incineration / deep burial Autoclave/microwave/incineration Disinfection (chemical treatment)+/autoclaving/microwaving and mutilation shredding Incineration/ destruction and drugs disposal in secured landfills

Category Waste Type Contaminated solid Category 6 waste Treatment and Disposal Method Incineration/autoclaving / microwaving Disinfection by chemical treatment+ Solid waste (disposable Category 7 microwaving/autoclaving & items other than sharps) mutilation shredding Liquid waste (generated from laboratory Disinfection by chemical treatment+ Category 8 washing, cleaning, and discharge into the drains housekeeping and disinfecting activity) Category 9 Incineration ash Disposal in municipal landfill Category 10 Chemical Wastes Chemical Treatment + and discharge in to drain for liquids and secured landfill for solids

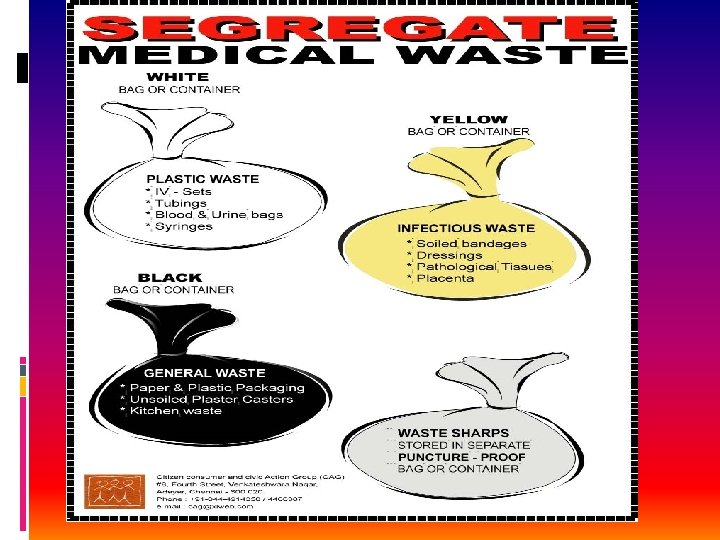
Color coding YELLOW RED Type of container PLASTIC BAG Disinfected container / plastic bag Blue/white Plastic bag/ translucent Puncture proof BLACK Plastic bag Waste category option treatment Cat 1, 2, 3, 6 Incineration / deep burial Cat 3, 6, 7 Autoclave/microwave /chemical treatment Cat 4, 7 Autoclave/microwave/chemical treatment Cat 5, 9, 10 Disposal in secure landfill


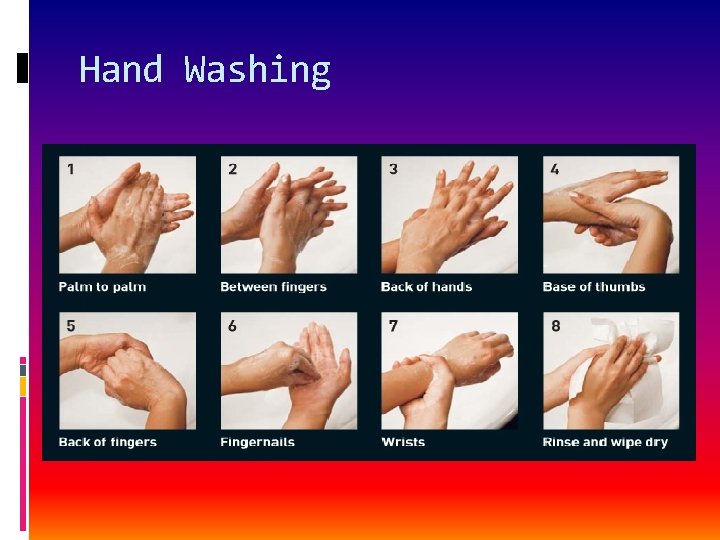
Hand Washing

Universal Precautions "Universal precautions, " as defined by CDC, are a set of precautions designed to prevent transmission of human immunodeficiency virus (HIV), hepatitis B virus (HBV), and other bloodborne pathogens when providing first aid or healthcare. Under universal precautions, blood and certain body fluids of all patients are considered potentially infectious for HIV, HBV and other bloodborne pathogens.

5 Steps of Universal Precautions Education Hand washing Use of protective barriers (Personal Protective Equipment (PPE)) Cleaning of contaminated surfaces Safe handling/disposal of contaminated material

1. Education The employer must provide training to protect the health and safety of the worker and provide for the safe handling and disposal of biological agents. Hygienic (washroom) facilities with water, soap and disposable towels or a hot air hand dryer must be provided at all our worksites. Protective surgical gloves (latex or vinyl) must be provided by the employer for workers who may be exposed to biological agents (i. e. body fluids from other people or from animals).

2. Hand Washing Frequent hand washing is one effective way to prevent the spread of infectious diseases in a workplace. Wash hands frequently and thoroughly, especially after contact with any body fluid or a contaminated surface. Wet, soap and lather hands for at least ten seconds. Wash and scrub under fingernails and cuticles with a small brush. Rinse hands thoroughly and dry.

3. Protective Barriers 1. Gloves Surgical quality latex or vinyl gloves must be provided by the employer for all contact with blood, body fluids and feces. Gloves are also necessary for disinfecting contaminated surfaces and disposing of used materials and biological waste. Wash hands thoroughly with warm water and soap after removing gloves. Change gloves after each task or exposure and dispose as contaminated waste. 2. Personal Protective Equipment Protective eye glasses and a mask must be provided and used where blood, body fluids or feces are likely to splash on the mucous membranes of the eyes, nose or mouth. Gowns, lab coats or aprons must be provided and worn where clothing is likely to be soiled. 3. Resuscitation Devices To minimize your exposure during emergency mouth-to-mouth resuscitation, mouthpieces or other resuscitation devices should be provided. Resuscitation devices should be a disposable type and available in every first aid kit.

4. Cleaning and disinfecting of contaminated areas Wear gloves and use disposable towels or other means of cleaning that will ensure against direct contact with blood, body fluids or feces. Decontaminate the area with an approved germicide or 1: 100 solution of household bleach. All used equipment must be thoroughly washed and disinfected.

5. Safe handling/disposal of contaminated material Special precautions must be observed when disposing of biological waste and materials contaminated with biological waste. The employer must have a written Disposal of Contaminated Waste Procedure for the safe disposal of contaminated waste which should include the following: Dispose of biological waste in a puncture-resistant container lined with a leak -proof plastic bag. Post a biological waste symbol on the container. Consider all biological waste as infectious. Wear puncture-resistant gloves and handle all contaminated wastes carefully to prevent body contact. Hold only the outside of the container when emptying it. Never reach into the container. Do not load the container beyond its capacity or compact the contents. Compaction may lead to additional contamination of the work area. Never mix biological waste with regular trash. Any object that could cut or puncture the skin such as needles or broken glass may carry infectious material and should be handled with caution. Dispose of 'sharps' in unbreakable, non-pierceable containers that have a lid. Never place 'sharps' in the regular trash.
